All good research breaks new ground, but rarely does the research unearth truths that challenge the foundation of a science.
That’s what Artem R. Oganov has done, and the professor of theoretical crystallography in the Department of Geosciences will have his work published in the Dec. 20 issue of the journal Science.
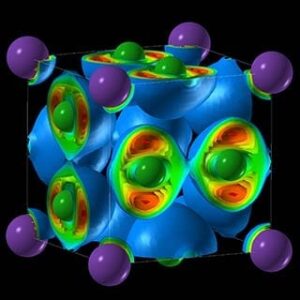
The paper titled “Unexpected stable stoichiometries of sodium chlorides,” documents his predictions about, and experiments in, compressing sodium chloride—rock salt—to form new compounds. These compounds validate his methodology for predicting the properties of objects—a methodology now used worldwide for computational material discovery—and hold the promise of novel materials and applications.
“I think this work is the beginning of a revolution in chemistry,” Oganov says. “We found, at low pressures achievable in the lab, perfectly stable compounds that contradict the classical rules of chemistry. If you apply the rather modest pressure of 200,000 atmospheres—for comparison purposes, the pressure at the center of the earth is 3.6 million atmospheres—everything we know from chemistry textbooks falls apart.”
Standard chemistry textbooks say that sodium and chlorine have very different electronegativities, and thus must form an ionic compound with a well-defined composition. Sodium’s charge is +1, chlorine’s charge is -1; sodium will give away an electron, chlorine wants to take an electron. According to chemistry texts and common sense, the only possible combination of these atoms in a compound is 1:1—rock salt, or NaCl.
“We found crazy compounds that violate textbook rules—NaCl3, NaCl7, Na3Cl2, Na2Cl, and Na3Cl,” says Weiwei Zhang, the lead author and visiting scholar at the Oganov lab and Stony Brook’s Center for Materials by Design, directed by Oganov. “These compounds are thermodynamically stable and, once made, remain indefinitely; nothing will make them fall apart. Classical chemistry forbids their very existence. Classical chemistry also says atoms try to fulfill the octet rule—elements gain or lose electrons to attain an electron configuration of the nearest noble gas, with complete outer electron shells that make them very stable. Well, here that rule is not satisfied.”
This opens all kinds of possibilities. Oganov posited that, if you mix NaCl with metallic sodium, compress in a diamond anvil cell, and heat, you will get sodium-rich compounds like Na3Cl. He likewise theorized that, if you take NaCl, mix it with pure chlorine, and compress and heat, you will get chlorine-rich compounds such as NaCl3. This is exactly what was seen in the experiments, which were performed by the team of Alexander F. Goncharov of Carnegie Institution of Washington, confirming Oganov’s predictions. “When you change the theoretical underpinnings of chemistry, that’s a big deal,” Goncharov says. “But what it also means is that we can make new materials with exotic properties.”
Among the compounds Oganov and his team created are two-dimensional metals, where electricity is conducted along the layers of the structure. “One of these materials—Na3Cl—has a fascinating structure,” he says. “It is comprised of layers of NaCl and layers of pure sodium. The NaCl layers act as insulators; the pure sodium layers conduct electricity. Systems with two-dimensional electrical conductivity have attracted a lot of interest.”
Like much of science, Oganov’s pursuit began with curiosity—and obstinacy.
The Latest on: Novel materials
[google_news title=”” keyword=”Novel materials” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Novel materials
- UMaine’s novel polymer 3D printer surpasses former Guinness World Recordon April 26, 2024 at 6:03 pm
Four times larger than its predecessor, FoF 1.0 offers new opportunities in composite materials, including development of recycled and bio-based materials.
- Book Club: Let’s Talk About ‘Good Material,’ by Dolly Aldertonon April 26, 2024 at 12:57 pm
The writer Dolly Alderton has long had an avid following in her native England, but with her best-selling comic novel “Good Material” she’s become a trans-Atlantic success.
- Author releases debut novel “The Waves Take You Home”on April 26, 2024 at 9:00 am
It’s no doubt a big accomplishment to publish a novel, especially when it’s the author’s first one. That’s the case for Colombia-born María Alejandra ...
- Thiol-ene click reaction offers a novel approach to fabricate elastic ferroelectricson April 26, 2024 at 6:23 am
A research group led by Prof. Li Runwei and Hu Benlin at the Ningbo Institute of Materials Technology and Engineering (NIMTE) of the Chinese Academy of Sciences has developed a facile and efficient ...
- Towards novel promising perovskite-type ferroelectric materials: High-pressure synthesis of rubidium niobateon April 25, 2024 at 1:37 pm
Capacitors are crucial components in electronic devices such as smartphones and computers. They are made of dielectric materials that polarize on the application of the voltage. Currently, barium ...
- Novel Anode Material Boosts Lithium-Ion Battery Performanceon April 25, 2024 at 9:16 am
Dr. Han Joong Tark and student researcher Lee Do Geun at Korea Electrotechnology Research Institute's Nano Hybrid Technology Research Center have devised a novel manufacturing method for ...
- Methane Eating Microbes - A Novel Solution For Greenhouse Gas Mitigationon April 25, 2024 at 7:00 am
Methane is a significant contributor to human-driven greenhouse gas emissions, but there is useful group of microbes that can help because they use methane for food.
- Near Infrared (NIR) Absorbing Material Market Set to Surge to US$ 764 Million by 2034, Expanding at 8.4% CAGRon April 24, 2024 at 7:52 pm
The near infrared (NIR) absorbing material market valuation is estimated to be US$ 764 million by 2034, projected at a CAGR of 8.4%.
- A novel universal light-based technique to control valley polarization in bulk materialson April 24, 2024 at 9:52 am
Electrons inside solid materials can only take certain values of energy. The allowed energy ranges are called "bands," and the space between them, the forbidden energies, is known as "band gaps." Both ...
- TerraSafe Materials and North Carolina State University Announce Licensing Agreement to Commercialize Novel Material Technologieson April 23, 2024 at 6:00 am
TerraSafe continues to grow its IP portfolio to commercialize sustainable packaging solutionsNEW YORK, April 23, 2024 (GLOBE NEWSWIRE) -- TerraSafe Materials, Inc. (“TerraSafe” or the “Company”), the ...
via Bing News