Cotton candy machines may hold the key for making life-sized artificial livers, kidneys, bones and other essential organs.
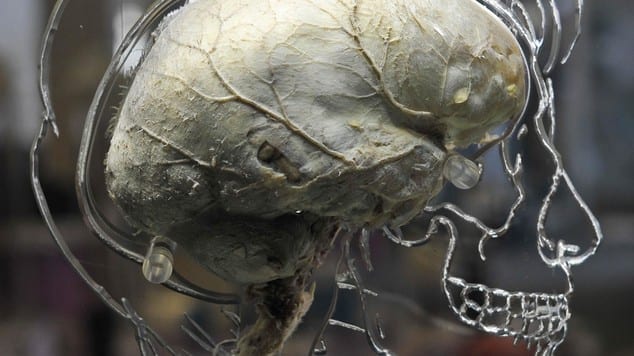
For several years, Leon Bellan, assistant professor of mechanical engineering at Vanderbilt University, has been tinkering with cotton candy machines, getting them to spin out networks of tiny threads comparable in size, density and complexity to the patterns formed by capillaries – the tiny, thin-walled vessels that deliver oxygen and nutrients to cells and carry away waste. His goal has been to make fiber networks that can be used as templates to produce the capillary systems required to create full-scale artificial organs.”
In an article published online on Feb. 4 by the Advanced Healthcare Materials journal, Bellan and colleagues report that they have succeeded in using this unorthodox technique to produce a three-dimensional artificial capillary system that can keep living cells viable and functional for more than a week, which is a dramatic improvement over current methods.
“Some people in the field think this approach is a little crazy,” said Bellan, “But now we’ve shown we can use this simple technique to make microfluidic networks that mimic the three-dimensional capillary system in the human body in a cell-friendly fashion. Generally, it’s not that difficult to make two-dimensional networks, but adding the third dimension is much harder; with this approach, we can make our system as three-dimensional as we like.”
Many tissue engineering researchers, including Bellan, are currently focusing their efforts on a class of materials similar to hair gel – water-based gels, called hydrogels – and using these materials as scaffolds to support cells within three-dimensional artificial organs.
Hydrogels are attractive because their properties can be tuned to closely mimic those of the natural extracellular matrix that surrounds cells in the body. Unlike solid polymer scaffolds, hydrogels support diffusion of necessary soluble compounds; however, oxygen, nutrients and wastes can only diffuse a limited distance through the gel. As a result, cells must be very close (within the width of human hair) to a source of nutrients and oxygen and a sink for the wastes they produce, otherwise they starve or suffocate.
So, to engineer tissues that have the thickness of real organs and keep cells alive throughout the entire scaffold, the researchers must build in a network of channels that allow fluids to flow through the system, mimicking the natural capillary system.
In the bottom-up process, scientists culture cells in a thin slab of gel, and after some time they spontaneously begin creating capillaries. Although this approach has the advantage of simplicity, it has one fundamental problem: It can take weeks for the cells to create such a network. So it isn’t possible to stack the cells too high or the ones in the center begin dying off before the crucial capillary network forms.
As a result, Bellan is using a top-down approach. He reports that his cotton-candy spinning method can produce channels ranging from three to 55 microns, with a mean diameter of 35 microns. “So far the other top-down approaches have only managed to create networks with microchannels larger than 100 microns, about ten times the size of capillaries,” he said. In addition, many of these other techniques are not able to form networks as complex as the cotton candy approach.
Learn more: Cotton candy machines may hold key for making artificial organs
The Latest on: Artificial organs
[google_news title=”” keyword=”artificial organs” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Artificial organs
- OpenAI considers allowing users to create AI-generated pornographyon May 9, 2024 at 3:49 pm
Critics say ChatGPT creator’s proposal to allow erotica, slurs and other adult content undermines its mission statement ...
- How eating ultra-processed foods could lead to increased risk of death, study showson May 9, 2024 at 10:43 am
Whole foods in their natural state -- such as fresh fruit and vegetables, grains and lean proteins -- are up against rapidly growing options for ready-to-eat, ultra-processed packaged bites that ...
- New fluidic system advances development of artificial blood vessels and biomedicine applicationson May 9, 2024 at 7:05 am
Nature consistently inspires engineering applications. Recently, a group of researchers from the Faculty of Engineering at the University of Hong Kong (HKU) drew new inspiration from the vascular ...
- Artificial Intelligenceon May 8, 2024 at 4:33 am
By Erica L. Green The prominent A.I. start-up is also joining an industrywide effort to spot content made with artificial intelligence. By Cade Metz and Tiffany Hsu The suit, which accuses the ...
- Patient in Ankara with rare disease relies on portable artificial lungon May 8, 2024 at 2:20 am
Aykut Inciler, a 60-year-old resident of Ankara, Türkiye, has been coping with pulmonary hypertension, a rare form of high blood pressure, for ...
- Fruit fly model identifies key regulators behind organ developmenton May 7, 2024 at 5:03 pm
A new computational model simulating fruit fly wing development has enabled researchers to identify previously hidden mechanisms behind organ generation. An research team developed a fruit fly model ...
- Two studies aim to fight cancer with artificial cellson May 5, 2024 at 5:00 pm
The artificial cells will interact with organoids, three-dimensional aggregates of cells normally used in research to replicate human tissues and organs as miniaturized and simplified models, which ...
- Research team develop porous sponge material for enhancing kidney hemostasis and repairon May 2, 2024 at 11:18 am
A POSTECH research team have crafted a material aimed at swiftly staunching kidney bleeding and facilitating wound recovery. Their research featured in the online edition of Biomaterials.
- First Look presents a fine look at the future (or is it now?) with THE A.I. AT DELPHI.on May 1, 2024 at 7:26 am
THE A.I. AT DELPHI, a new play by Bella Poynton, directed by Jeffrey Coyle, presented by First Look Buffalo, starring: Melinda Capeles, Lisa Ludwig, Jon May, and Anthony ...
- artificial intelligenceon April 30, 2024 at 8:02 am
Microsoft plans to invest $3.3 billion in an artificial intelligence-related initiative in Wisconsin, giving a boost to President Joe Biden’s job-creation efforts in the key election ...
via Bing News