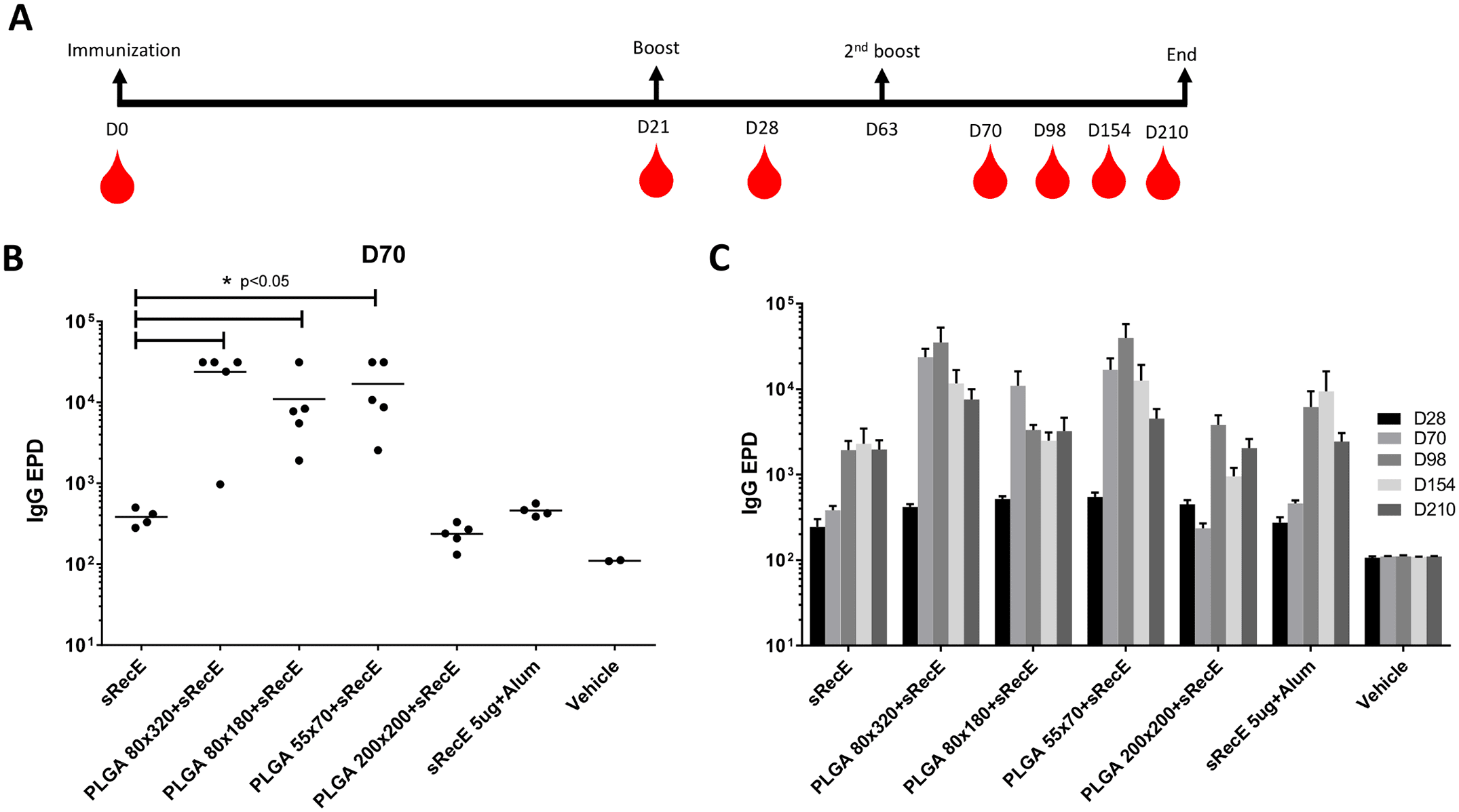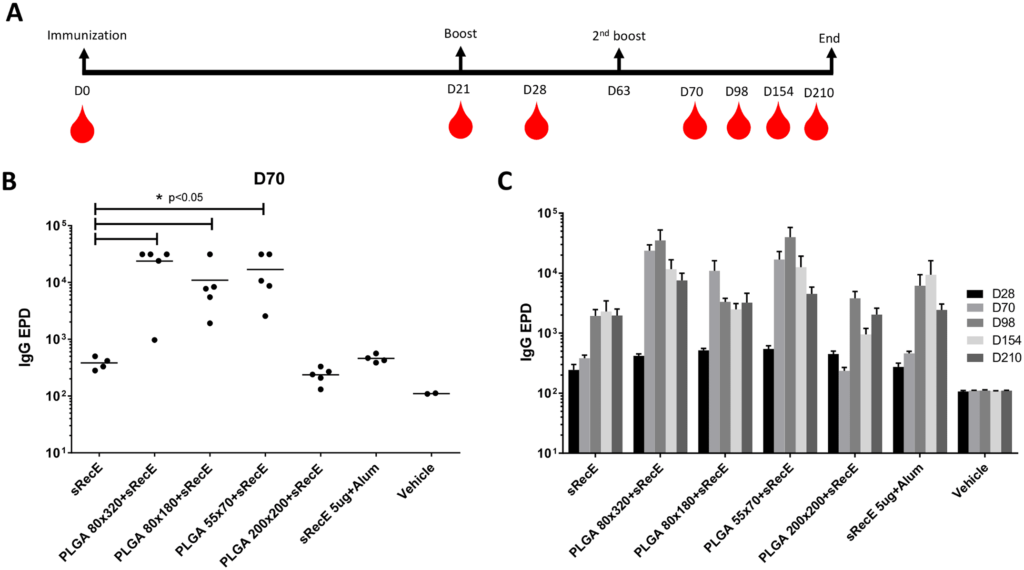
A) TEM analysis of 80×320 nm, 80×180 nm, 55×70 nm and 200×200 nm PLGA particles. B) sRecE was adsorbed to the PLGA nanoparticles in variable sRecE/PLGA (w/w%) ratios. Increasing sRecE/PLGA ratios were obtained by fixing sRecE and decreasing the particle mass. Adsorption efficiency was determined by the amount of non-adsorbed sRecE after the spinning down the particles
Dengue virus (DENV) is the causative agent of dengue fever and dengue hemorrhagic fever. The virus is endemic in over 120 countries, causing over 350 million infections per year.
Dengue vaccine development is challenging because of the need to induce simultaneous protection against four antigenically distinct DENV serotypes and evidence that, under some conditions, vaccination can enhance disease due to specific immunity to the virus. While several live-attenuated tetravalent dengue virus vaccines display partial efficacy, it has been challenging to induce balanced protective immunity to all 4 serotypes. Instead of using whole-virus formulations, we are exploring the potentials for a particulate subunit vaccine, based on DENV E-protein displayed on nanoparticles that have been precisely molded using Particle Replication in Non-wetting Template (PRINT) technology.
Here we describe immunization studies with a DENV2-nanoparticle vaccine candidate. The ectodomain of DENV2-E protein was expressed as a secreted recombinant protein (sRecE), purified and adsorbed to poly (lactic-co-glycolic acid) (PLGA) nanoparticles of different sizes and shape. We show that PRINT nanoparticle adsorbed sRecE without any adjuvant induces higher IgG titers and a more potent DENV2-specific neutralizing antibody response compared to the soluble sRecE protein alone. Antigen trafficking indicate that PRINT nanoparticle display of sRecE prolongs the bio-availability of the antigen in the draining lymph nodes by creating an antigen depot. Our results demonstrate that PRINT nanoparticles are a promising platform for delivering subunit vaccines against flaviviruses such as dengue and Zika.
Author Summary
Dengue virus (DENV) is transmitted by mosquitoes and is endemic in over 120 countries, causing over 350 million infections yearly. Most infections are clinically unapparent, but under specific conditions, dengue can cause severe and lethal disease. DENV has 4 distinct serotypes and secondary DENV infections are associated with hemorrhagic fever and dengue shock syndrome. This enhancement of infection complicates vaccine development and makes it necessary to induce protective immunity against all 4 serotypes. Since whole virus vaccine candidates struggle to induce protective immunity, we are developing a nanoparticle display vaccine approach. We have expressed, purified and characterized a soluble recombinant E-protein (sRecE). Regardless of nanoparticle shape or size, particulation of sRecE enhances DENV specific IgG titers and induces a robust, long lasting neutralizing antibody response and by adsorbing sRecE to the nanoparticles, we prolong the exposure of sRecE to the immune system.
Nanoparticle display shows great promise in dengue vaccine development and possibly other mosquito-borne viruses like zika virus.
The Latest on: Nanoparticle vaccines
[google_news title=”” keyword=”Nanoparticle vaccines” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Nanoparticle vaccines
- Personalised Mrna Vaccines: A Revolutionary New Approach In Melanoma Treatmenton May 2, 2024 at 11:45 am
A personalised mRNA vaccine to treat melanoma has now reached late-stage trials in the UK. This is just the latest step in improving the cure rate o ...
- Brain cancer in children is notoriously hard to treat—a new mRNA cancer vaccine triggers an attack from withinon May 2, 2024 at 8:58 am
Our cancer vaccine seeks to address these issues in a number of ways. Also, instead of injecting nanoparticles into the skin, muscle or directly into the tumor, as is commonly done for many ...
- An experimental vaccine increases survival rates by up to 50% in four people with highly-aggressive brain canceron May 2, 2024 at 5:09 am
Spanish biochemist Héctor Méndez and his colleagues from the University of Florida have achieved ‘promising’ results in their initial human trial ...
- RNA glioblastoma vaccine shows promise in first human trialon May 2, 2024 at 3:01 am
The vaccine – developed by researchers at the University of Florida (UF) in the US – is based on mRNA taken from the patient’s tumour that is delivered in a lipid nanoparticle (LNP) casing that ...
- New mRNA Vaccines in Development for Cancer and Infectionson May 2, 2024 at 2:22 am
Prelog also discussed personalized vaccines for cancer immunotherapy. Personalized mRNA vaccines are tailored to the patient's genetic characteristics and antigens. They could be used in cancer ...
- mRNA Brain Cancer Vaccine Shows Promise In Early Trials In Humans – And Dogson May 2, 2024 at 1:12 am
A new mRNA vaccine candidate for aggressive brain cancer has shown promise in a small clinical trial – but it was a clinical trial with a twist. As well as testing the vaccine in a handful of human ...
- Q&A: AstraZeneca's COVID-19 vaccine and blood clots—what you need to knowon May 1, 2024 at 9:24 am
Pharmaceutical giant AstraZeneca admitted in court this week that its COVID-19 vaccine can cause a rare but deadly blood-clotting condition that has become the central focus of a class-action lawsuit ...
- Vaccine Developers Leverage mRNA and Other Powerful Technologieson April 30, 2024 at 5:00 pm
Also, lipid nanoparticle technology is becoming safer and more stable, helping it to become the technology of choice for delivering mRNA-based vaccines. There are also opportunities to use artificial ...
- Tolerizing vaccines for autoimmune diseases tempt BMS into $1.8bn collaborationon April 30, 2024 at 7:10 am
Bristol Myers Squibb partners with Repertoire Immune Medicines to develop innovative vaccines aimed at treating autoimmune disorders more selectively and effectively ...
via Bing News