Credit: David Song/NYU
Targeting the membrane of a virus, rather than its proteins, could lead to a new generation of antivirals
Antiviral therapies are notoriously difficult to develop, as viruses can quickly mutate to become resistant to drugs. But what if a new generation of antivirals ignores the fast-mutating proteins on the surface of viruses and instead disrupts their protective layers?
“We found an Achilles heel of many viruses: their bubble-like membranes. Exploiting this vulnerability and disrupting the membrane is a promising mechanism of action for developing new antivirals,” said Kent Kirshenbaum, professor of chemistry at NYU and the study’s senior author.
In a new study published Aug. 2 in the journal ACS Infectious Diseases, the researchers show how a group of novel molecules inspired by our own immune system inactivates several viruses, including Zika and chikungunya. Their approach may not only lead to drugs that can be used against many viruses, but could also help overcome antiviral resistance.
The urgent need for new antivirals
Viruses have different proteins on their surfaces that are often the targets of therapeutics like monoclonal antibodies and vaccines. But targeting these proteins has limitations, as viruses can quickly evolve, changing the properties of the proteins and making treatments less effective. These limitations were on display when new SARS-CoV-2 variants emerged that evaded both the drugs and the vaccines developed against the original virus.
“There is an urgent need for antiviral agents that act in new ways to inactivate viruses,” said Kirshenbaum. “Ideally, new antivirals won’t be specific to one virus or protein, so they will be ready to treat new viruses that emerge without delay and will be able to overcome the development of resistance.”
“We need to develop this next generation of drugs now and have them on the shelves in order to be ready for the next pandemic threat—and there will be another one, for sure,” added Kirshenbaum.
Drawing inspiration from our immune systems
Our innate immune system combats pathogens by producing antimicrobial peptides, the body’s first line of defense against bacteria, fungi, and viruses. Most viruses that cause disease are encapsulated in membranes made of lipids, and antimicrobial peptides work by disrupting or even bursting these membranes.
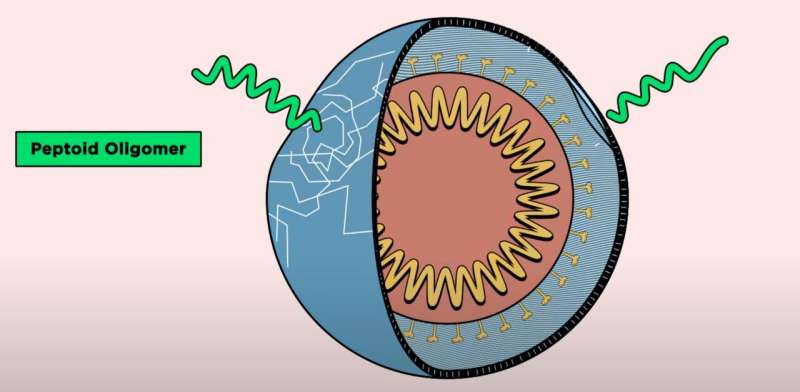
While antimicrobial peptides can be synthesized in the lab, they are rarely used to treat infectious diseases in humans because they break down easily and can be toxic to healthy cells. Instead, scientists have developed synthetic materials called peptoids, which have similar chemical backbones to peptides but are better able to break through virus membranes and are less likely to degrade.
“We began to think about how to mimic natural peptides and create molecules with many of the same structural and functional features as peptides, but are composed of something that our bodies won’t be able to rapidly degrade,” said Kirshenbaum.
The researchers investigated seven peptoids, many originally discovered in the lab of Annelise Barron, a co-author of the study who currently leads a lab at Stanford. The NYU team studied the antiviral effects of the peptoids against four viruses: three enveloped in membranes (Zika, Rift Valley fever, and chikungunya) and one without (coxsackievirus B3).
“We were particularly interested in studying these viruses as they have no available treatment options,” said Patrick Tate, a chemistry PhD student at NYU and the study’s first author.
How peptoids disrupt viral membranes and avoid other cells
The membranes surrounding viruses are made of different molecules than the virus itself, as lipids are acquired from the host to form membranes. One such lipid, phosphatidylserine, is present in the membrane on the outside of viruses, but is sequestered towards the interior of human cells under normal conditions.
“Because phosphatidylserine is found on the exterior of viruses, it can be a specific target for peptoids to recognize viruses, but not recognize—and therefore spare—our own cells,” said Tate. “Moreover, because viruses acquire lipids from the host rather than encoding from their own genomes, they have better potential to avoid antiviral resistance.”
The researchers tested seven peptoids against the four viruses. They found that the peptoids inactivated all three enveloped viruses—Zika, Rift Valley fever, and chikungunya—by disrupting the virus membrane, but did not disrupt coxsackievirus B3, the only virus without a membrane.
Moreover, chikungunya virus containing higher levels of phosphatidylserine in its membrane was more susceptible to the peptoids. In contrast, a membrane formed exclusively with a different lipid named phosphatidylcholine was not disrupted by the peptoids, suggesting that phosphatidylserine is crucial in order for peptoids to reduce viral activity.
“We’re now starting to understand how peptoids actually exert their antiviral effect—specifically, through the recognition of phosphatidylserine,” said Tate.
The researchers are continuing pre-clinical studies to evaluate the potential of these molecules in fighting viruses and to understand if they can overcome the development of resistance. Their peptoid-focused approach may hold promise for treating a wide range of viruses with membranes that can be difficult to treat, including Ebola, SARS-CoV-2, and herpes.
Original Article: Novel Molecules Fight Viruses by Bursting Their Bubble-like Membranes
More from: New York University | Loyola University Chicago | University of Louisville
The Latest Updates from Bing News
Go deeper with Bing News on:
Antiviral therapies
- Poseida Highlights Strong Progress on Its Genetic Medicine Programs at the American Society for Gene and Cell Therapy 27th Annual Meeting
Poseida Therapeutics, Inc. (Nasdaq: PSTX), a clinical-stage allogeneic cell therapy and genetic medicines company advancing differentiated non-viral treatments for patients with cancer and rare ...
- Chris Cuomo brutally mocked for taking U-turn on anti-viral drug Ivermectin: ‘He’s such a hypocrite’
NewsNation anchor Chris Cuomo recently claimed that he is facing side-effects caused by a COVID-19 vaccine. And now in his latest appearance on Patrick Bet-David's podcast, Cuomo revealed that he ...
- ASGCT: Ring's 'new class of viral vector' is re-dosable in primates, overcoming key challenge of AAVs
Kahvejian told Ong that one of the firm’s companies, Ring Therapeutics, was exploring ways to harness a family of benign, human-dwelling viruses called anelloviruses and use them as viral vectors for ...
- A Novel Broad-Spectrum Antiviral with Activity Against Smallpox/Mpox
NanoViricides, Inc. (the "Company") (www.nanoviricides.com) is a development stage company that is creating special purpose nanomaterials for antiviral therapy. The Company's novel nanoviricide® class ...
- Gut-Brain Treatment Goes Viral
It’s not just gut bacteria that connect our guts to our brains. Tiny viruses also have an outsized role to play.
Go deeper with Bing News on:
Antivirals
- How the Flu Virus Sneaks Into the Brain and What We Can Do About It
Some influenza-associated brain disorders may be caused by the virus entering the brain and making proteins, suggesting that antivirals that block protein production might be an effective treatment, ...
- How the Flu Virus Invades the Brain
Researchers uncovered how the influenza virus may infiltrate the brain, leading to a serious condition known as influenza-associated encephalopathy (IAE).
- Promising new treatment strategy for deadly flu-related brain disorders
Researchers from Osaka University have found that a brain disorder associated with flu (influenza-associated encephalopathy, or IAE) can be caused by the influenza virus entering the brain from the ...
- Four reasons to be concerned (but not freak out) about the bird flu
Government officials say they are closely monitoring the bird flu virus in food, livestock and people but experts say more needs to be done.
- The Era of Direct-acting Antivirals Has Begun
Lower Limit of Detection (LLOD) Versus Lower Limit of Quantification (LLOQ) The LLOD is the HCV RNA concentration at which less than 5% of the samples that contain a known amount of an RNA ...









