UNSW researchers have helped show how carbon dioxide can be broken down cheaply and efficiently via a process that dissolves captured CO2 gas into a solvent around nanoparticles of gallium.
A global collaboration, led by researchers from UNSW, has shown how liquid gallium can be used to help achieve the important goal of net zero carbon emissions.
Engineers from UNSW have helped to discover a cheap new way to capture and convert CO2 greenhouse emissions using liquid metal.
The process can be done at room temperature and uses liquid gallium to convert the carbon dioxide into oxygen and a high-value solid carbon product that can later be used in batteries, or in construction, or aircraft manufacturing.
A team from the School of Chemical Engineering, led by Professor Kourosh Kalantar-Zadeh, worked in collaboration with researchers at University of California, Los Angeles (UCLA), North Carolina State University, RMIT, University of Melbourne, Queensland University of Technology, and the Australian Synchrotron (ANSTO).
Their findings have been published in the Advanced Materials journal and Professor Kalantar-Zadeh and his team say the new technology has the potential to be used in a wide variety of ways to significantly reduce the levels of greenhouse gases in the atmosphere.
“We see very strong industrial applications with regards to decarbonisation. This technology offers an unprecedent process for capturing and converting CO2 at an exceptionally competitive cost,” said Junma Tang, the first author of the paper.
“The applications could be in cars to convert polluting exhaust gases, or even at a much larger scale at industrial sites where CO2 emissions could be immediately captured and processed using this technology.
“We have already scaled this system up to two-and-a-half litres dimensions, which can deal with around 0.1 litre of CO2 per minute. And we’ve tested that running continuously for a whole month and the efficiency of the system did not degrade.”
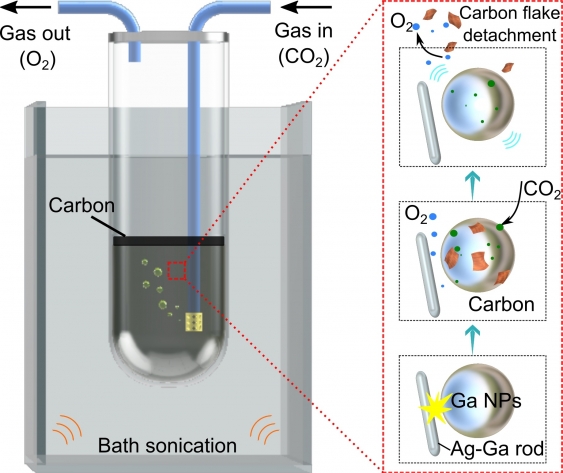
The newly discovered process dissolves captured CO2 gas into a solvent around nanoparticles of gallium, which exist in liquid state above 30°C.
The reactor also contains nano-sized solid silver rods that are the key to generating the triboelectrochemical reactions that take place once mechanical energy (e.g. stirring/mixing) is introduced.
A triboelectrochemical reaction occurs in solid–liquid interfaces due to friction between the two surfaces, with an electric field also created that sparks a chemical reaction.
The reactions break the carbon dioxide into oxygen gas, as well as carbonaceous sheets which ‘float’ to the surface of the container due to differences in density and can therefore be easily extracted.
In their paper, the research team show a 92 per cent efficiency in converting a tonne of CO2 as described, using a low energy input. They estimate it equates to a cost of around $100 per tonne of CO2.
In order to commercialise the research, a spin-out company called LM Plus has been established with the support of UNSW’s Knowledge Exchange – a program that helps transform research discoveries into successful innovations to benefit society, along with seed investment from Uniseed.
UNSW has become a shareholder as part of a license agreement with the company.
“This is a very green process which also produces a high-value carbonaceous sheet which can then be sold and used to make electrodes in batteries, or for carbon fibre materials that are used in high-performance products like aircraft, racing cars, and luxury vehicles,” said Paul Butler, Uniseed’s Investment Manager and director of LM Plus.
“What we are working towards now is to raise funds to build a larger size proof-of-concept for this system to work within a 40-foot container –the size of a truck trailer – that could ultimately help industrial sites immediately capture any CO2 emissions and convert them.”
LM Plus hopes to be able to build that much larger system within 15 months and is already in talks with potential commercial partners about other ways of implementing the new technology.
Original Article: Liquid metal proven to be cheap and efficient CO2 converter
More from: University of New South Wales | University of California Los Angeles | North Carolina State University | Royal Melbourne Institute of Technology | University of Melbourne | Queensland University of Technology | Australian Synchrotron
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Capturing and converting CO2
- Q3 2024 Applied Industrial Technologies Inc Earnings Callon April 25, 2024 at 9:10 pm
Welcome to the fiscal 2024 third quarter earnings call for Applied Industrial Technologies. My name is Shayan, and I'll be your operator for today's call. (Operator Instructions) Please note that this ...
- Bureau Veritas: Strong Start to the Year; 2024 Outlook Confirmedon April 24, 2024 at 5:20 pm
NEUILLY-SUR-SEINE, France, April 25, 2024--(BUSINESS WIRE)--Q1 2024 Key figures 1 › Revenue of EUR 1,439.5 million in the first quarter of 2024, up 2.5% year-on-year and up 8.0% organically › Strong ...
- Third Avenue International Real Estate Value Fund Q1 2024 Portfolio Manager Commentaryon April 23, 2024 at 2:15 am
Q1, the Third Avenue Real Estate Value Fund generated a return of +2.81% (after fees) versus -1.11% (before fees).
- Check out Some of the Mind-Blowing Visuals as Phish Bounced Around the Vegas Sphereon April 22, 2024 at 5:38 am
The latest from the world's largest LED screen in our running blog. Don't miss a story from inside (and outside) the Sphere.
- FTSE 100 Live 19 April: Index closes up for day, down for week; S&P below 5000on April 21, 2024 at 11:22 pm
The FTSE 100 index is set to fall about 0.9% as traders react to developments in the Middle East and a weak finish for US stocks last night. Brent Crude rose by about $4 at one point to a peak of ...
Go deeper with Google Headlines on:
Capturing and converting CO2
[google_news title=”” keyword=”capturing and converting CO2” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Liquid gallium
- Making Diamonds Doesn’t Require Extreme Pressure Anymoreon April 26, 2024 at 10:45 am
A new method of making diamonds doesn’t require extreme pressure. It involves diamond growth without traditional high-pressure conditions.
- Lab-grown diamond created in less than 3 hours — will it revolutionize the jewelry industry?on April 26, 2024 at 10:27 am
Diamonds are a girl’s best friend— but they’re this scientist’s best creation. Researchers have discovered a faster method to making diamonds in a laboratory without the extreme conditions usually ...
- Scientists can make perfectly good diamonds in just 150 minuteson April 25, 2024 at 10:32 pm
A team of scientists were able to create a completely authentic diamond in only 150 minutes, move over billions of years of pressure.
- Instant diamonds that take ‘under three hours to grow’ invented by scientists to avoid ‘billion-year’ wait for real gemson April 25, 2024 at 4:39 pm
A DAZZLING breakthrough has figured out how to produce diamonds in just 150 minutes compared to the billion-year natural process. The researchers behind the invention are confident they’ll ...
- Diamonds can now be grown in labon April 25, 2024 at 11:45 am
A new method for creating diamonds in the lab without severe pressure has been devised by a research team led by Rod Ruoff at the Institute of Basic Science at the Ulsan National Institute ...
Go deeper with Google Headlines on:
Liquid gallium
[google_news title=”” keyword=”liquid gallium” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]










