Marce Schreier/EPFL
A promising avenue for the future of clean energy is to store it in the form of carbon-based fuels produced from renewable sources, effectively enabling the clean use of liquid fuels such as gasoline.
A first step is the electrolysis of carbon dioxide into oxygen and carbon monoxide, which can be subsequently be transformed into liquid fuels. But current CO-forming catalysts are either not selective enough or too expensive to be industrially viable.
EPFL scientists have now developed an Earth-abundant catalyst based on copper-oxide nanowires modified with tin oxide. A solar-driven system set up using this catalyst was able to split CO2 with an efficiency of 13.4%. The work is published in Nature Energy, and is expected to help worldwide efforts to synthetically produce carbon-based fuels from CO2 and water.
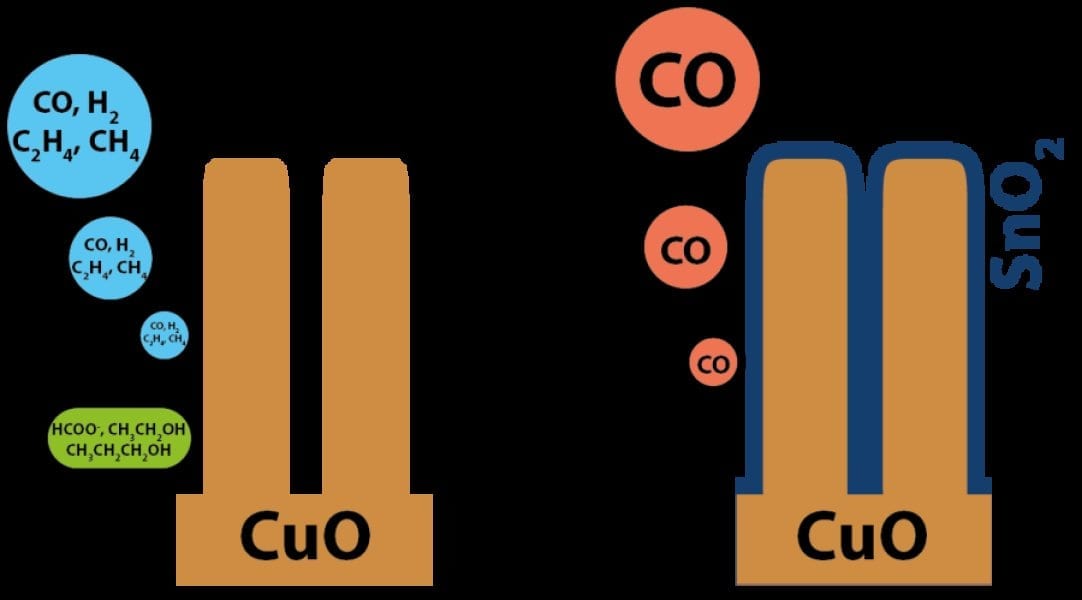
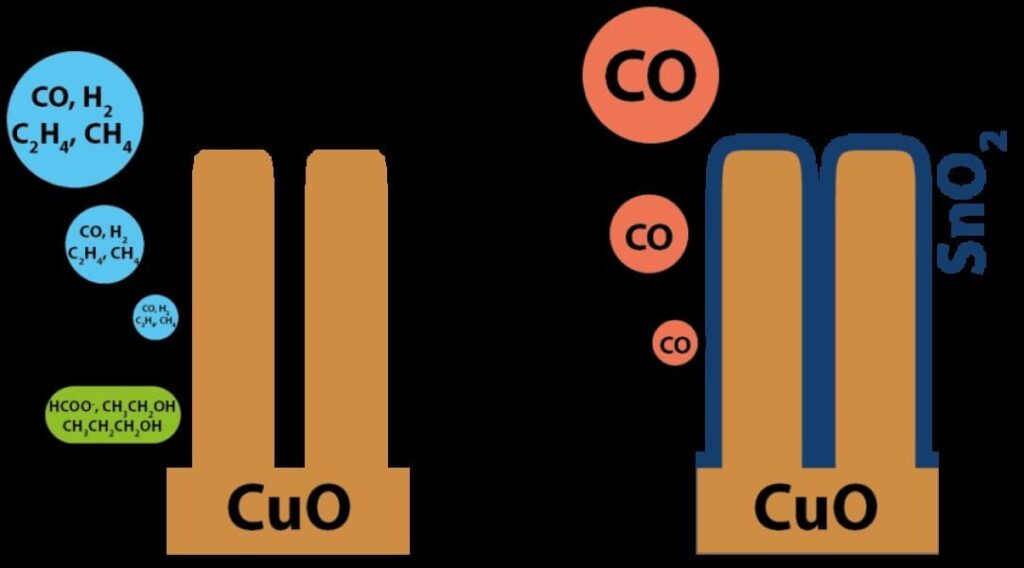
The research was carried out by the lab of Michael Grätzel at EPFL. Grätzel is known worldwide for the invention of dye-sensitized solar cells (“Grätzel cells”). The new catalyst, developed by PhD student Marcel Schreier, postdoc Jingshan Luo, and several co-workers, is made by depositing atomic layers of tin oxide on copper oxide nanowires. Tin oxide suppresses the generation of side-products, which are commonly observed from copper oxide catalysts, leading to the sole production of CO in the electroreduction of CO2.
The catalyst was integrated into a CO2 electrolysis system and linked to a triple-junction solar cell (GaInP/GaInAs/Ge) to make a CO2 photo-electrolyzer. Importantly, the system uses the same catalyst as the cathode that reduces CO2 to CO and the anode that oxidizes water to oxygen through what is known as the “oxygen evolution reaction”. The gases are separated with a bipolar membrane. Using only Earth-abundant materials to catalyze both reactions, this design keeps the cost of the system low.
The system was able to selectively convert CO2 to CO with an efficiency of 13.4% using solar energy. The catalyst also reached a Faradaic efficiency of up to 90%, which describes how efficiently electrical charge is transferred to the desired product in an electrocatalysis system like the one developed here. “The work sets a new benchmark for solar-driven CO2 reduction,” says Luo.
“This is the first time that such a bi-functional and low-cost catalyst is demonstrated,” adds Schreier. “Very few catalysts — except expensive ones, like gold and silver — can selectively transform CO2 to CO in water, which is crucial for industrial applications.”
Learn more: Splitting carbon dioxide using low-cost catalyst materials
[osd_subscribe categories=’CO2-to-fuel’ placeholder=’Email Address’ button_text=’Subscribe Now for any new posts on the topic “CO2 TO FUEL’]
The Latest on: Splitting CO2 into CO and oxygen
[google_news title=”” keyword=”splitting CO2 into CO and oxygen” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- Progress! U.S. energy-related CO2 emissions dropped 3% in 2023
According to the EIA, more than 80% of the emissions reductions occured in the electric power sector due to decreased coal-fired generation.
- Higher CO2 levels mean viruses live longer, infect more
Carbon dioxide is key to how long airborne viruses hang around in the air and, therefore, their likelihood of spreading. Opening a window may be a more scientific way to avoid the spread of ...
- Venus Unveiled: Carbon and Oxygen Caught Escaping Into Space
New findings from BepiColombo show escape of heavy ions from Venus, hinting at complex atmospheric dynamics. A fleeting visit of the European Space Agency (ESA)/Japan Aerospace Exploration Agency (JAX ...
- Virus lifespan and transmission boosted by high CO2 levels
The findings reveal that carbon dioxide (CO2) levels play a critical role in the lifespan and transmission of airborne viruses.
- New electrochemical reactor converts CO2 into usable materials
One of the chemical approaches to DAC involves the use of potassium hydroxide (KOH). This liquid alkaline solution turns the carbon dioxide molecules into more complex compounds called carbonates.
via Google News and Bing News