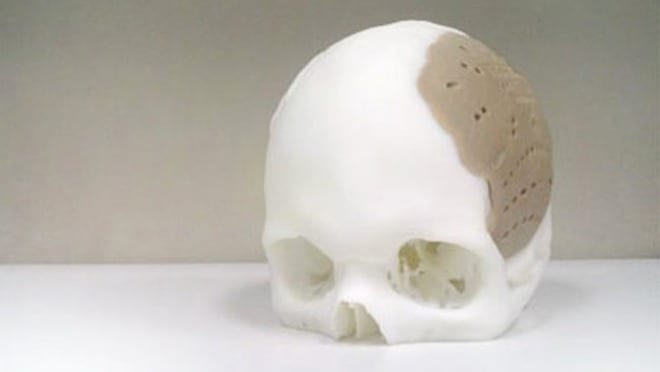
OsteoFab is the coolest 3-D printed material you’d never want to use.
Invented by plastics research firm Oxford Performance Materials (OPM), this first-of-its-kind polyketone can be used by 3-D printers to repair large sections of a damaged skull. It was recently cleared for use by the FDA and is being used in critical surgical procedures all over the world.
The typical OsteoFab customer has been in a car accident or suffered other physical trauma. In the emergency room, doctors stabilize the patient and and get a CAT scan. The OPM engineering team builds on the data from the scan and creates printable CAD files that feature screw holes and scaffolding necessary for implantation. A surgeon approves the design and it’s printed at OPM using a selective laser sintering 3-D printer. A technician laser-scans the final part for quality control and ships it to the hospital where it’s sterilized and implanted.
The treatment can take several months and and costs $8,000-15,000, but also provides benefits no other process can match. Since the implant is custom built to fit the patient, it makes surgery quicker which saves money and can help reduce the chance of complications. It also reduces cosmetic defects caused by off-the-shelf parts.
Despite this futuristic application, OPM’s super-high-performance thermoplastic has less dramatic beginnings. “We’re at the top of the polymer food chain,” says OPM president Scott DeFelice. “Our material has ended up in some interesting spots, like semiconductor manufacturing when you need pure materials with no outgassing, or in high pressure, high temperature pipe connectors at the bottom of the North Sea.”
The polymer was also deemed well suited for spinal implants since it is biocompatible, has mechanical properties similar to bone, and also provokes a positive cellular response that allows new bone to grow on top of the implants. But, there were also limitations — each implant was labor intensive, which introduced the opportunity for defects.
“Seven years ago I picked up an article and it was about surgeons dreaming of printing implants,” explains DeFelice, who says he immediately recognized the opportunity and called on his team of polymer scientists, mechanical engineers, and clinical experts to explore the potential. “We looked at our materials technology and realized that we could make some changes and it could work,” he says.
Unlike most 3-D printing entrepreneurs who can jut put their products on Kickstarter or an ecommerce page, OPM had to submit their product to the FDA for evaluation. “Their process was very thoughtful,” says DeFelice. “They understood the technology and benefits, both for the patient and the opportunity for savings by adopting patient specific solutions.”
He also adds that it doesn’t hurt that the president is talking about 3-D printing. The material was cleared for use by the FDA in February, but has been available in Europe and used successfully in procedures already.
The Latest Bing News on:
OsteoFab
- Feed has no items.
The Latest Google Headlines on:
OsteoFab
[google_news title=”” keyword=”OsteoFab” num_posts=”10″ blurb_length=”0″ show_thumb=”left”] [/vc_column_text]The Latest Bing News on:
3-D Printed Skull Implants
- 3D printers are helping surgeons create custom cranial implantson April 26, 2024 at 11:28 am
I know that if I suffer a severe injury, especially to my skull, all I really need is a 3D printer. Now that I’m in my 40s, I’m more acutely aware of the inherent risk of injury in normal, everyday ...
- Global 3D Printed Medical Implants Industryon April 23, 2024 at 8:06 am
Global 3D Printed Medical Implants Industry is projected to reach worth of US$ 2,494.5 million by 2032, expanding at a CAGR of 12.5% by 2032 ...
- 3-D Printed Cranial Implant Gets 510(k) Clearanceon April 22, 2024 at 4:09 pm
D-printed PEEK implants using the EXT 220 MED were successfully demonstrated in nearly 40 cranioplasties across Europe.
- FDA Approves Innovative 3D-Printed Cranial Implants for US Market Entryon April 16, 2024 at 11:10 pm
VSP® PEEK Cranial Implant is designed to address defects in the skull resulting from traumatic injuries like fractures or gunshot wounds.
- In a first, FDA approves 3D-printed cranial implants to enter USon April 16, 2024 at 9:23 am
Now, the Food and Drug Administration (FDA) has cleared a customized 3D-printed solution to precisely fit a patient’s skull. The clearance by the FDA has paved the way for the w ...
- 3D Systems achieves FDA 510(k) clearance for 3D printed PEEK cranial implanton April 16, 2024 at 5:19 am
The VSP PEEK Cranial Implant solution includes a complete workflow comprising segmentation and 3D modelling software, the 3D Systems EXT 220 MED 3D printer, Evonik VESTAKEEP i4 3DF PEEK and a ...
- 3D Systems obtains 510(k) clearance for 3D-printed cranial implantson April 16, 2024 at 4:19 am
3D Systems has received 510 (k) clearance from the US Food and Drug Administration (FDA) for its VSP PEEK Cranial Implant. The patient-specific implant includes the EXT 220 MED 3D printer and ...
- 3D Systems announces FDA clearance for 3D printed PEEK cranial implantson April 16, 2024 at 2:16 am
The FDA has provided 510(k) clearance for 3D Systems 3D-printed, patient-specific cranial implant solution — VSP PEEK Cranial Implant.
- FDA clears 3D-printed, patient-specific cranial implant from 3D Systemson April 15, 2024 at 8:41 am
D Systems announced today that the FDA granted 510(k) clearance for its 3D-printed, patient-specific VSP PEEK cranial implant.
- 3D Systems Announces FDA Clearance for World’s First 3D-Printed PEEK Cranial Implantson April 15, 2024 at 7:30 am
FDA clearance enables wide-spread adoption of 3D Systems self-contained, cleanroom environment-based printing system, the EXT 220 MED, with ...
The Latest Google Headlines on:
3-D Printed Skull Implants
[google_news title=”” keyword=”3-D Printed Skull Implants” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]