UNSW researchers have helped show how carbon dioxide can be broken down cheaply and efficiently via a process that dissolves captured CO2 gas into a solvent around nanoparticles of gallium.
A global collaboration, led by researchers from UNSW, has shown how liquid gallium can be used to help achieve the important goal of net zero carbon emissions.
Engineers from UNSW have helped to discover a cheap new way to capture and convert CO2 greenhouse emissions using liquid metal.
The process can be done at room temperature and uses liquid gallium to convert the carbon dioxide into oxygen and a high-value solid carbon product that can later be used in batteries, or in construction, or aircraft manufacturing.
A team from the School of Chemical Engineering, led by Professor Kourosh Kalantar-Zadeh, worked in collaboration with researchers at University of California, Los Angeles (UCLA), North Carolina State University, RMIT, University of Melbourne, Queensland University of Technology, and the Australian Synchrotron (ANSTO).
Their findings have been published in the Advanced Materials journal and Professor Kalantar-Zadeh and his team say the new technology has the potential to be used in a wide variety of ways to significantly reduce the levels of greenhouse gases in the atmosphere.
“We see very strong industrial applications with regards to decarbonisation. This technology offers an unprecedent process for capturing and converting CO2 at an exceptionally competitive cost,” said Junma Tang, the first author of the paper.
“The applications could be in cars to convert polluting exhaust gases, or even at a much larger scale at industrial sites where CO2 emissions could be immediately captured and processed using this technology.
“We have already scaled this system up to two-and-a-half litres dimensions, which can deal with around 0.1 litre of CO2 per minute. And we’ve tested that running continuously for a whole month and the efficiency of the system did not degrade.”
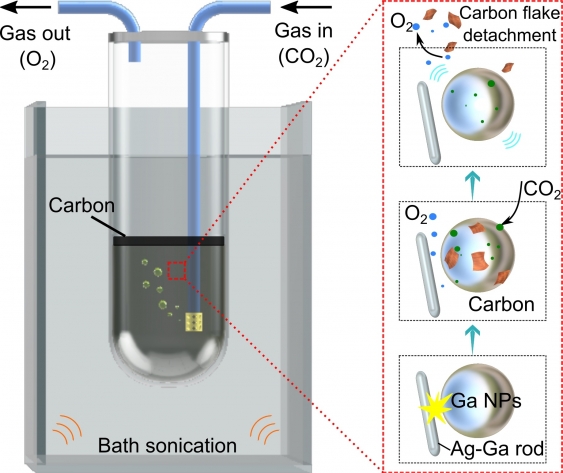
The newly discovered process dissolves captured CO2 gas into a solvent around nanoparticles of gallium, which exist in liquid state above 30°C.
The reactor also contains nano-sized solid silver rods that are the key to generating the triboelectrochemical reactions that take place once mechanical energy (e.g. stirring/mixing) is introduced.
A triboelectrochemical reaction occurs in solid–liquid interfaces due to friction between the two surfaces, with an electric field also created that sparks a chemical reaction.
The reactions break the carbon dioxide into oxygen gas, as well as carbonaceous sheets which ‘float’ to the surface of the container due to differences in density and can therefore be easily extracted.
In their paper, the research team show a 92 per cent efficiency in converting a tonne of CO2 as described, using a low energy input. They estimate it equates to a cost of around $100 per tonne of CO2.
In order to commercialise the research, a spin-out company called LM Plus has been established with the support of UNSW’s Knowledge Exchange – a program that helps transform research discoveries into successful innovations to benefit society, along with seed investment from Uniseed.
UNSW has become a shareholder as part of a license agreement with the company.
“This is a very green process which also produces a high-value carbonaceous sheet which can then be sold and used to make electrodes in batteries, or for carbon fibre materials that are used in high-performance products like aircraft, racing cars, and luxury vehicles,” said Paul Butler, Uniseed’s Investment Manager and director of LM Plus.
“What we are working towards now is to raise funds to build a larger size proof-of-concept for this system to work within a 40-foot container –the size of a truck trailer – that could ultimately help industrial sites immediately capture any CO2 emissions and convert them.”
LM Plus hopes to be able to build that much larger system within 15 months and is already in talks with potential commercial partners about other ways of implementing the new technology.
Original Article: Liquid metal proven to be cheap and efficient CO2 converter
More from: University of New South Wales | University of California Los Angeles | North Carolina State University | Royal Melbourne Institute of Technology | University of Melbourne | Queensland University of Technology | Australian Synchrotron
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Capturing and converting CO2
- Q1 2024 Avidxchange Holdings Inc Earnings Callon May 8, 2024 at 9:36 pm
Tien-Tsin Huang; Analyst; JPMorgan Chase & Co. James Faucette; Analyst; Morgan Stanley & Co. LLC ...
- The 2024 NHL Draft Heads to the Vegas Sphereon May 8, 2024 at 11:05 am
Welcome to 2024, where the Vegas Sphere is still one of the largest talking points in Pro AV. The Pro AV wonder has now hosted U2, helped enhance the first-ever Super Bowl weekend in Las Vegas, and ...
- The science of capturing carbon dioxideon May 6, 2024 at 5:30 am
Carbon dioxide can be converted into useful elements, which could help fight the threat of a greenhouse effect.
- Titan sub disaster may have been caused by ‘micro-buckling’, says engineeron May 3, 2024 at 5:01 pm
Friend of Titan sub victim recalls harrowing moment he learnt of death live on TV A civil engineer has proposed that the fatal implosion of the Titan submersible last year may have been caused by ...
- New Black Ops 2 Aassualt Rifles,Sub-Machine Guns,Light Machine Guns and Pistolson April 25, 2024 at 5:00 pm
Snow to hit parts of UK as temperatures predicted to fall to -3°C Wall Street plunges after US inflation shock Here’s how much water you should really be drinking each day How much money do ...
Go deeper with Google Headlines on:
Capturing and converting CO2
[google_news title=”” keyword=”capturing and converting CO2” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Liquid gallium
- Asia Pacific Gallium Arsenide Wafer Market to Reach USD 2,635.2 Million By 2032, at 16.6% CAGR: Astute Analyticaon May 10, 2024 at 3:31 am
Gallium arsenide wafer market presents a compelling opportunity for growth, driven by its superior performance in high-frequency applications critical for 5G, advanced computing, and next-generation ...
- Researchers discover spontaneous liquefaction of solid metal–liquid metal interfaces in colloidal binary alloyson May 6, 2024 at 7:37 am
The boundary between solid metal and liquid metal can be much less "solid" than we ever suspected. RMIT researchers have discovered that the liquid-solid boundary can fluctuate back and forth, with ...
- Surfaces on the move: dynamic liquefactionon May 4, 2024 at 5:00 pm
The boundary between solid metal and liquid metal can be much less ‘solid’ than we ever suspected. RMIT researchers have discovered that the liquid-solid boundary can fluctuate back and forth, with ...
- Liquid metal synthesis of diamonds achieved at atmospheric pressureon April 30, 2024 at 1:30 am
Diamonds have been synthesised at atmospheric pressure by researchers in South Korea. The researchers do not yet fully understand the underlying mechanism that forms these diamond films, but it ...
- Instant diamonds that take ‘under three hours to grow’ invented by scientists to avoid ‘billion-year’ wait for real gemson April 25, 2024 at 4:39 pm
A DAZZLING breakthrough has figured out how to produce diamonds in just 150 minutes compared to the billion-year natural process. The researchers behind the invention are confident they’ll ...
Go deeper with Google Headlines on:
Liquid gallium
[google_news title=”” keyword=”liquid gallium” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]