CREDIT
Debra Weinstein, Sao-Mai Nguyen-Mau, and Vincent Lee
New discovery could help prevent the formation of infectious bacterial films on hospital equipment
Bacteria are best known as free-living single cells, but in reality their lives are much more complex. To survive in harsh environments, many species of bacteria will band together and form a biofilm–a collection of cells held together by a tough web of fibers that offers protection from all manner of threats, including antibiotics. A familiar biofilm is the dental plaque that forms on teeth between brushings, but biofilms can form almost anywhere given the right conditions.
Biofilms are a huge problem in the health care industry. When disease-causing bacteria establish a biofilm on sensitive equipment, it can be impossible to sterilize the devices, raising rates of infection and necessitating expensive replacements. So researchers look for ways to break down the defenses of biofilms to prevent them from establishing a foothold.
Now, a University of Maryland-led team has found an important link in the biofilm formation process: an enzyme that shuts down the signals that bacteria use to form a biofilm. The findings, reported in the August 24, 2015 Early Online Edition of the Proceedings of the National Academy of Sciences, have far-reaching implications for the development of new treatments, and could one day help make biofilm-related complications a distant memory.
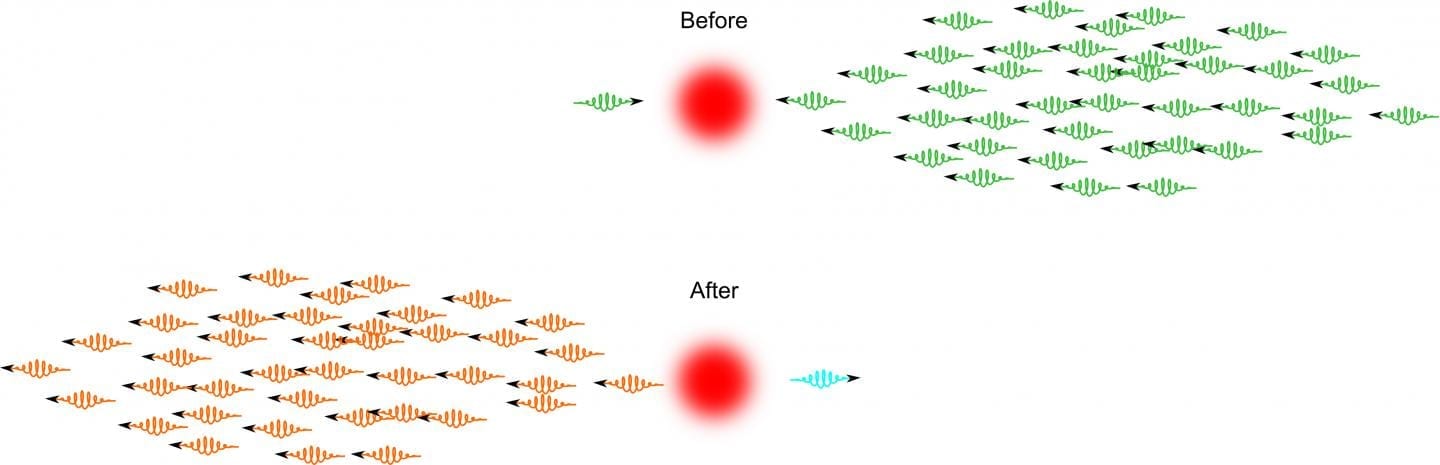
“Bacteria form biofilms because they sense a change in their environment. They do this by generating a signaling molecule, which binds to a receptor that turns on the response,” said Mona Orr, the lead author of the study and a UMD biological sciences graduate student. “But you need a way to turn off the switch–to remove the signal when it’s no longer needed. We’ve identified the enzyme that completes the process of turning off the switch.”
The well-known switch that activates biofilm formation is a signaling molecule called Cyclic-di-GMP, also known as c-di-GMP. Many species of disease-causing bacteria use c-di-GMP to signal the formation of biofilms, including Escherichia coli, Salmonella enterica and Vibrio cholerae.
But Orr and her colleagues are the first to identify the molecule that completes the process of clearing c-di-GMP from the cell, thus ending the biofilm signaling process. The molecule is an enzyme called oligoribonuclease, and much like c-di-GMP, oligoribonuclease is also common among disease-causing bacterial species.
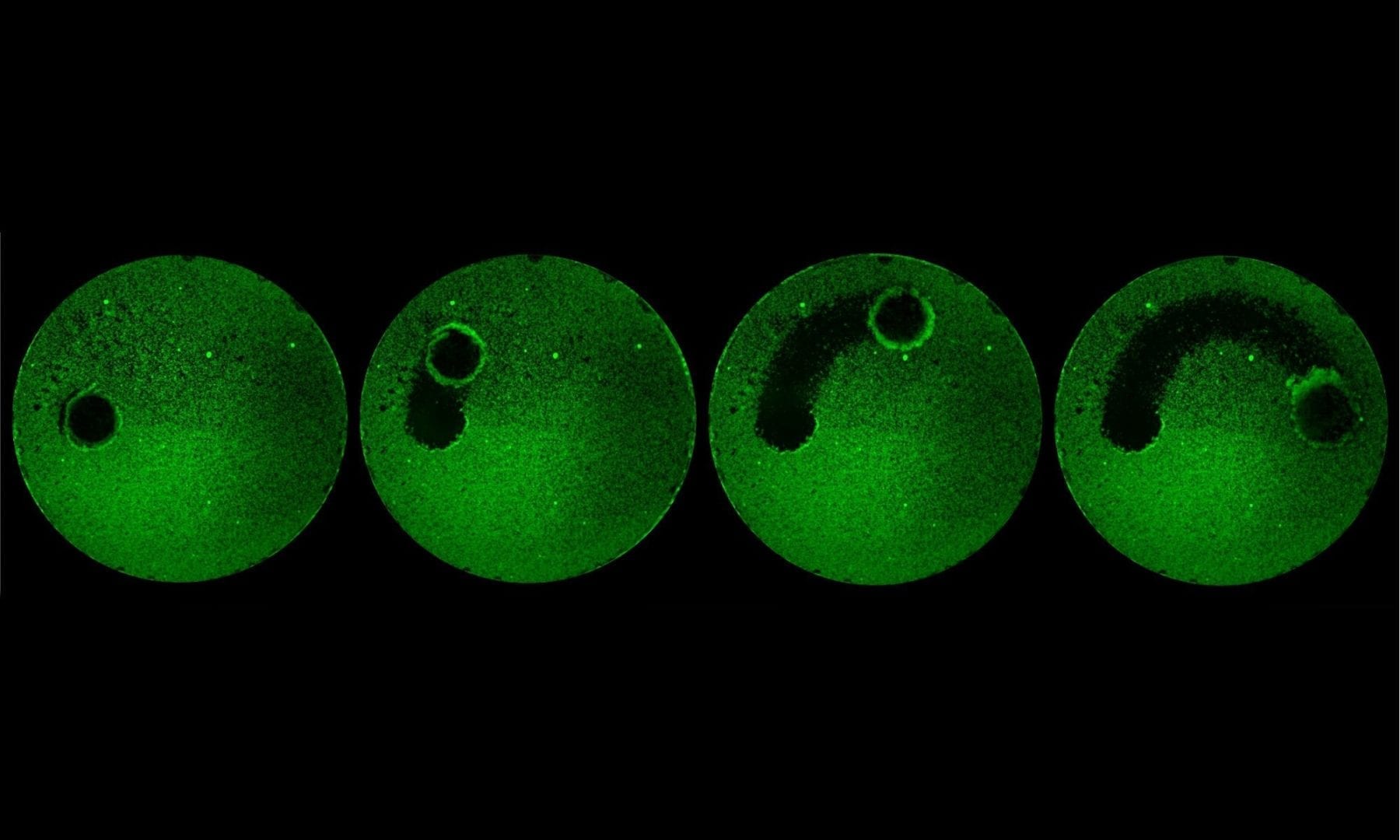
The team studied the process in the bacteria Pseudomonas aeruginosa, a common species known to cause infections in hospital patients. But because of the genetic and physiological similarities between P. aeruginosa and other infectious species, the researchers believe that oligoribonuclease serves the same function across a wide variety of bacteria.
“You can think of this process in terms of water filling a sink. The rate of water from the faucet is just as important as the size of the drain in determining the level of water in the sink,” said Vincent Lee, a co-author of the study and an associate professor in the UMD Department of Cell Biology and Molecular Genetics and the Maryland Pathogen Research Institute. “The level of c-di-GMP in the cell is analogous to the amount of water in the sink. Because no one knew what the drain was, our findings create a complete picture of the signaling process.”
Orr, Lee and their colleagues from the UMD Department of Chemistry and Biochemistry and Michigan State University focused their work on P. aeruginosa because it is well studied and can survive under a variety of conditions, making it notoriously difficult to control. Contact lens wearers might already be familiar with P. aeruginosa, as it commonly forms infectious, green-tinted biofilms on older lenses or those that have not been cleaned properly.
The team found that oligoribonuclease is necessary for the second of a two-step process. The first, which converts c-di-GMP into an intermediate molecule called pGpG, was already known. Orr, Lee and their colleagues have now filled in the important second step in this process: oligoribonuclease breaks apart pGpG and thus completely shuts off the signaling pathway.
The result suggests that oligoribonuclease could be used to help design new antibiotics, disinfectants, and surface treatments to control biofilms. Such measures could prevent infections and preclude the need for frequent replacement of expensive hospital equipment. Because biofilms can also form on implanted medical devices, such as pacemakers and synthetic joints, effective treatments against biofilms could eliminate the need for costly and risky replacement surgeries.
Read more: UMD-led study identifies the off switch for biofilm formation
The Latest on: Biofilm
[google_news title=”” keyword=”biofilm” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Biofilm
- 6 Reasons Why Your Shower Drain May Smell According to Plumbing Proson May 7, 2024 at 5:14 am
See the folks at Budget Heating, Cooling, & Plumbing put a Zip It drain cleaner to use in this video: Related: These Kitchen Staples Will Unclog a Bathtub Drain Quickly Without Harsh Chemicals Biofilm ...
- Hulu to Remove 71 Titles In May - Here's the 5 You Should Watch Before They Leaveon May 6, 2024 at 4:59 pm
Leaving May 31. Elvis (2022) Austin Butler is sensational as the King of Rock n’ Roll in this raucous Baz Luhrmann-directed biofilm which chronicles Presley’s rise and fall. Tom Hanks plays Colonel ...
- Max to Add 11 New Titles This Week - Here's the 3 Most Interestingon May 6, 2024 at 7:18 am
Here’s the three most interesting, in my humble opinion: The Iron Claw (2023) The 2023 biofilm stars Zach Efron and Jeremy Allen White as sons of wrestling promoter Fritz Von Erich who groomed them to ...
- Surprise! This Innocent-Looking Bathroom Item Is Actually The Germiest Part Of Your Bathroomon May 5, 2024 at 10:06 am
“You probably are going to have more of a chance of your face touching a shower curtain than a bath mat,” Tetro said. “So while the bath mat might be the germiest place, the shower curtain is actually ...
- Multispecies biofilm including Candida albicans. (IMAGE)on May 3, 2024 at 8:01 am
Disclaimer: AAAS and EurekAlert! are not responsible for the accuracy of news releases posted to EurekAlert! by contributing institutions or for the use of any information through the EurekAlert ...
- Antimicrobial peptide from cows shows potential for treating hypervirulent bacteriaon May 2, 2024 at 6:34 am
University of Central Florida College of Medicine researcher Renee Fleeman is on a mission to kill drug-resistant bacteria, and her latest study has identified a therapy that can penetrate the slime ...
- Should you try oil pulling to boost your oral health? Dentists explain benefits and side effectson April 30, 2024 at 3:36 pm
Oil pulling is a centuries-old technique that’s trending on social media for its dental health benefits. Experts explain how it works and what the science says.
- Solved! What Is the Pink Mold in My Shower, and How Do I Get Rid of It?on April 23, 2024 at 9:14 am
The discoloration comes from a biofilm—specifically, a bacterial colony of Serratia marcescens. Though it’s not actually mold, it’s commonly referred to as pink mold. Is pink mold ...
- The Extracellular Bastions of Bacteria — A Biofilm Way of Lifeon March 11, 2022 at 11:38 am
Bacteria permeate virtually every corner of the Earth. Most bacteria live in a biofilm state to enhance their survival and propagation, and to perform essential tasks for many ecosystems.
- Supercharged: the biofilm anodeon March 29, 2019 at 9:12 am
Reporting in Biotechnology & Bioengineering, Kato Marcus and colleagues describe the development of a mathematical model for the anode biofilm that can be used to investigate how electrons ...
via Bing News