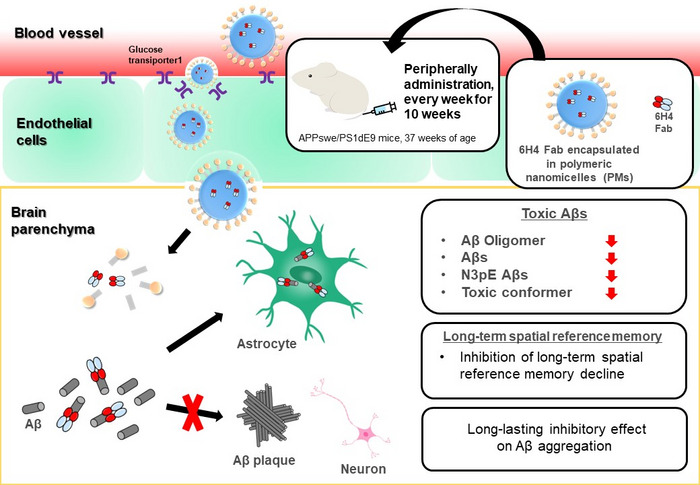
The newly developed antibody 6H4 fragments (Fabs) specific to A? oligomers, encapsulated in the polymeric nanomicelles (PMs), were peripherally administrated to AD model mouse for 10 weeks. In mice that were administered anti-A?O 6H4 Fabs in the PMs, various toxic A? species (A? oligomers, A?42s, N3pE A?s, A?s with toxic conformer) and the presence of A? plaques with dense cores in the brain were significantly reduced. Additionally, suppression of the progression of pathological processes of AD was supported by the prevention of cognitive-behavioral decline in the spatial memory tests.
CREDIT: Department of Neurology and Neurological Science, TMDU
Researchers led by Tokyo Medical and Dental University (TMDU) find that antibody fragments encapsulated in nanomicelles cross the blood–brain barrier and reduce the levels of toxic A? species in the brain of an Alzheimer’s disease model mouse
Sometimes the best things in life come by chance, when we happen to be in the right place at the right time. Now, researchers from Japan have found a way to ensure that new medications are delivered to the right place in the body and at the right timepoint in disease progression, so that they have the best effect.
In a study published recently in the Journal of Nanobiotechnology, researchers led by Tokyo Medical and Dental University (TMDU) have revealed that a novel delivery system delivers treatment to where it is needed most in a mouse model of Alzheimer’s disease (AD).
AD is a common neurodegenerative disease that causes dementia. It is characterized by the accumulation of a protein called amyloid ? (A?) in the brain, and a number of different toxic forms of A? have been identified that impair brain function, notably A? oligomers (A?Os).
“Multiple clinical trials have attempted to use an anti-A? antibody to treat AD, but the results have been unsatisfactory,” says lead author of the study Akiko Amano. “One potential explanation for this is that the blood–brain barrier (BBB) prevents most full-length antibodies from entering the brain.”
To address this challenge, the researchers previously developed glucosylated (sugar-linked) polymeric nanomicelles (PMs), which are tiny, hollow balls that could successfully cross the BBB via transcytosis in mouse brain capillary endothelial cells; this process was mediated by glucose-transporter-1 and induced by an increase in blood glucose levels after the mice experienced fasting conditions. In this study, Takanori Yokota and colleagues filled PMs with fragments of an anti-A?O antibody, injected them into a mouse model of AD, and assessed the effects on the brain and on behavior.
“The results were very clear,” explains senior author Nobuo Sanjo. “Administration of anti-A?O antibody fragments through PMs significantly reduced the amounts of various toxic A? species. In addition, the A? plaques that did form were smaller and less dense than those seen in untreated mice.”
Next, the researchers analyzed the behavior of the mice and found that the mice treated with the antibody fragment-filled PMs had better learning and spatial memory than untreated mice. “Our findings suggest that delivering sufficient levels of antibodies to the brain using PMs can reduce toxic A? species and slow AD progression in mice,” says Amano.
Given that the failure of anti-A? antibodies to improve cognitive function in human clinical trials was likely because of an insufficient supply of the antibodies in the brain, PM-encapsulated antibody fragments could represent an effective way to prevent AD progression. In addition, new candidates for AD treatment that degrade toxic A?s and reduce their toxic effects could also be delivered to the brain using the same PM-based system.
Original Article: Transporting antibodies across the blood–brain barrier to treat Alzheimer’s disease
More from: Tokyo Medical and Dental University
The Latest Updates from Bing News
Go deeper with Bing News on:
Alzheimer’s disease
- New tool links Alzheimer's disease types to rate of cognitive decline
Mayo Clinic researchers have discovered a series of brain changes characterized by unique clinical features and immune cell behaviors using a new corticolimbic index tool for Alzheimer's disease, a leading cause of dementia.
- How exactly does a high-fat diet increase Alzheimer's risk?
New research in mouse models of Alzheimer's disease suggests that a high-fat diet causes molecular changes that disrupt brain health.
Go deeper with Bing News on:
PM-encapsulated antibody fragments
- Best Biotech Penny Stocks
Looking for the best biotech penny stocks? The most important innovation of the 1990s was the internet — today, it would be a vaccine for COVID-19. If these times have anything to say about it ...
- Therapeutic Antibodies – Webinars and Online Events
This exclusive Technology Networks webinar focuses on the most recent and innovative strategies for therapeutic antibody development and production. Your speaker, Professor Brockwell, will describe ...
- Recombinant lipid-tagged antibody fragments as functional cell-surface receptors
To establish whether molecules other than scFv antibody fragments could be lipid-modified and inserted into cell membranes, we cloned the gene encoding human interleukin-2 (IL-2) into vector pLP2 ...
- Method created to better remove protein causing Alzheimer’s
The researchers, primarily from Tokyo Medical and Dental University and the Innovation Center of Nanomedicine, encapsulated fragments of an antibody that can attach itself to the protein and ...
- Antibody Specificity, Affinity and Kinetics in a Single Experiment
The smaller antibody formats such as single-chain fragment variables, nanobodies or other scaffolds that can reach hidden targets or act as intrabodies to target intracellular antigens must strike a ...