“These described methods allow us to do this in two to three hours, a vast improvement over standard culturing practice, which can take as much as two weeks to provide a diagnosis.”
A handheld diagnostic device that Massachusetts General Hospital (MGH) investigators first developed to diagnose cancer has been adapted to rapidly diagnose tuberculosis (TB) and other important infectious bacteria. Two papers appearing in the journals Nature Communications and Nature Nanotechnology describe portable devices that combine microfluidic technology with nuclear magnetic resonance (NMR) to not only diagnose these important infections but also determine the presence of antibiotic-resistant bacterial strains.
“Rapidly identifying the pathogen responsible for an infection and testing for the presence of resistance are critical not only for diagnosis but also for deciding which antibiotics to give a patient,” says Ralph Weissleder, MD, PhD, director of the MGH Center for Systems Biology (CSB) and co-senior author of both papers. “These described methods allow us to do this in two to three hours, a vast improvement over standard culturing practice, which can take as much as two weeks to provide a diagnosis.”
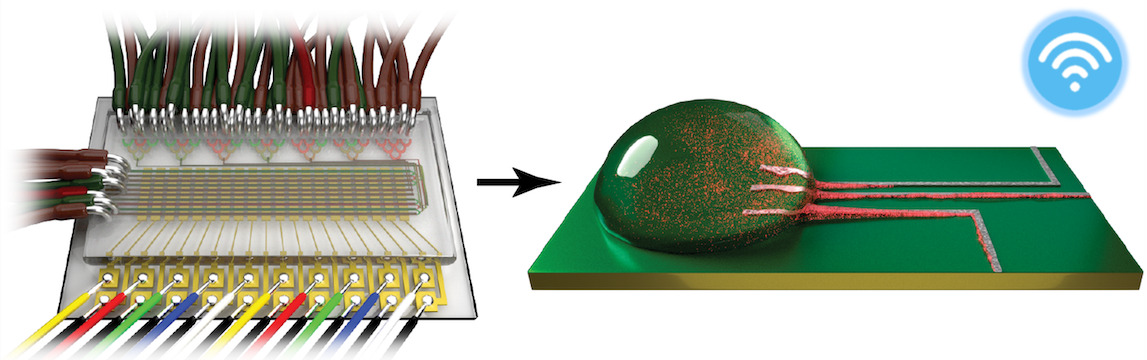
Investigators at the MGH CSB previously developed portable devices capable of detecting cancer biomarkers in the blood or in very small tissue samples. Target cells or molecules are first labeled with magnetic nanoparticles, and the sample is then passed through a micro NMR system capable of detecting and quantifying levels of the target. But initial efforts to adapt the system to bacterial diagnosis had trouble finding antibodies – the detection method used in the earlier studies – that would accurately detect the specific bacteria. Instead the team switched to targeting specific nucleic acid sequences.
The system described in the Nature Communications paper, published on April 23, detects DNA from the tuberculosis bacteria in small sputum samples. After DNA is extracted from the sample, any of the target sequence that is present is amplified using a standard procedure, then captured by polymer beads containing complementary nucleic acid sequences and labeled with magnetic nanoparticles with sequences that bind to other portions of the target DNA. The miniature NMR coil incorporated into the device – which is about the size of a standard laboratory slide – detects any TB bacterial DNA present in the sample.
Tests of the device on samples from patients known to have TB and from healthy controls identified all positive samples with no false positives in less than three hours. Existing diagnostic procedures can take weeks to provide results and can miss up to 40 percent of infected patients. Results were even stronger for patients infected with both TB and HIV – probably because infection with both pathogens leads to high levels of the TB bacteria – and specialized nucleic acid probes developed by the research team were able to distinguish treatment-resistant bacterial strains.
The Latest Bing News on:
Handheld diagnostic device
- Massage Guns the Likely Culprit in Two Cases of Acute Vertigoon May 9, 2024 at 4:59 pm
That's the diagnostic challenge described by David Elisha ... massage guns remain unexplored in existing literature," Elisha and Nazarian noted. Handheld massage devices apply significantly more force ...
- MAICO Ero•Scan Pro + Tymp System from e3 Diagnosticson May 9, 2024 at 4:59 pm
The Ero•Scan Pro + Tymp is a hand-held device that fits into any practice, performing hearing screening or diagnostic testing. With the push of one button, easily run up to three (3) tests ...
- New Therapeutic Promise For Fibromyalgiaon May 6, 2024 at 5:00 am
This month you might see the purple colors of Fibromyalgia Awareness Month, when people around the world try to raise awareness for fibromyalgia and other chronic pain conditions. The month is a time ...
- FDA Approves First Fully Autonomous Handheld AI device for Diabetic Retinopathy Screeningon May 3, 2024 at 7:00 pm
AEYE Health, the leading company for AI retinal imaging and diagnostics, is thrilled to announce that it received the first ever FDA clearance for a fully autonomous AI that diagnoses ...
- OPTOMED RELEASES FIRST FDA-CLEARED HANDHELD AI FUNDUS CAMERA FOR DETECTION OF MORE THAN MILD DIABETIC RETINOPATHYon May 1, 2024 at 7:00 am
Optomed USA, Inc., a medical technology company, introduces the Optomed Aurora AEYE, a handheld AI fundus camera that provides instant detection of more than mild diabetic retinopathy. With the ...
- FDA approves the first fully autonomous AI for portable diabetic retinopathy screeningon April 30, 2024 at 3:29 pm
DS, enables autonomous diabetic retinopathy screening using handheld cameras, revolutionizing eye care accessibility and outcomes.
- Could patients be seen by hospital consultants in their own homes?on April 28, 2024 at 10:01 pm
A hospital consultant explains how health professionals can deliver hospital-level care in people’s homes with the help of digital diagnostic and monitoring devices ...
- Omdia research handheld gaming devices will reach 29.3 million in 2028on April 24, 2024 at 12:02 pm
Omdia's Games Handhelds Database helps unravel handheld gaming's newfound complexity, sizing all key devices and market categories ... Inc. , a leading diagnostic solutions company with a focus in ...
- Ravenshaw University researchers invent portable device for instant cancer detectionon March 24, 2024 at 10:53 pm
BHUBANESWAR: A group of researchers from Ravenshaw University has invented a unique diagnostic device that could revolutionise cancer detection in the country. The portable device can detect cancer ...
- FDA Approves AI-Powered Handheld Device That Can Detect Skin Canceron January 17, 2024 at 7:36 am
The FDA has approved DermaSensor’s first-of-its-kind handheld device that uses artificial ... for skin cancer and should not be the only diagnostic criterion. The FDA is also requiring that ...
The Latest Google Headlines on:
Handheld diagnostic device
[google_news title=”” keyword=”handheld diagnostic device” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
The Latest Bing News on:
Diagnostic device
- Global Pelvic Floor Diagnostics Industryon May 13, 2024 at 9:03 am
Global Pelvic Floor Diagnostics Industry is expected to hit US$ 3,753.3 Million at CAGR of 6.2% during forecast period 2023 to 2033 | FMI ...
- Global Diagnostic X-Ray System Market Growth, Forecasted to Reach USD 20.46 Billion by 2032 at a 6.1% of CAGRon May 13, 2024 at 7:21 am
The global Diagnostic X-Ray System market is on track for substantial growth, with an expected valuation of USD 11.30 billion by the end of 2022. Projections indicate further expansion at a Compound ...
- Dementia Action Week 2024: the future of Alzheimer’s diagnosticson May 13, 2024 at 4:29 am
As the debate over using tau levels to guide patient treatment persists, the landscape for Alzheimer’s diagnostics continues to move ahead.
- Sleep Apnea Devices Market, Industry Trends and Global Forecasts 2024-2035: How Contactless Sleep Apnea Devices Are Transforming Treatmenton May 13, 2024 at 1:44 am
The global sleep apnea market highlights the distribution of this segment across different types of products, such as sleep apnea medical devices and drug therapies. The sleep apnea devices market ...
- Qiagen’s QIAstat-Dx respiratory diagnostic wins FDA clearanceon May 13, 2024 at 12:02 am
In September 2023, Glasgow-based diagnostics spinout Microplate Dx raised £2.5m ($3.1m) in seed funding to develop a point-of-care device to identify effective antibiotics for bacterial infections.
- Targeted Diagnostic Botox Injections Can Identify Trigger Sites for Migraineon May 9, 2024 at 2:01 pm
Diagnostic targeted Botox injections have high positive predictive value for migraine trigger site localization, according to a study published in the May issue of Plastic and Reconstructive Surgery.
- Library hosting free device diagnostic day on May 18on May 9, 2024 at 5:53 am
BOONE — The Watauga County Public Library is hosting a free computer check-up on May 18 from 9 a.m. to 2 p.m.
- A diagnostic tool for ‘forgotten’ disease of endometriosison May 7, 2024 at 12:54 am
EndoCure is combining ultrasound and AI to allow physicians to clearly see the tiny lesions invisible to standard imaging systems – a tool that could transform diagnosis.
- Blood diagnostics device modeled on leeches could be use to detect malariaon May 2, 2024 at 9:22 am
Researchers at ETH Zurich have developed a safe and inexpensive device for reliable blood measurements. It works using a suction cup and could also be employed to diagnose the tropical disease malaria ...
- A new vet diagnostic tool swiftly identifies deadly swine illnesson May 1, 2024 at 9:49 am
Scientists are leveraging the Swine Disease Reporting System to rapidly detect new strains of porcine reproductive and respiratory syndrome virus.
The Latest Google Headlines on:
Diagnostic device
[google_news title=”” keyword=”diagnostic device” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]