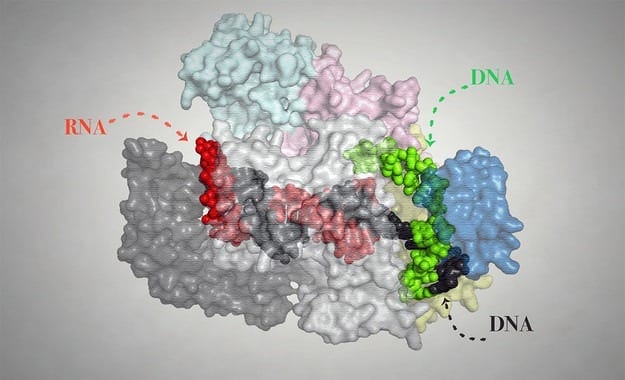
In an era of widespread genetic sequencing, the ability to edit and alter an organism’s DNA is a powerful way to explore the information within and how it guides biological function.
A paper from the University of Wisconsin-Madison in the August issue of the journal GENETICS takes genome editing to a new level in fruit flies, demonstrating a remarkable level of fine control and, importantly, the transmission of those engineered genetic changes across generations.
Both features are key for driving the utility and spread of an approach that promises to give researchers new insights into the basic workings of biological systems, including embryonic development, nervous system function, and the understanding of human disease.
“Genome engineering allows you to change gene function in a very targeted way, so you can probe function at a level of detail” that wasn’t previously possible, says Melissa Harrison, an assistant professor of biomolecular chemistry in the UW-Madison School of Medicine and Public Health and one of the three senior authors of the new study.
Disrupting individual genes has long been used as a way to study their roles in biological function and disease. The new approach, based on molecules that drive a type of bacterial immune response, provides a technical advance that allows scientists to readily engineer genetic sequences in very detailed ways, including adding or removing short bits of DNA in chosen locations, introducing specific mutations, adding trackable tags, or changing the sequences that regulate when or where a gene is active.
The approach used in the new study, called the CRISPR RNA/Cas9 system, has developed unusually fast. First reported just one year ago by scientists at the Howard Hughes Medical Institute and University of California, Berkeley, it has already been applied to most traditional biological model systems, including yeast, zebrafish, mice, the nematode C. elegans, and human cells. The Wisconsin paper was the first to describe it in fruit flies and to show that the resulting genetic changes could be passed from one generation to the next.
“There was a need in the community to have a technique that you could use to generate targeted mutations,” says Jill Wildonger, a UW-Madison assistant professor of biochemistry and another senior author of the paper. “The need was there and this was the technical advance that everyone had been waiting for.”
“The reason this has progressed so quickly is that many researchers — us included — were working on other, more complicated, approaches to do exactly the same thing when this came out,” adds genetics assistant professor Kate O’Connor-Giles, the third senior author. “This is invaluable for anyone wanting to study gene function in any organism and it is also likely to be transferable to the clinical realm and gene therapy.”
The CRISPR RNA/Cas9 system directs a DNA-clipping enzyme called Cas9 to snip the DNA at a targeted sequence. This cut then stimulates the cell’s existing DNA repair machinery to fill in the break while integrating the desired genetic tweaks. The process can be tailored to edit down to the level of a single base pair — the rough equivalent of changing a single letter in a document with a word processor.
The broad applicability of the system is aided by a relatively simple design that can be customized through creation of a short RNA sequence to target a specific sequence in the genome to generate the desired changes. Previous genome editing methods have relied on making custom proteins, which is costly and slow.
“This is so readily transferable that it’s highly likely to enable gene knockout and other genome modifications in any organism,” including those that have not previously been used for laboratory work, says O’Connor-Giles. “It’s going to turn non-model organisms into genetic model organisms.”
That ease may also pay off in the clinic. “It can be very difficult and time-consuming to generate models to study all the gene variants associated with human diseases,” says Wildonger. “With this genome editing approach, if we work in collaboration with a clinician to find [clinically relevant] mutations, we can rapidly translate these into a fruit fly model to see what’s happening at the cellular and molecular level.”
The Latest Bing News on:
Genome editing
- What It'll Take to Create 21st-Century Mammoths, Dodos, and Thylacineson April 27, 2024 at 8:16 am
We don’t fully understand how to create an artificial placenta at the moment. We don’t really understand the intricacies of the developmental process. This is all information that we will learn along ...
- Genome Editing Genome Engineering Market Role of Artificial Intelligence in Survey Design Innovations and Trendson April 25, 2024 at 8:30 pm
The latest research report from Report Ocean titled “Genome Editing Genome Engineering Market: Trends Analysis from 2024 to 2032”. This report delivers accurate economic projections, worldwide ...
- This AI Just Designed a More Precise CRISPR Gene Editor for Human Cells From Scratchon April 25, 2024 at 1:25 pm
Based on large language models—the tech behind the popular ChatGPT—Profluent's AI designed a new gene editor and put it to work in human cells.
- Intellia (NTLA) to Report Q1 Earnings: What's in the Cards?on April 25, 2024 at 8:21 am
We expect investors to focus on Intellia Therapeutics, Inc.’s NTLA progress with the development of its portfolio of CRISPR-based gene-edited therapies when it reports first-quarter 2024 earnings.
- Regeneron, Mammoth Launch In Vivo CRISPR Gene Editing Collaborationon April 25, 2024 at 8:00 am
Regeneron Pharmaceuticals will use Mammoth Biosciences’ CRISPR-based ultracompact gene editing platform to develop in vivo therapies for tissues and cell types beyond the liver.
- Advancing crop breeding through targeted genome modificationon April 25, 2024 at 6:36 am
Modern crop breeding is entering a new era of genome design, led by genome editing technologies as mainstream tools for targeted genome modification. In a review published in Nature Reviews Genetics, ...
- AI-designed gene editing tools successfully modify human DNAon April 24, 2024 at 5:51 am
Medically, AI is helping us with everything from identifying abnormal heart rhythms before they happen to spotting skin cancer. But do we really need it to get involved with our genome? Protein-design ...
- GU Medical Center Distinguished Scientist Seminar Series Illuminates Uses of Gene Editing in Cancer Researchon April 23, 2024 at 5:00 pm
A researcher highlighted gene editing and its therapeutic applications in a talk April 18. Raj Chari, director of the Genome Modification Core Laboratory at the Frederick National Laboratory for ...
- Probing the Inner World of Cells Provides Unexpected Boost to Prime Editingon April 23, 2024 at 4:02 am
While probing the inner workings of cells, Dr. Britt Adamson, assistant professor of molecular biology at Princeton University, and colleagues discovered a way to boost the efficiency of prime editing ...
- Profluent Successfully Edits Human Genome with OpenCRISPR-1, the World’s First AI-Created and Open-Source Gene Editoron April 22, 2024 at 9:30 am
With this launch, Profluent demonstrates the first successful precision editing of the human genome with customizable gene editors designed from scratch with AI. OpenCRISPR-1 is an AI-created gene ...
The Latest Google Headlines on:
Genome editing
[google_news title=”” keyword=”Genome editing” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
The Latest Bing News on:
CRISPR RNA/Cas9
- Experts develop way to harness CRISPR technology to deal with antimicrobial resistanceon April 26, 2024 at 3:10 pm
Antimicrobial resistance (AMR) is continuing to increase globally, with rates of AMR in most pathogens increasing and threatening a future in which every day medical procedures may no longer be ...
- Buy Rating on Crispr Therapeutics AG Backed by Exa-cel’s Precision and Favorable Safety Profileon April 25, 2024 at 11:24 pm
Salim Syed, an analyst from Mizuho Securities, maintained the Buy rating on Crispr Therapeutics AG (CRSP – Research Report). The ...
- This AI Just Designed a More Precise CRISPR Gene Editor for Human Cells From Scratchon April 25, 2024 at 1:25 pm
Based on large language models—the tech behind the popular ChatGPT—Profluent's AI designed a new gene editor and put it to work in human cells.
- Regeneron Pays Mammoth $100M to Access CRISPR Enzymes for AAV Deliveryon April 25, 2024 at 8:18 am
Regeneron will use Mammoth Biosciences’ tiny Cas enzymes to deliver in vivo CRISPR-based gene editing therapies to tissues and cell types beyond the liver, the companies announced Thursday.
- Regeneron, Mammoth Launch In Vivo CRISPR Gene Editing Collaborationon April 25, 2024 at 8:00 am
Regeneron Pharmaceuticals will use Mammoth Biosciences’ CRISPR-based ultracompact gene editing platform to develop in vivo therapies for tissues and cell types beyond the liver.
- GU Medical Center Distinguished Scientist Seminar Series Illuminates Uses of Gene Editing in Cancer Researchon April 23, 2024 at 5:00 pm
an RNA sequence called a guide RNA will help Cas9, a specialized enzyme, navigate to the site where it must cleave the virus’ DNA. Chari said the discovery of CRISPR-Cas9 revolutionized gene editing. ...
- Profluent combines LLMs and CRISPR for open-source AI gene editing projecton April 23, 2024 at 11:00 am
Profluent has staked a claim at the intersection of some of the buzziest terms in biopharma. | Profluent has staked a claim at the intersection of some of the buzziest terms in biopharma. The protein ...
- A small factor makes a big impact on genome editingon April 21, 2024 at 5:00 pm
Prime editing systems minimally consist of two components: a modified version of the protein element of CRISPR/Cas9 and a ribonucleic acid (RNA) molecule called a pegRNA. These components work ...
- CRISPR Therapeutics Stock (NASDAQ:CRSP) Pushes Lower, But Fundamentals Are Intacton April 19, 2024 at 4:31 pm
CRISPR Therapeutics (NASDAQ:CRSP) stock has been slowly heading lower in recent months, and I don’t think that’s fair. Having reached highs near ...
- MEGA CRISPR: Engineering Better Immunotherapies with RNA Editingon March 31, 2024 at 5:00 pm
3 However, CRISPR-associated nuclease 9 (Cas9)-based genome cutting tools face unique safety ... which could possibly only be addressed if we find methods to engineer RNA.” “Our years of experience on ...
The Latest Google Headlines on:
CRISPR RNA/Cas9
[google_news title=”” keyword=”CRISPR RNA/Cas9″ num_posts=”10″ blurb_length=”0″ show_thumb=”left”]