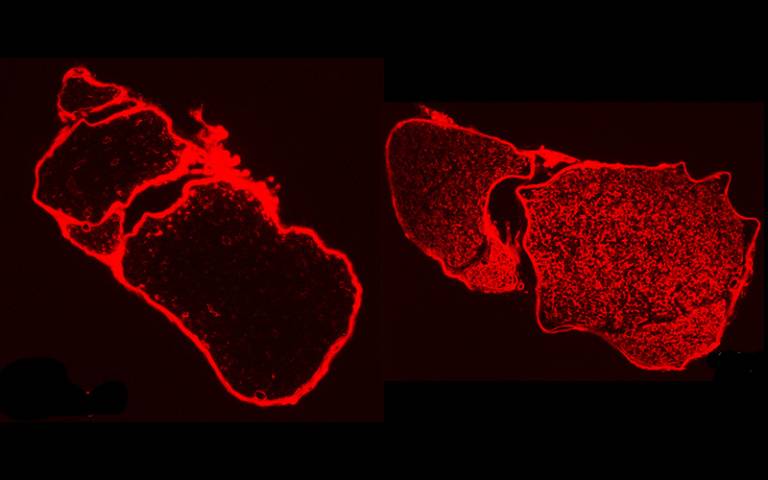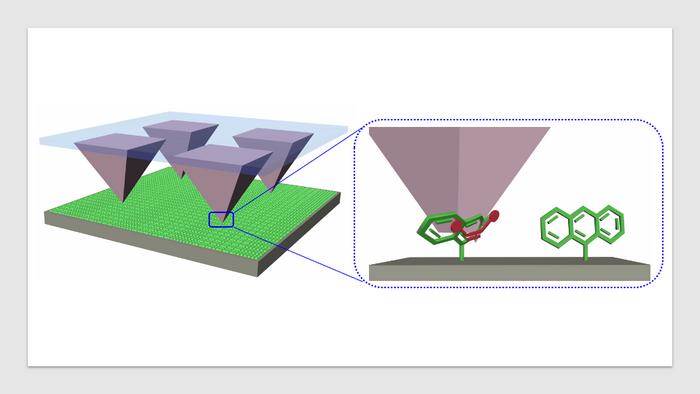
Tip arrays transfer a dienophile molecules (red) onto an anthracene-modified (green) surface. Upon contact, the tips form nanoreactors, where pressure is applied that accelerates the Diels-Alder cycloaddition reactions. For their study, the authors took monolayers of molecules placed on silicon wafers and pushed reactive molecules into them using tip arrays, which created new chemicals. The experimental setup allowed the researchers to precisely control the pressure between the molecules, which led to a new understanding of what occurs in these reactions.
(Image credit: Yerzhan Zholdassov)
The production of chemicals accounts for 40% of all energy currently used in manufacturing, and the process also results in toxic solvent waste that pollutes the environment and poses health risks to humans and animals. A newly published study in the journal Science details a novel mechanochemistry method that has the ability to manufacture chemicals without those deleterious effects.
Researchers with the Nanoscience Initiative at the Advanced Science Research Center at the CUNY Graduate Center (CUNY ASRC), the University of Pennsylvania, and the University of California-Merced took a unique approach that advances the opportunity to use mechanochemistry in large-scale production. The technique uses organic chemistry and nanotechnology to push molecules together and create chemicals without the use of costly solvents that pollute the environment. The research team’s findings have major implications for numerous manufacturing sectors, including the production of pharmaceuticals and materials for a variety of medical and industrial purposes.
“This is a really exciting breakthrough, because the discovery makes mechanochemistry a reliable means of producing chemicals, and it allows us to do so without the harmful byproducts and large energy demands of current manufacturing techniques,” said the study’s lead author Adam Braunschweig, a professor of Chemistry and Biochemistry with the CUNY ASRC Nanoscience Initiative and Hunter College Department of Chemistry.
“When we pushed on the molecules, we found that they twisted into new, more reactive shapes that require less energy to combine and produce a desired chemical,” said first author Yerzhan Zholdassov, a doctoral student with the Braunschweig Lab.
The experiment allowed researchers to measure the amount of force needed to create a predictable and reliable chemical reaction and show that mechanochemistry is a viable and scalable technique for manufacturing chemicals in a more sustainable, cost-efficient manner. The new technique can also be used to create new drugs and materials that can’t be created using current techniques that rely on solvents.
Co-author Robert Carpick, a professor in the University of Pennsylvania’s Department of Mechanical Engineering and Applied Mechanics who collaborated on this project, added: “This discovery was not possible without chemists teaming up with mechanical engineers in a truly cross-disciplinary way. The chemists were critical to designing and conducting the experiments, but we had to combine their forefront chemistry knowledge with advanced mechanics analysis to understand – through experiments and theory – how mechanical forces are accelerating chemical reactions here. The teamwork made the difference.”
Original Article: A novel method for squeezing molecules together could significantly reduce chemical manufacturing waste and its negative environmental impacts
More from: CUNY Graduate Center | University of Pennsylvania | University of California Merced
The Latest Updates from Bing News
Go deeper with Bing News on:
Mechanochemistry
- Scientists are shaking up lithium extraction with a different kind of chemistry
When people think of chemistry, the image that typically comes to mind is a variety of colored liquids in beakers, flasks, and test tubes in a lab. But in actual practice, chemistry can involve ...
- Free tool uses machine learning to pick better molecules for testing new reactions
“Developing a web application is a great way to bring these ideas to a larger part of the community,” says Connor Coley, a computational chemist at the Massachusetts Institute of Technology. He has ...
- Could mechanochemistry have saved Abbott Laboratories $250 million?
The disappearing polymorph of HIV drug ritonavir can now be recovered from its more stable nuisance form by a 15-minute ball milling procedure. Careful control of the milling conditions enabled ...
- A closer look at mechanochemistry
Ferdi Schüth and his team at the Max Planck Institut für Kohlenforschung in Mülheim/Germany have been studying the phenomena of mechanochemistry for several years. But what actually happens at ...
- Mechanochemistry strips cargo molecules from a loaded rotaxane
“Being able to release multiple molecules in a single elongation event—this is completely new in mechanochemistry,” De Bo says. For rotaxanes with a single docking station, ultrasound freed ...
Go deeper with Bing News on:
Accelerating chemical reactions
- Researchers create nanomembrane to increase reaction rate in chemical production
Flow-through reactors packed with enzymes can produce certain chemicals in a gentle and careful way. However, their performance has so far been limited. A research team from the Helmholtz-Zentrum ...
- Air-Stable Ruthenium Catalyst to Revolutionize Industrial Chemistry
Researchers at The University of Manchester have developed an air-stable ruthenium catalyst with wide-ranging applications, promising to streamline optimization processes and support new scientific ...
- Scientists unveil ruthenium catalyst for new reaction discovery and optimization
Researchers at The University of Manchester have developed a new catalyst which has been shown to have a wide variety of uses and the potential to streamline optimization processes in industry and ...
- How scientists are accelerating chemistry discoveries with automation
The advance could help speed the discovery of new pharmaceutical drugs and accelerate the development of new chemical reactions. A new automated workflow developed by scientists at Lawrence ...
- Scientists unveil cutting-edge ruthenium catalyst for new reaction discovery and optimisation
Catalysts, often considered the unsung heroes of chemistry, are instrumental in accelerating chemical reactions, and play a crucial role in the creation of most manufactured products. For example, the ...