via Wikipedia
Researchers from Genethon, the AFM-Téléthon laboratory, Inserm (UMR 1089, Nantes) and the University of London (Royal Holloway) demonstrated the efficacy of an innovative gene therapy in the treatment of Duchenne muscular dystrophy. Indeed, after injecting microdystrophin (a “shortened” version of the dystrophin gene) via a drug vector, the researchers managed to restore muscle strength and stabilise the clinical symptoms in dogs naturally affected by Duchenne muscular dystrophy.
This work published in Nature Communications has been achieved thanks to the donations of the French Téléthon.
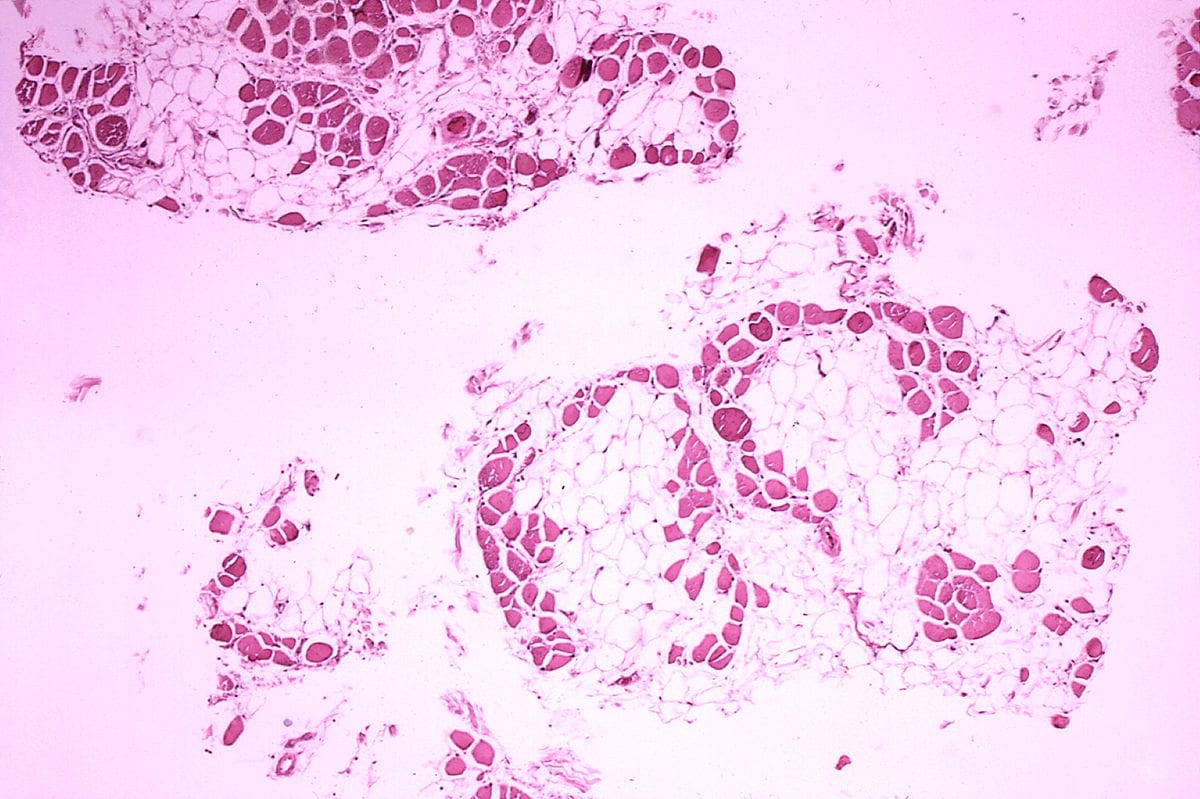
Teams at Genethon developed, in collaboration with a team at Royal Holloway University of London led by Pr. Dickson, and produced, a gene therapy drug combining an AAV-type viral vector with a shortened version of the dystrophin gene (approximately 4,000 base pairs), allowing the production of a functional protein. Dr Le Guiner’s team tested this innovative treatment in 12 dogs naturally affected by Duchenne muscular dystrophy. By injecting this microdystrophin intravenously, and hence into the whole body of the dogs, the researchers observed that dystrophin expression returned to a high level, and muscle function was significantly restored, with stabilisation of the clinical symptoms observed for over 2 years following injection of the drug. No immunosuppressive treatment was administered beforehand, and no sideeffects were observed.
Some Golden Retrievers develop Duchenne muscular dystrophy naturally. The successful treatment of these dogs, which show the same clinical symptoms as children with this disease, and are of a similar weight, is a decisive step toward developing the same treatment in children. “This preclinical study demonstrates the safety and efficacy of microdystrophin, and makes it possible to consider developing a clinical trial in patients. Indeed, this is the first time that it has been possible to treat the whole body of a large sized animal with this protein. Moreover, this innovative approach allows treatment of all patients with Duchenne muscular dystrophy, regardless of the genetic mutation responsible,” says Caroline Le Guiner, the main author of this study.
Learn more: Gene therapy: Microdystrophin restores muscle strength in Duchenne muscular dystrophy
The Latest on: Duchenne muscular dystrophy
[google_news title=”” keyword=”Duchenne muscular dystrophy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
- These 15 One-of-a-Kind Watches Won’t Appear at the OnlyWatch Auction. We Asked the Brands Why.on April 26, 2024 at 9:00 am
We asked 15 brands why their unique timepieces for OnlyWatch will not appear at the rescheduled auction in Geneva next month, and what the fate of those watches might be.
- Patients With DMD Receiving Eteplirsen Show Prolonged Survival, Study Findson April 25, 2024 at 2:03 pm
The study provides evidence of survival benefits among patients with Duchenne muscular dystrophy (DMD) receiving eteplirsen compared with the natural history of the condition.
- DeWine: Ohio first state in the nation to screen newborn babies for Duchenne Muscular Dystrophyon April 24, 2024 at 9:00 pm
Ohio Gov. Mike DeWine on Monday announced that Ohio will become the first state in the nation to begin screening all newborn babies for Duchenne Muscular Dystrophy. The provision was included in HR 33 ...
- Gov. DeWine announces Ohio will be first state to screen newborns for DMDon April 24, 2024 at 7:23 am
Gov. Mike DeWine announced Tuesday that the state of Ohio will become the first in the United States to screen all newborn babies for Duchenne muscular dystrophy.
- Glucocorticoid use can aid breathing ability in Duchenne adults: Studyon April 23, 2024 at 8:28 am
Glucocorticoid use was seen to help maintain breathing ability and arm function in adults in the late stages of Duchenne muscular dystrophy.
- Duchenne UK and Parent Project Muscular Dystrophy Award $500,000 to Evaluate Safety and Tolerability of Muscle Progenitor Cells in Phase 1 Trialon April 23, 2024 at 7:01 am
Parent Project Muscular Dystrophy (PPMD), a US nonprofit organization, and the UK charity Duchenne UK, two leading organizations dedicated to ending Duchenne muscular dystrophy (Duchenne), are excited ...
- My son's smile gives me hope for Duchenne muscular dystrophy. A new law could help otherson April 23, 2024 at 6:01 am
Finally, when we were approved for evaluation, we received a devastating diagnosis: Duchenne muscular dystrophy. Duchenne is a rare, progressive disorder in which muscle cells are in a constant ...
- My son's smile gives me hope for Duchenne muscular dystrophy. A new law could help otherson April 23, 2024 at 5:01 am
Opinion: Newborn testing can give families a head start on giving a child with the degenerative muscular disease a shot at the best outcome in life.
- An FDA pathway can accelerate innovation for Duchenne muscular dystrophyon April 22, 2024 at 1:31 am
Few treatments are available for people with Duchenne muscular dystrophy. The FDA's accelerated approval program can help change that.
via Google News and Bing News