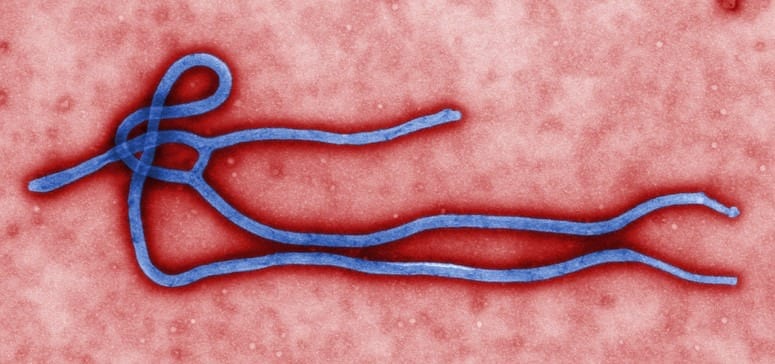
The worst Ebola virus disease outbreak in history ended in 2016 after infecting 28,600 people and killing about 11,300 worldwide.
The outbreak led to urgent action by medical experts across the world to combat this devastating disease; including the setting up of trials of vaccines to stop the disease taking hold.
This global commitment to develop a vaccine against the disease suggested eight options, out of a starting pool of 15 candidates, should be evaluated in clinical trials worldwide by the end of 2015.
Professor Sanjeev Krishna, of St George’s University of London’s Institute for Infection and Immunity, said: “An unprecedented Ebola outbreak showed how it is possible for academics, non-governmental organisations, industry and funders to work effectively together very quickly in times of medical crisis. The results of the trial show how a vaccine could best be used to tackle this terrible disease effectively.
“We need a system of specialists, medical experts and organisers that maintains vigilance against outbreak diseases like Ebola.
“We should continue to improve ways to make, evaluate and deliver vaccines when they are needed, often in parts of the world lacking in infrastructure for diagnosing infections and providing treatments.”
He explained that considering the persistent replication of the vaccine which is called rVSV-?GP-ZEBOV in children and adolescents, further studies investigating lower doses in this population are warranted.
The vaccine contains a non-infectious portion of a gene from the Zaire Ebola virus. The St George’s researchers worked with colleagues on a vaccine trial in Gabon.
In addition, lower vaccine doses should be considered when boosting individuals with pre-existing antibodies to Ebolavirus glycoprotein, a finding that has emerged after the vaccine was tested in a country that has experienced Ebolavirus outbreaks in the past.
The vaccine was one of two being examined as a ‘candidate’ option by the World Health Organisation to identify urgently a vaccine to combat the Ebola virus outbreak in West Africa.
The clinical trial was led by colleagues at University of Tübingen in Germany, coordinated by Professor Peter Kremsner with their partner institute CERMEL in Lambaréné, Gabon.
Learn more: Ebola vaccine tested in adults and children in Africa hailed a success
The Latest on: Ebola vaccine
[google_news title=”” keyword=”Ebola vaccine” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- Ebola's exponential growthon May 8, 2024 at 5:00 pm
Until and unless a vaccine becomes available in very large quantities, the only way to stop the exponential spread of Ebola in the affected countries is to isolate the victims, a task that is very ...
- Urgent considerations for booster vaccination strategies against Ebola virus diseaseon May 8, 2024 at 4:30 pm
With two endorsed and prophylactic vaccines against Zaire ebolavirus (referred to hereafter as EBOV), the number of individuals vaccinated against EBOV worldwide is estimated to range between 500 000 ...
- AstraZeneca Covid vaccine to be withdrawn after admitting rare side effecton May 8, 2024 at 6:23 am
AstraZeneca is set to pull its Covid vaccine after acknowledging it could cause a rare and potentially fatal side effect. The pharmaceutical heavyweight voluntarily withdrew its "market authorization" ...
- Newsday - Fears over the use of a new Ebola vaccine in DRCongo - BBC Soundson May 8, 2024 at 1:41 am
An MP explains why there's scepticism about the goal of vaccinations ...
- Vaccine Developers Leverage mRNA and Other Powerful Technologieson April 30, 2024 at 5:00 pm
Broderick has worked for many years on vaccines for Ebola, Zika, Lassa fever, and Middle East respiratory syndrome. “For an infectious disease vaccine, what you really want to see is what we call ...
- Conference Report - I. Investigating New Vaccines: Ebola and HIVon April 26, 2024 at 5:00 pm
In our efforts to protect ourselves from diseases caused by viruses, we are constantly faced by the challenges posed by a nature that is steadily changing and adapting. These challenges can take ...
- Soligenix Extends Patent Protection for its Filovirus Vaccine Platform to the United Kingdom and South Africaon April 25, 2024 at 4:30 am
Includes thermostabilized Ebola vaccines MarVax™ and SuVax™ Includes nanoemulsion adjuvant compatible with lyophilization "Our next generation combined vaccine platform includes three major ...
- Soligenix’s vaccine for Sudan ebolavirus granted orphan drug designation in USon April 11, 2024 at 5:00 pm
The FDA has granted orphan drug designation to the active ingredient in Soligenix Inc.’s Suvax, a subunit protein vaccine of recombinantly expressed Sudan ebolavirus (SUDV) glycoprotein, for the ...
- Conference Report - I. Investigating New Vaccines: Ebola and HIVon April 10, 2024 at 5:00 pm
The hunt for an efficacious vaccine against HIV is still eluding scientists, despite the huge amount of knowledge accumulated about this virus and its role in the pathogenesis of AIDS. Candidate ...
- Sudan Ebola Virus Vaccine Candidate Gets Orphan Drug Statuson April 10, 2024 at 5:00 pm
The vaccine candidate has been shown to be stable for at least 2 years when stored at temperatures of 40 degrees Celsius (104 degrees Fahrenheit). The Food and Drug Administration (FDA ...
via Google News and Bing News