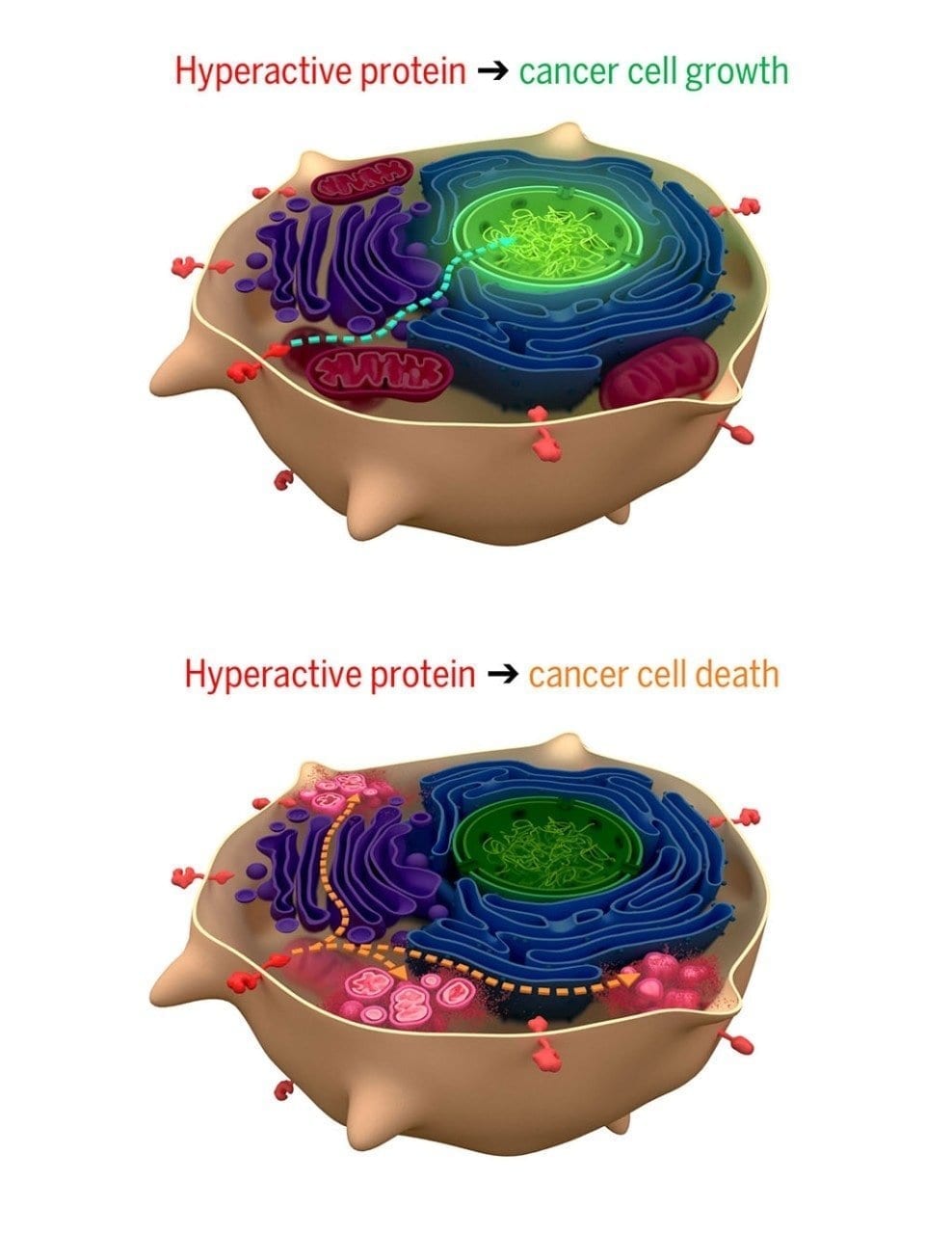
An investigational cancer drug that targets tumors caused by mutations in the KRAS gene will be evaluated in phase 2 clinical trials, following promising safety and efficacy results in preliminary human studies and excellent results in animal studies.
The drug, developed by Amgen and currently referred to as AMG 510, is the first therapy to reach clinical trials that inhibits a mutant KRAS protein. Errors in the KRAS gene, which encodes a crucial cell signaling protein, are one of the most common causes of cancer.
The particular mutant inhibited by AMG 510 – called KRAS(G12C) – is present in approximately 13% of lung adenocarcinomas, 3% of colorectal cancers, and 2% of other solid tumors.
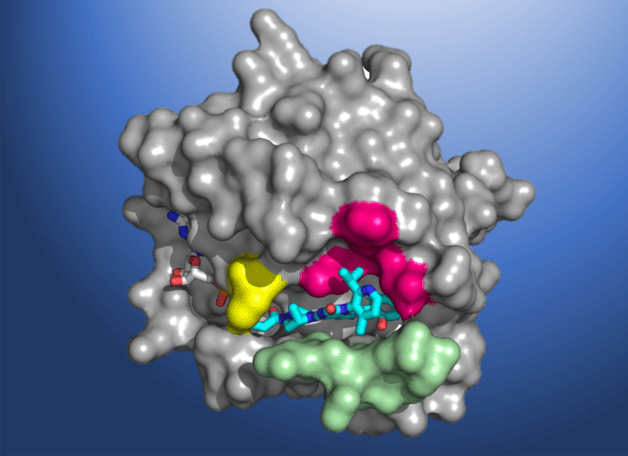
Despite its significant role in the pathogenesis of cancer, scientists have been unable to design KRAS-specific therapeutics due to the shape of the protein – it has an exceptionally smooth surface with no obvious regions for a drug molecule to bind. Seeking to develop a long-sought direct inhibitor, researchers at Amgen conducted X-ray crystallography of KRAS(G12C) proteins at Berkeley Lab’s Advanced Light Source (ALS). The high-resolution structural maps generated using the data acquired at the beamlines helped Amgen make the breakthrough discovery of a small pocket on the molecule. In subsequent studies, the beamline data allowed scientists to investigate atomic-level molecular interactions between KRAS(G12C) and potential inhibiting compounds that bind in this pocket. AMG 510 emerged as a very promising candidate after a multiyear drug agent optimization program.
“It’s rare that a compound gets all the way through the development process and becomes a drug. So, for the BCSB team, it feels great to see our (small) contribution finding its way to fight diseases,” said Marc Allaire, one of the Berkeley Lab biophysicists who operate the BCSB beamlines.
Learn more: Unique Cancer Drug Discovered With Help From Advanced Light Source Begins Historical Clinical Trial
Go deeper with Bing News on:
Investigational cancer drug
- Replay wins IND clearance for PRAME T cell cancer therapy
Genome writing company Replay Bio has had its investigational new drug (IND) application cleared by the US Food ... Syena is working alongside the University of Texas MD Anderson Cancer Center to fund ...
- FDA Drug Approval Decisions Expected in June 2024
Tarlatamab is an investigational first-in-class, bispecific T-cell engager immunotherapy designed to target delta-like ligand 3 on small cell lung cancer (SCLC) cells and CD3 on T cells. The ...
- Trials to watch: Four ALS drugs to keep an eye on
There are a total of 12 candidates in mid to late-stage trials for ALS poised to enter the market in the next five years.
- FDA approves immunotherapy drug combo for non-muscle invasive bladder cancer
The U.S. Food and Drug Administration (FDA) has approved the immunotherapy-boosting drug N-803, which is marketed under the brand name Anktiva, to be used in combination with the ...
- Survival gains seen with assignment to experimental group in cancer trials
For patients with solid tumors, assignment to an experimental group in trials of investigational drugs yields significant survival gains, according to a review published online April 30 in the Annuals ...
Go deeper with Google Headlines on:
Investigational cancer drug
[google_news title=”” keyword=”investigational cancer drug” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Cancer drug
- Johnson & Johnson proposes to pay $6.5B to settle talc ovarian cancer lawsuits in US
Through a bankruptcy filing of its subsidiary company, J&J will be able to resolve about 99.75% of the pending talc lawsuits against the company and its affiliates in the U.S.
- Two-punch treatment delivers blood cancer knockout: Study shows drug combo eradicates cancer cells in lab-based tests
A novel combination of two cancer drugs has shown great potential as a future treatment for patients with acute myeloid leukemia (AML), one of the most common types of blood cancers. A new study by WEHI researchers has revealed the combination of two ...
Go deeper with Google Headlines on:
Cancer drug
[google_news title=”” keyword=”cancer drug” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]