A molecular system that holds great promise for the capture and storage of carbon dioxide has been modified so that it now also holds great promise as a catalyst for converting captured carbon dioxide into valuable chemical products.
The sponge-like quality of a COF’s vast internal surface area enables the system to absorb and store enormous quantities of targeted molecules, such as carbon dioxide.
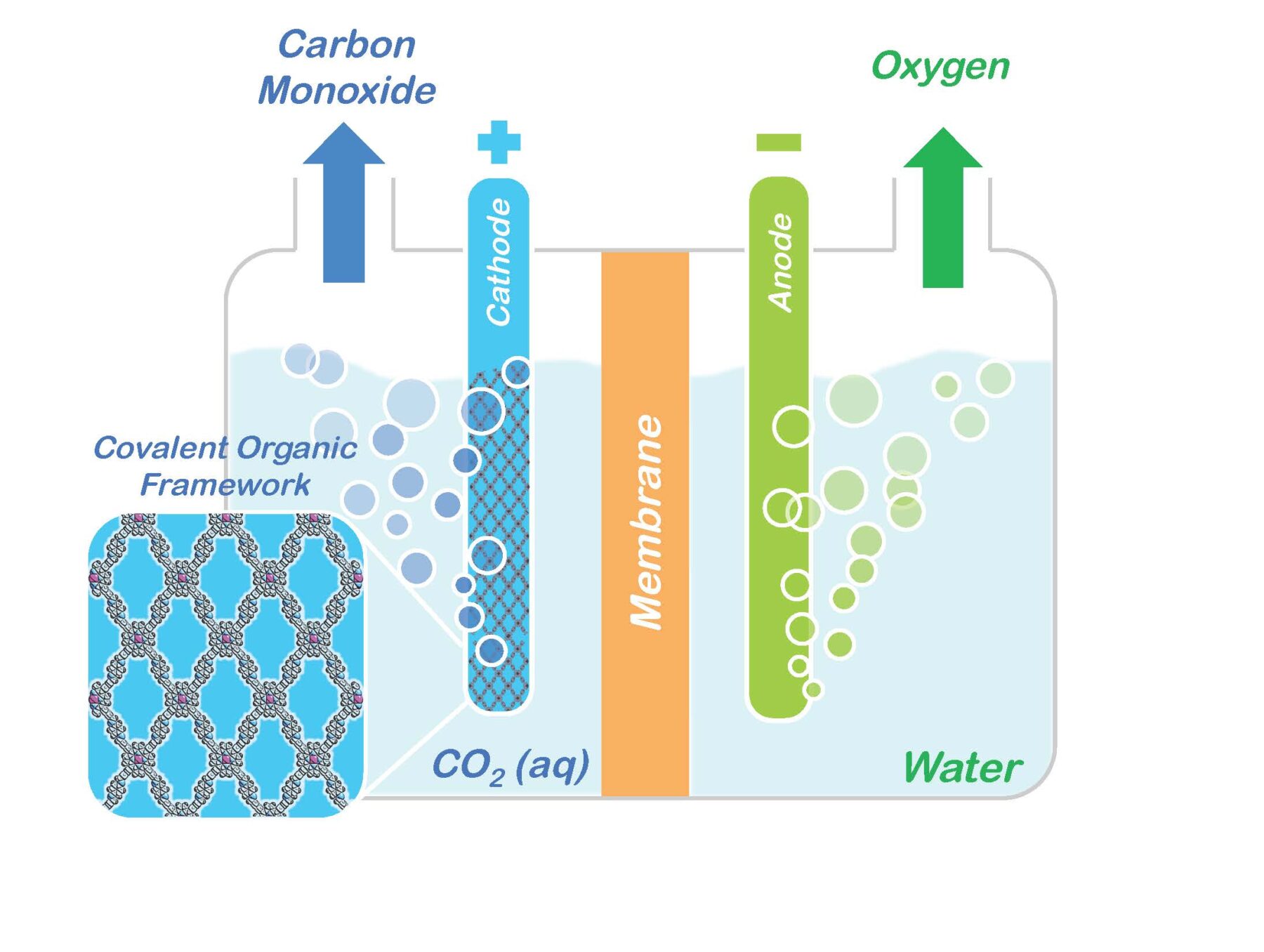
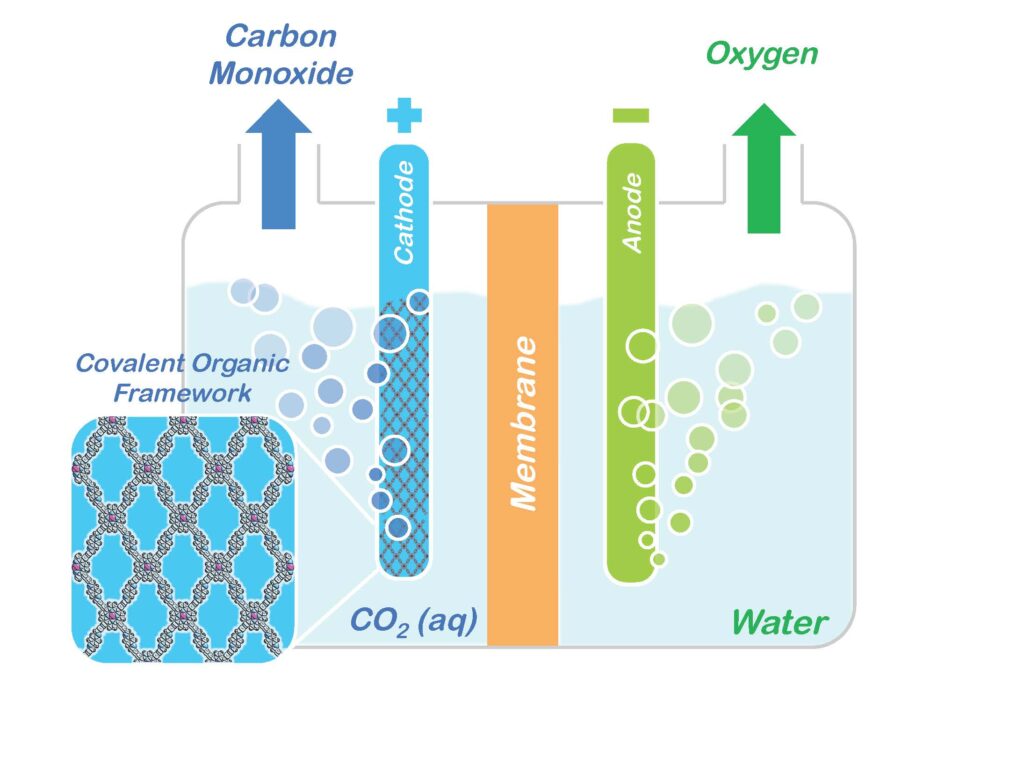
Researchers with the U.S. Department of Energy (DOE)’s Lawrence Berkeley National Laboratory (Berkeley Lab) have incorporated molecules of carbon dioxide reduction catalysts into the sponge-like crystals of covalent organic frameworks (COFs). This creates a molecular system that not only absorbs carbon dioxide, but also selectively reduces it to carbon monoxide, which serves as a primary building block for a wide range of chemical products including fuels, pharmaceuticals and plastics.
“There have been many attempts to develop homogeneous or heterogeneous catalysts for carbon dioxide, but the beauty of using COFs is that we can mix-and-match the best of both worlds, meaning we have molecular control by choice of catalysts plus the robust crystalline nature of the COF,” says Christopher Chang, a chemist with Berkeley Lab’s Chemical Sciences Division, and a co-leader of this study. “To date, such porous materials have mainly been used for carbon capture and separation, but in showing they can also be used for carbon dioxide catalysis, our results open up a huge range of potential applications in catalysis and energy.”
Chang and Omar Yaghi, a chemist with Berkeley Lab’s Materials Sciences Division who invented COFs, are the corresponding authors of a paper in Science that describes this research in detail. The paper is titled “Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water.” Lead authors are Song Lin, Christian Diercks and Yue-Biao Zhang. Other co-authors are Nikolay Kornienko, Eva Nichols, Yingbo Zhao, Aubrey Paris, Dohyung Kim and Peidong Yang.
Chang and Yaghi both hold appointments with the University of California (UC) Berkeley. Chang is also a Howard Hughes Medical Institute (HHMI) investigator. Yaghi is co-director of the Kavli Energy NanoScience Institute (Kavli-ENSI) at UC Berkeley.
The notoriety of carbon dioxide for its impact on the atmosphere and global climate change has overshadowed its value as an abundant, renewable, nontoxic and nonflammable source of carbon for the manufacturing of widely used chemical products. With the reduction of atmospheric carbon dioxide emissions in mind, Yaghi and his research group at the University of Michigan in 2005 designed and developed the first COFs as a means of separating carbon dioxide from flue gases. A COF is a porous three-dimensional crystal consisting of a tightly folded, compact framework that features an extraordinarily large internal surface area – a COF the size of a sugar cube were it to be opened and unfolded would blanket a football field.
The sponge-like quality of a COF’s vast internal surface area enables the system to absorb and store enormous quantities of targeted molecules, such as carbon dioxide.
Read more: Soaking Up Carbon Dioxide and Turning it into Valuable Products
The Latest on: Covalent organic frameworks
[google_news title=”” keyword=”covalent organic frameworks” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Covalent organic frameworks
- Ultrathin samples with surface phonon polariton enhance photoinduced dipole forceon May 6, 2024 at 12:53 pm
While analyzing the infrared photoinduced force response of quartz, Dr. Jian Li observed a unique spectral response that is different from the far field infrared absorption spectrum.
- When does a conductor not conduct? Switching a 2D metal-organic framework from an insulator to a metalon April 29, 2024 at 7:40 am
An Australian-led study has found unusual insulating behavior in a new atomically-thin material—and the ability to switch it on and off.
- Metal–organic frameworks articles from across Nature Portfolioon April 25, 2024 at 5:00 pm
Using liquid gallium as an atomically smooth substrate enables the deposition of single-crystal layers of conducting two-dimensional metal–organic frameworks. Two-dimensional covalent organic ...
- Recent advances and perspectives of covalent organic frameworks for alkali-ion batterieson March 21, 2024 at 7:01 am
Xu said that covalent organic framework (COF) materials exhibit several advantageous characteristics and demonstrate excellent electrochemical performance. Their unique network framework provides ...
- Study shows 3D-covalent organic frameworks can be tuned using structural isomerson March 6, 2024 at 7:21 am
More information: Xiaohan Wang et al, Triple Isomerism in 3D Covalent Organic Frameworks, Journal of the American Chemical Society (2024). DOI: 10.1021/jacs.3c13863 Provided by Tokyo Institute of ...
- What is a MOF (metal organic framework)?on January 21, 2024 at 11:49 pm
Metal-organic frameworks (MOFs) are organic-inorganic hybrid crystalline porous materials that consist of a regular array of positively charged metal ions surrounded by organic 'linker' molecules. The ...
- Organic Chemistryon January 18, 2024 at 11:10 am
Organic chemistry is the study of the structure, properties, composition, reactions, and preparation of carbon-containing compounds. Most organic compounds contain carbon and hydrogen, but they may ...
- A Bond Worth Forming: The Rise of Targeted Covalent Inhibitorson September 22, 2023 at 12:07 pm
Prior to 2000, many drugs that formed a covalent bond with targets did so non-selectively, which often led to undesired side effects when compared to non-covalent drugs. This led to the belief that ...
- What is covalent bonding?on March 14, 2023 at 8:31 pm
Each atom then has a complete outer shell of electrons. The slideshow shows how a covalent bond forms between a hydrogen atom and a chlorine atom, making hydrogen chloride. A simple molecule ...
- Ashlee J. Howarth, PhDon December 12, 2022 at 1:21 pm
covalent–organic frameworks (COFs), and metal–organic frameworks (MOFs). Recent literature will be used to highlight potential applications and key advances relevant to each class of porous materials.
via Bing News