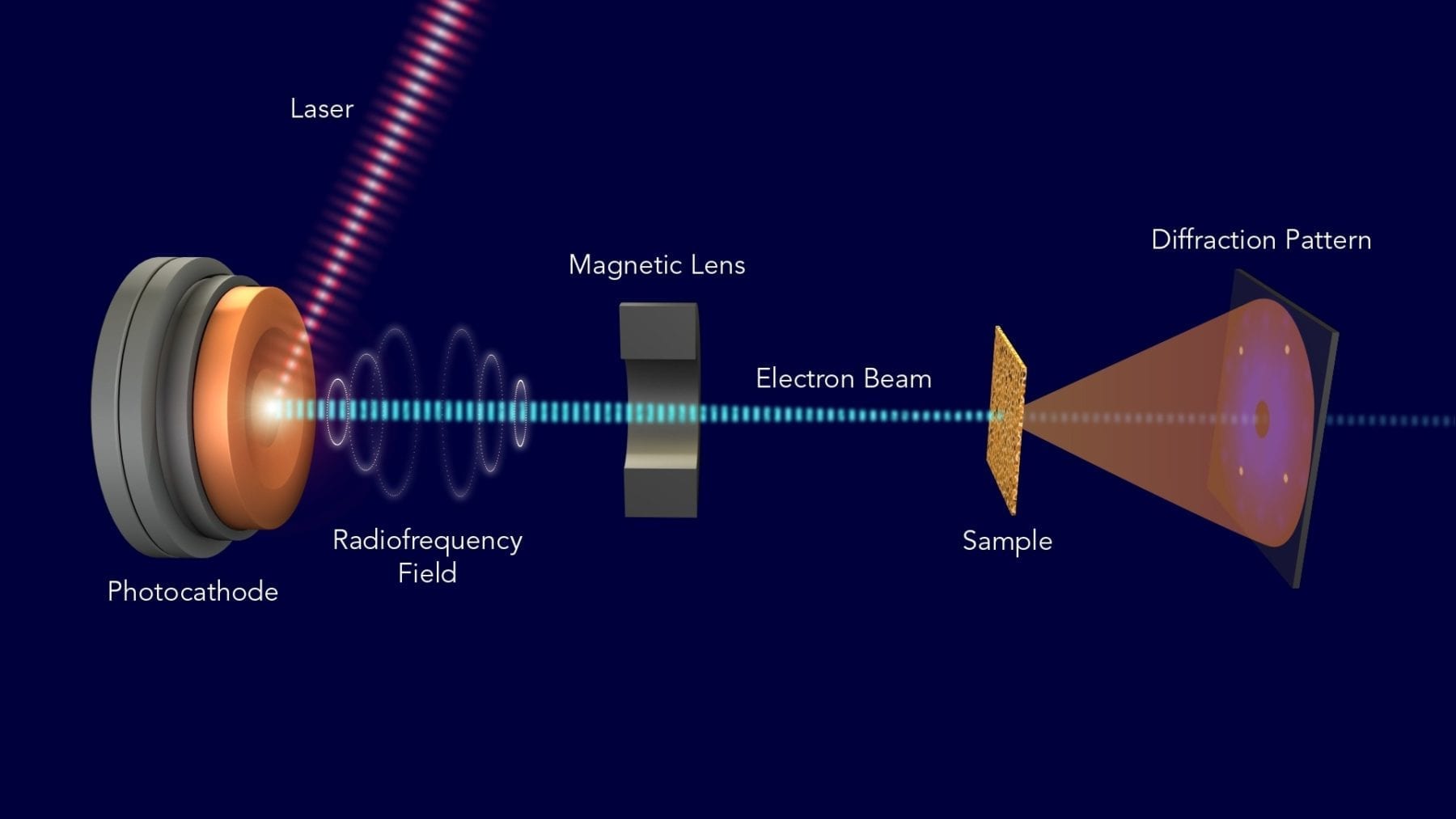
The term “designer drug” may soon refer less to the illicit kind and more to custom creations by the pharmaceutical industry. Aprecia Pharmaceuticals Company has just had its proprietary ZipDose Technology platform approved by the US Food and Drug Administration (FDA). This marks the very first instance that the FDA has given the green light for a 3D-printed drug product.
The use of 3D printing in the medical field is somewhat new, but growing rapidly. Bio-printing has proven to be an efficient way to create living tissues, and 3D-printed implants are able to deliver drugs more effectively. Now, with the FDA’s approval, Aprecia can continue the creation of its 3D-printed drug, possibly paving the way for future drugs.
Spritam levetiracetam is an oral prescription for the treatment of seizures and those affected by epilepsy. Aprecia’s ZipDose Technology enables the delivery of this drug, up to 1,000 mg per dose, with just a sip of water. This rapid disintegration method is achieved by 3D printing Spritam with a porous formulation. As a result, each dose is easy to take and does not require any measuring.
Read more: Approved by the FDA, 3D-printed drug set to change future of medication
The Latest on: 3D-printed drug
[google_news title=”” keyword=”3D-printed drug” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: 3D-printed drug
- Bird flu virus traces found in 1 in 5 samples of pasteurized milk: FDAon April 26, 2024 at 11:10 am
The Food and Drug Administration has found that about 1 in 5 samples of retail milk contain traces of highly contagious bird flu, though these findings may not be indicative of an infectious risk ...
- Enjoy STEM Family Day with the Einstein Projecton April 26, 2024 at 8:29 am
Perform robotics, 3D printing, science experiments, and more with STEM Family Day. UWGB STEM student organizations, Brown County Extension, and the Einstein Project are hosting a ...
- Technion breakthrough for better drug delivery and tissue implantationon April 23, 2024 at 2:11 pm
Researchers develop ultrasound for non-invasive method for bio-printing live cells and tissues deep within the body.
- Global 3D Printed Drugs Industryon April 23, 2024 at 7:40 am
Global 3D Printed Drugs Industry is expected to reach US$ 742.88 Million in 2032, growing at a CAGR of 9.2% From 2022 to 2032 ...
- 3D-printed ‘ghost guns’ keep showing up at N.J. crime scenes, state probe findson April 17, 2024 at 4:18 pm
The State Commission of Investigation, an independent New Jersey government watchdog, held a hearing in Trenton on Tuesday as part of its probe into the proliferation of “ghost guns” and 3D-printable ...
- Man accused of 3D printing universal mailbox keys in Harris County facing several fraud-related chargeson April 17, 2024 at 11:55 am
A Texas man accused of making 3D printed copies of counterfeit universal mail keys, known as arrow keys, has been indicted by a Harris County grand jury.
- FDA Approves Innovative 3D-Printed Cranial Implants for US Market Entryon April 16, 2024 at 11:10 pm
VSP® PEEK Cranial Implant is designed to address defects in the skull resulting from traumatic injuries like fractures or gunshot wounds.
- In a first, FDA approves 3D-printed cranial implants to enter USon April 16, 2024 at 9:23 am
Now, the Food and Drug Administration (FDA) has cleared a customized 3D-printed solution to precisely fit a patient’s skull. The clearance by the FDA has paved the way for the w ...
- 3D Systems obtains 510(k) clearance for 3D-printed cranial implantson April 16, 2024 at 4:19 am
3D Systems has received 510 (k) clearance from the US Food and Drug Administration (FDA) for its VSP PEEK Cranial Implant. The patient-specific implant includes the EXT 220 MED 3D printer and ...
- 3D Systems Announces FDA Clearance for World’s First 3D-Printed PEEK Cranial Implantson April 15, 2024 at 7:30 am
FDA clearance enables wide-spread adoption of 3D Systems self-contained, cleanroom environment-based printing system, the EXT 220 MED, with ...
via Bing News