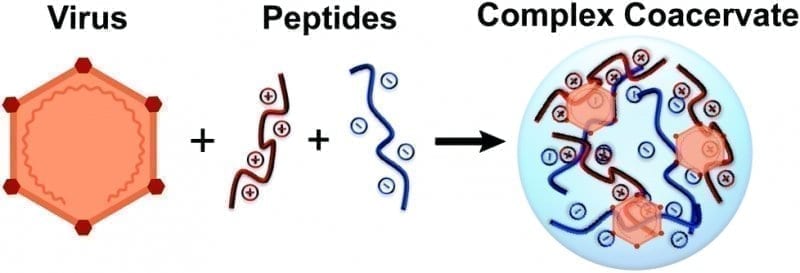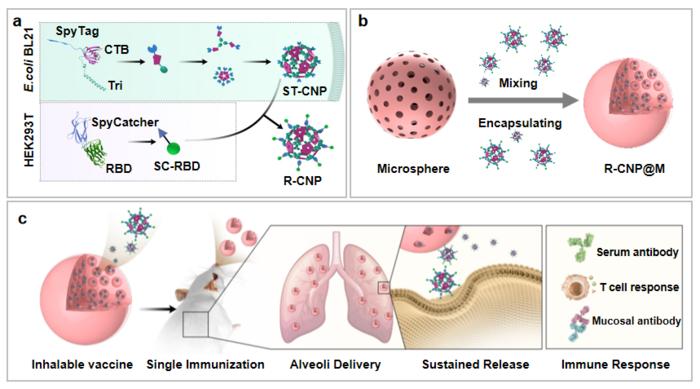
Construction and mechanism of single-dose, dry-powder inhalation vaccine
CREDIT: Image by MA Guanghui’s group
Researchers develop a novel dry-powder inhaled vaccine platform
Researchers from the Institute of Process Engineering (IPE) of the Chinese Academy of Sciences have proposed a new “nano-micro composite” delivery concept for vaccines. Based on this idea, they have developed a single-dose, dry-powder, inhalable vaccine platform using nano-micro composite multilevel structures, which has been successfully prepared in the laboratory, and the vaccine has been shown to be effective in blocking respiratory viral infection and transmission in animal models. This platform holds great promise for combating future emerging and epidemic infectious diseases.
This study was published in Nature on Dec 13.
In recent years, researchers have made significant progress in developing vaccines for infectious respiratory diseases. However, most of these vaccines are administered through intramuscular injection, which primarily induces a humoral immune response and relies on blood antibodies to neutralize the virus. Unfortunately, this approach fails to trigger a mucosal immune response and establish a robust immune barrier in the respiratory tract.
Moreover, adjuvants commonly used in current vaccines, e.g., aluminum adjuvants, are unable to induce cellular immune responses and are ineffective in combating rapid viral mutations. Additionally, the current liquid form of vaccines necessitates strict low-temperature storage conditions, and the two- or three-dose vaccination schedule also impacts the overall vaccination rate.
To address these problems, interdisciplinary integration and innovative research concepts are needed to develop safer and more efficient vaccines for respiratory infections.
Taking this approach, Prof. WEI Wei and Prof. MA Guanghui, from the State Key Laboratory of Biochemical Engineering at IPE, in cross-disciplinary collaboration with Prof. WANG Hengliang and Prof. ZHU Li, from the State Key Laboratory of Pathogen and Biosecurity, have developed a new vaccine platform to tackle these challenges.
This platform combines biodegradable microspheres with protein nanoparticles. The surface of these nanoparticles can simultaneously display multiple antigens, thus inducing a broad-spectrum immune response and expanding the range of vaccine protection. It also allows for the quick and convenient development of other respiratory virus vaccines due to the flexibility of antigen presentation.
Moreover, the platform’s unique nano-micro composite structure enables an efficient immune response in the lungs by facilitating high-performance delivery. Once the antigen-nanoparticles are released, they can be efficiently taken up by antigen-presenting cells.
Additionally, this dry powder vaccine significantly reduces storage and transportation costs, making it suitable for areas with limited refrigeration facilities, thus improving immunization rates.
Furthermore, the sustained release of antigens in the vaccine induces long-lasting humoral, cellular, and mucosal immunity with just a single inhalation. In later cooperation with Prof. HE Zhanlong of the Institute of Medical Biology of the Chinese Academy of Medical Sciences, the researchers developed a model of airborne protection, close contact protection, and airborne transmission blocking, which described the benefits of inhaled vaccine for preventing the virus infection and transmission.
“The components of this nano-micro system used natural proteins and approved polymer materials, and the effectiveness and safety of the vaccine have been systematically studied in non-human primates, indicating its great potential for clinical translation.” said Prof. WEI Wei.
Original Article: Researchers develop a novel dry-powder inhaled vaccine platform
More from: Chinese Academy of Sciences
The Latest Updates from Bing News
Go deeper with Bing News on:
Dry-powder inhaled vaccine
- TFF Pharmaceuticals Announces Pricing of $4.8 Million Public Offering
The gross proceeds from the offering, before deducting the placement agent's fees and other offering expenses, are expected to be approximately $4.8 million. The Company intends to use the net ...
- Insmed gets grant for treatment of gram-positive pulmonary bacterial infections
Discover Insmed Inc's groundbreaking patent for lipo-glycopeptide derivatives to combat bacterial infections in the lungs, including cystic fibrosis patients. Learn about the innovative method using ...
Go deeper with Bing News on:
Inhalable vaccine platform
- Gritstone bio Highlights the Durability and Potential Broad Utility of its Self-amplifying mRNA (samRNA) COVID-19 Vaccine at ESCMID Global 2024
IgG levels and neutralizing antibodies against variants of concern persisted for at least 12 months, consistent with previous findings ---- ...
- GSK Says Pfizer Infringed Five Patents Relating to Comirnaty COVID Vaccine
GlaxoSmithKline filed a four-count civil action for patent infringement in the United States District Court for the District of Delaware late last week seeking damages for Pfizer and BioNTech’s ...
- Vaccines News
It utilizes a nanoliposome vaccine platform that underwent phase 2 and phase 3 clinical trials in South Korea and the ... How Children's Birthdays Help Show the Best Month for Flu Shots Feb. 22 ...
- China approves world’s first inhalable Covid-19 vaccine
Chinese drug regulators have approved the world’s first inhalable Covid-19 vaccine, made by Tianjin-based manufacturer Cansino Biologics. The National Medical Products Administration gave the go ...
- Applied DNA Awarded Contract by HDT Bio For Rapid Vaccine Development Program
Under the terms of the contract, Applied DNA will supply Linea™ DNA IVT templates to HDT Bio for use in conjunction with its LION™ formulated repRNA (self-replicating RNA) vaccination platform. The ...