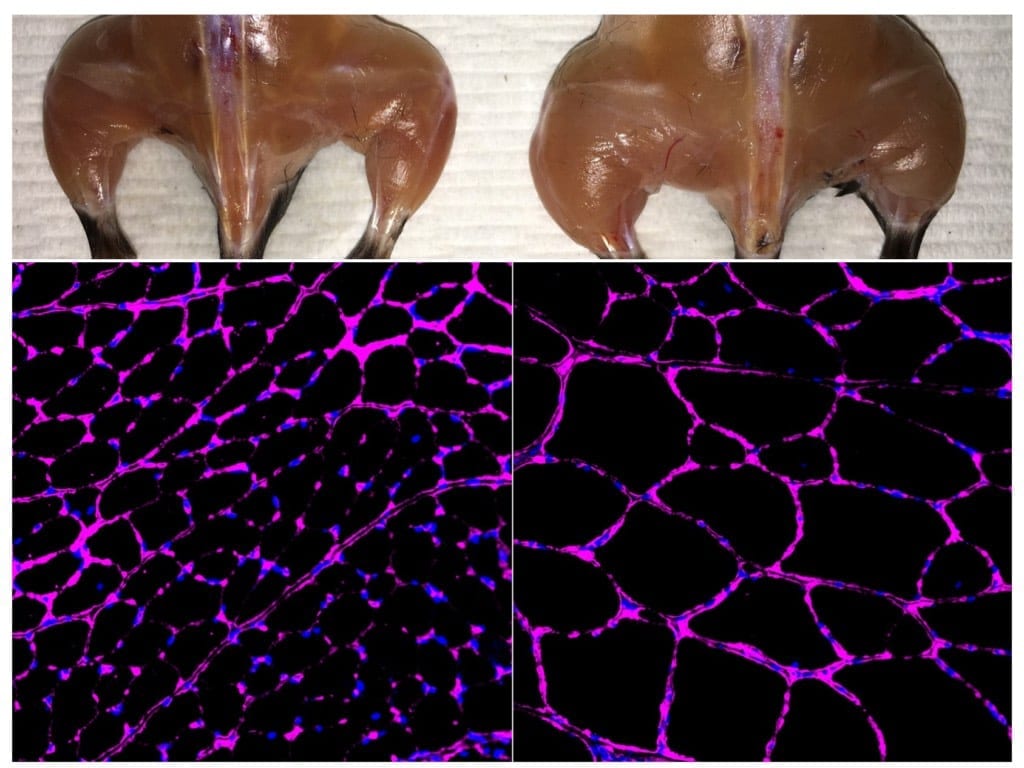
Muscular dystrophy, which affects approximately 250,000 people in the U.S., occurs when damaged muscle tissue is replaced with fibrous, fatty or bony tissue and loses function. For years, scientists have searched for a way to successfully treat the most common form of the disease, Duchenne Muscular Dystrophy (DMD), which primarily affects boys. Now, a team of University of Missouri researchers have successfully treated dogs with DMD and say that human clinical trials are being planned in the next few years.
“This is the most common muscle disease in boys, and there is currently no effective therapy,” saidDongsheng Duan, the study leader and the Margaret Proctor Mulligan Professor in Medical Research at the MU School of Medicine. “This discovery took our research team more than 10 years, but we believe we are on the cusp of having a treatment for the disease.”
Patients with Duchenne muscular dystrophy have a gene mutation that disrupts the production of a protein known as “dystrophin.” Absence of dystrophin starts a chain reaction that eventually leads to muscle cell degeneration and death. Affected boys lose their ability to walk and breathe as they get older. This places significant limitations on individuals afflicted with the disease. Dystrophin also is one of the largest genes in the human body.
“Due to its size, it is impossible to deliver the entire gene with a gene therapy vector, which is the vehicle that carries the therapeutic gene to the correct site in the body,” Duan said. “Through previous research, we were able to develop a miniature version of this gene called a microgene. This minimized dystrophin protected all muscles in the body of diseased mice.”
However, it took the team more than 10 years to develop a strategy that can safely send the micro-dystrophin to every muscle in a dog that is afflicted by the disease. The dog has a body size similar to that of an affected boy. Success in the dog will set the foundation for human tests.
In this latest study, the MU team demonstrated for the first time that a common virus can deliver the microgene to all muscles in the body of a diseased dog. The dogs were injected with the virus when they were two to three months old and just starting to show signs of DMD. The dogs are now six to seven months old and continue to develop normally.
“The virus we are using is one of the most common viruses; it is also a virus that produces no symptoms in the human body, making this a safe way to spread the dystrophin gene throughout the body,” Duan said. “These dogs develop DMD naturally in a similar manner as humans. It’s important to treat DMD early before the disease does a lot of damage as this therapy has the greatest impact at the early stages in life.”
The Latest on: Muscular Dystrophy
[google_news title=”” keyword=”Muscular Dystrophy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Muscular Dystrophy
- Second Death - Pfizer Reports Young Boy's Death After One Year Of Gene Therapy Treatment In Muscle Wasting Disorder Trialon May 8, 2024 at 10:09 am
A young patient died due to cardiac arrest after receiving Pfizer Inc’s (NYSE:PFE) experimental gene therapy in a mid-stage trial for Duchenne muscular dystrophy (DMD), the company told in a letter to ...
- Help Chattanooga firefighters 'Fill the Boot' in support of Muscular Dystrophy Associationon May 8, 2024 at 8:18 am
It’s that time of year where firefighters nationwide are joining forces to "Fill the Boot" for the Muscular Dystrophy Association.The Chattanooga Fire Departmen ...
- Child dies in gene therapy trial; Pfizer pauses studyon May 8, 2024 at 7:31 am
Death of boy in Pfizer's phase 2 study of gene therapy for Duchenne muscular dystrophy reported by patient advocacy group. Pfizer pauses dosing in another trial ...
- Pfizer Halts Dosing in Late-Stage DMD Study Following Death of Young Patienton May 8, 2024 at 5:24 am
Pfizer’s investigational Duchenne muscular dystrophy gene therapy fordadistrogene movaparvovec has been hit with another patient death, forcing the pharma to pause dosing in its Phase III study.
- Boy Dosed with Pfizer DMD Gene Therapy Dies a Year after Phase II Trialon May 7, 2024 at 11:19 pm
Pfizer has paused enrollment in a Phase III trial assessing its Duchenne muscular dystrophy (DMD) gene therapy candidate fordadistrogene movaparvovec after acknowledging the sudden death of a young ...
- Young boy dies in trial for Pfizer Duchenne gene therapyon May 7, 2024 at 2:57 pm
A boy with Duchenne muscular dystrophy died after receiving a Pfizer experimental gene therapy. The company is pausing its trial in older boys.
- Patient dies in Pfizer study of Duchenne gene therapyon May 7, 2024 at 2:39 pm
Pfizer said the patient, a young boy who was treated earlier last year, had died suddenly. The company is working with trial researchers to investigate further.
- UPDATE: Young boy's fatal cardiac arrest prompts dosing pause in Pfizer's phase 3 gene therapy trialon May 7, 2024 at 1:29 pm
A young boy with Duchenne muscular dystrophy (DMD) participating in Pfizer’s phase 2 gene therapy trial has died, the pharma shared in a May 7 ...
- Pfizer Pauses Gene Therapy Trial for Muscle Disease After Deathon May 7, 2024 at 1:27 pm
Pfizer Inc. paused a study of an experimental gene therapy for muscular dystrophy after a child who received it died suddenly.
- My Turn | Illinois can lead the way in the fight for a cure to Duchenne Muscular Dystrophyon May 7, 2024 at 2:45 am
"Knowing that both of our sons have what is essentially a death sentence was and is devastating," writes Daniel Flessner of Roberts.
via Bing News