via EPFL
Bioengineers at EPFL have created miniature intestines in a dish that match up anatomically and functionally to the real thing better than any other lab-grown tissue models. The biological complexity and longevity of the new organoid technology is an important step towards enabling drug testing, personalized medicine, and perhaps, one day, transplantations.
Organoids are fast-becoming one of the most cutting-edge tools of modern life sciences. The idea is to use stem cells to build miniature tissues and organs that accurately resemble and behave like their real counterparts.
One can immediately appreciate the value of organoids for both research and medicine: from basic biological research to drug development and testing, organoids could complement animal testing by providing healthy or diseased human tissues, expediting the lengthy journey from lab to clinical trial. Beyond that, there is already the whisper of organoid technology perhaps being used for replacing damaged tissues or even organs in the future: take stems cells from the patient and grow them into a new liver, heart, kidney, or lung.
So far, established methods of making organoids come with considerable drawbacks: stem cells develop uncontrollably into circular and closed tissues that have a short lifespan, as well as non-physiological size and shape, all of which result in overall anatomical and/or physiological inconsistency with real-life organs.
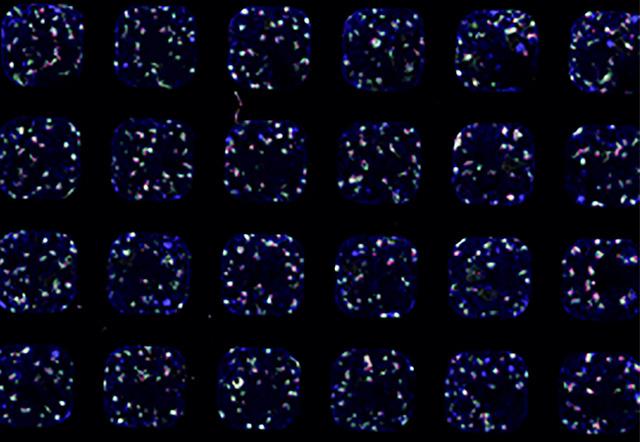
Now, scientists from the group led by Matthias Lütolf at EPFL‘s Institute of Bioengineering have found a way to “guide” stem cells to form an intestinal organoid that looks and functions just like a real tissue. Published in Nature, the method exploits the ability of stem cells to grow and organize themselves along a tube-shaped scaffold that mimics the surface of the native tissue, placed inside a microfluidic chip (a chip with little channels in which small amounts of fluids can be precisely manipulated).
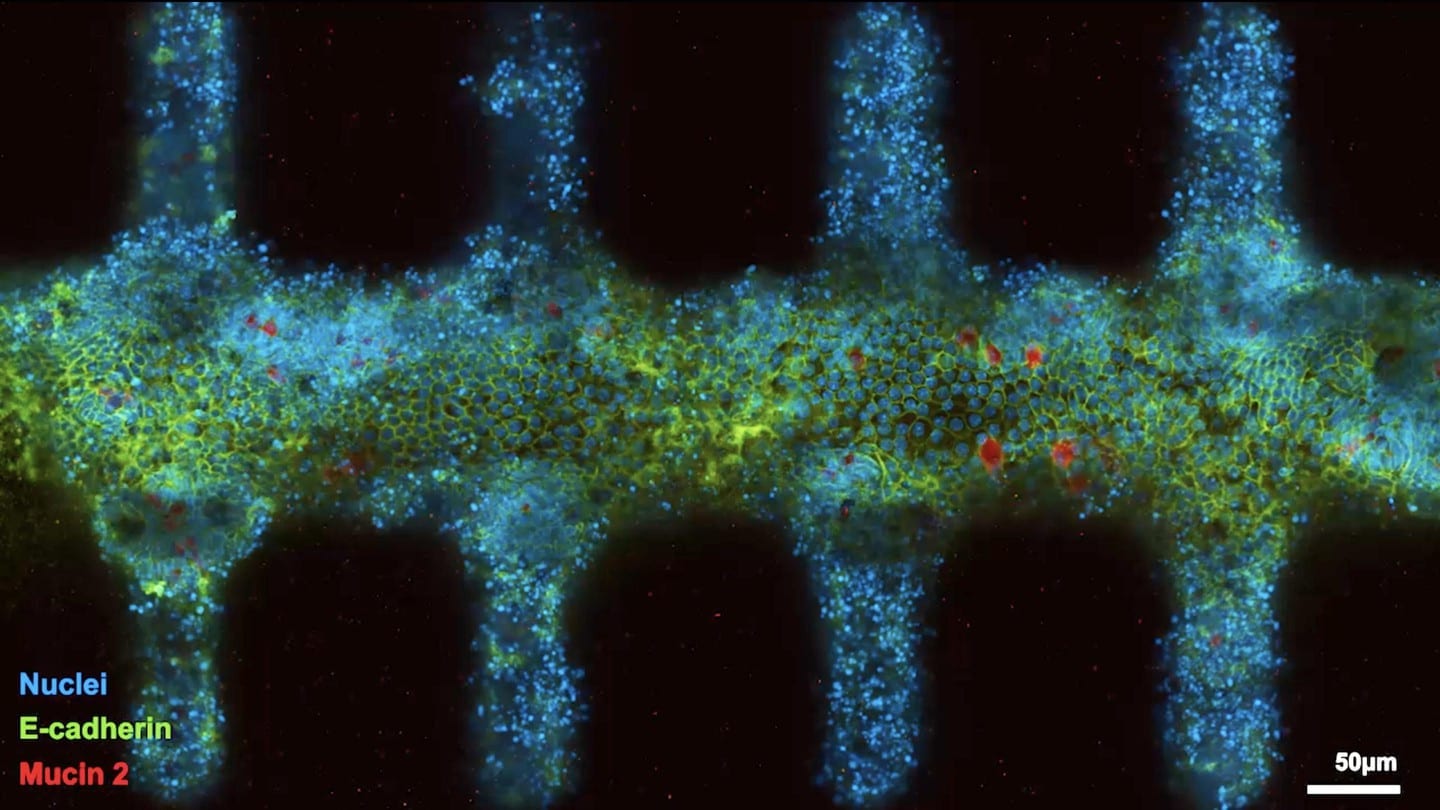
The EPFL researchers used a laser to sculpt this gut-shaped scaffold within a hydrogel, a soft mix of crosslinked proteins found in the gut’s extracellular matrix supporting the cells in the native tissue. Aside from being the substrate on which the stem cells could grow, the hydrogel thus also provides the form or “geometry” that would build the final intestinal tissue.
Once seeded in the gut-like scaffold, within hours, the stem cells spread across the scaffold, forming a continuous layer of cells with its characteristic crypt structures and villus-like domains. Then came the surprise: the scientists found that, the stem cells just “knew” how to arrange themselves in order to form a functional tiny gut.
“It looks like the geometry of the hydrogel scaffold, with its crypt-shaped cavities, directly influences the behavior of the stem cells so that they are maintained in the cavities and differentiate in the areas outside, just like in the native tissue,” says Lütolf. The stem cells didn’t just adopt to the shape of the scaffold, they produced all the key differentiated cell types found in the real gut, with some rare and specialized cell types normally not found in organoids.
Intestinal tissues are known for the highest cell turnover rates in the body, resulting in a massive amount of shed dead cells accumulating in the lumen of the classical organoids that grow as closed spheres and require weekly breaking down into small fragments to maintain them in culture. “The introduction of a microfluidic system allowed us to efficiently perfuse these mini-guts and establish a long-lived homeostatic organoid system in which cell birth and death are balanced,” says Mike Nikolaev, the first author of the paper.
The researchers demonstrate that these miniature intestines share many functional features with their in vivo counterparts. For example, they can regenerate after massive tissue damage and they can be used to model inflammatory processes or host-microbe interactions in a way not previously possible with any other tissue model grown in the laboratory.
In addition, this approach is broadly applicable for the growth of miniature tissues from stem cells derived from other organs such as the lung, liver or pancreas, and from biopsies of human patients. “Our work shows that tissue engineering can be used to control organoid development and build next-gen organoids with high physiological relevance, opening up exciting perspectives for disease modelling, drug discovery, diagnostics and regenerative medicine,” says Lütolf.
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Organoids
- Global Organoids and Spheroids Market: Journey to US$4,567.0 Million Revenue by 2031
Global Organoids and Spheroids Market was valued at US$ 771.4 million in 2022 and is projected to attain a valuation of US$ 4,567.0 Million by 2031 at a CAGR of 22.42% During the Forecast Period ...
- Human Organoids Market CAGR of 17.5%, Big Money to be Made as Market Size Continues to Surge
Human Organoids Market is valued approximately at USD 723.40 million in 2019 and is anticipated to grow with a healthy growth rate of more than 17.5% over the forecast period 2020-2027. Organoids are ...
- Fetal Organoids Generated From Human Amniotic Fluid
Over the last decade, organoids have become an increasingly popular platform for modeling organ function and disease. 3 However, the generation of fetal organoids is complicated by ethical and legal ...
- Scientists develop new organoid model to study thymus function
Researchers from the Organoid group have developed a new organoid model that can be used to study the thymus. The organoids are derived from mouse thymus tissue, specifically model thymic epithelial ...
- Lab-made miniorgans take scientists a step closer to curing cancer
In the fight to cure cancer, scientists need better models. And they might just get them — human-derived organoids — thanks to light.
Go deeper with Google Headlines on:
Organoids
[google_news title=”” keyword=”organoids” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Organoid technology
- Technology & Innovation
Technological innovations have produced robots capable of jobs that, until recently, only humans could perform. The present research explores the psychology of "botsourcing"—the replacement of human ...
- Researchers develop first model of the brain's information highways
Like country roads, small connections link neighboring nerve cells, while, like highways, thick nerve bundles connect different regions of the brain. These thick, heavily used nerve bundles, which for ...
- New Organoid Model Offers Insights Into Immune System Training
Researchers from the Organoid group have developed a new organoid model that can be used to study the thymus. The organoids, derived from mouse thymus tissue, specifically model thymic epithelial ...
- Scientists develop new organoid model to study thymus function
Researchers from the Organoid group have developed a new organoid model that can be used to study the thymus. The organoids are derived from mouse thymus tissue, specifically model thymic epithelial ...
- Scientists develop new organoid model to study thymus function
Cell Reports, 2024. H.C. is the head of Pharma Research and Early Developmentat Roche, Basel, and holds several patents related to organoid technology. The full disclosure is given at https://www ...
Go deeper with Google Headlines on:
Organoid technology
[google_news title=”” keyword=”organoid technology” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]