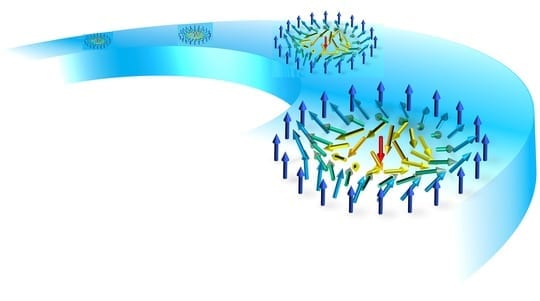
A new kind of antibody targets a feature shared by proteins thought to cause the most damage in Alzheimer’s disease, Parkinson’s disease, and related conditions, creating potential for a unified treatment approach.
This is the finding of a study led by researchers from NYU School of Medicine and published online August 29 in Scientific Reports.
The new study is based on decades of work arguing that the contribution to disease of key proteins—amyloid beta and tau in Alzheimer’s disease, alpha-synuclein in Parkinson’s disease, and prion proteins in conditions like mad cow disease—is driven by certain, toxic forms dominated by a common structure: bundles of “beta sheets” in clumped proteins.
In tissues from autopsied patients with these diseases and in live mice, experiments demonstrated how the study antibodies target and remove only these toxic forms, say the authors, and without triggering the immune toxicity that has frustrated treatment development efforts to date.
“In an atmosphere where countless treatments have failed in clinical trials over the last 15 years, the fact that our approach continues to be effective in rigorous tests should be of interest to our peers and the industry, even if it runs contrary to conventional thinking,” says corresponding author Fernando Goni, PhD, research associate professor in the Department of Neurology at NYU Langone Health.
“While we still have a number of milestones to reach before this work is ready for clinical testing, our results suggest that these antibodies may halt key pathological mechanisms across several neurological diseases and regardless of disease stage,” says corresponding author Thomas Wisniewski, MD, director of NYU Langone’s Center for Cognitive Neurology, Silberstein Alzheimer’s Institute, and Alzheimer’s Disease Center.
New Approach to Antibody Design
The study focuses on proteins that form important structures in the brain. The instant they form as chains of amino acids, proteins fold into complex shapes needed to do their jobs. Unfortunately, proteins can also “misfold” for countless reasons, such as genetic abnormalities, toxins, age-associated cell processes, and inflammation that eventually cause the diseases addressed by the current study. Cells and tissues die as misshapen proteins stop working and build up, but the field has struggled to pinpoint which of these shifting forms to target as the key drivers of disease.
Many research efforts, including the current study, seek to design antibodies shaped to attach to and remove the right targets. Past and ongoing attempts have targeted the initial, short chains of amino acids that serve as basic, repeating structural units, or monomers, of each misfolded protein. Still others targeted end-stage fibrils, each made of thousands of monomers, which accumulate in plaques and tangles that tissues cannot eliminate. Neither approach has yielded an effective therapy.
In that light, Dr. Goni, Dr. Wisniewski, and colleagues designed their antibodies to target instead the “oligomers” formed as several misfolded monomers associate and acquire the “beta-sheet” shape, but before they are large enough to fibrilize. These intermediate forms may be uniquely toxic, say many in the field, because, unlike fibrils, they can dissolve, move in and out of cells, and from one cell to another. This mobility may explain the “prion-like” progression seen in misfolding diseases where abnormal proteins cause normal ones to misfold in a domino effect that damages nerve cells and their connections in the brain.
Importantly, growing toxic oligomers of amyloid beta, tau, alpha synuclein, and prion protein become increasingly dominated by the twisted strands of amino acids called beta sheets, which have spatial arrangements that let the strands stack up.
To design new kinds of antibodies, the research team zeroed in on a small 13-amino acid peptide, derived from the extremely rare genetic disease called British amyloidosis, but not present in the rest of the human population. They converted it into a larger, stable oligomer with more than 90 percent “beta-sheet” structure—the p13Bri immunogen—that could be “seen” by the mammalian immune system, and that could trigger a specific antibody response to solve problems encountered with standard approaches. By immunizing mice with p13Bri at high doses, they forced the production of extremely rare antibodies against beta sheets in toxic oligomers.
The researchers say that their rare antibodies, activated by a protein fragment seen only in a rare disease, have almost zero chance of triggering unwanted immune responses to normal proteins with similar sequences (autotoxicity), the downfall of many previous attempts. Finally, the team screened their lead antibodies against tissues taken from the brains of human patients with Alzheimer’s disease, Parkinson’s disease, and prion diseases. Only the six monoclonal antibodies that reacted to toxic oligomers from at least two misfolded proteins from two diseases were selected for further study.
“This publication details the first system for making antibodies that truly target only toxic oligomers of misfolded proteins dominated by beta sheets across several diseases, and without regard to the amino acid makeup of each misfolded protein’s monomer,” says Dr. Goni.
Learn more: New Antibodies Target Structures Shared by Proteins Thought to Worsen Major Neurological Diseases
The Latest on: Neurological diseases
[google_news title=”” keyword=”neurological diseases” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- From understanding Parkinson’s Disease to the increased rate of cancer cases, here are this week’s talkbackson April 27, 2024 at 7:22 pm
WGAL News 8 talks to professionals to give a better understanding of important events impacting the Susquehanna Valley.
- These Popular Recreational Activities Could Be Increasing Your Risk of a Deadly Neurological Diseaseon April 27, 2024 at 2:45 pm
Activities could be modifiable risk factors for the disease. A study from Michigan Medicine suggests that participating in recreational activities — including golfing, gardening or yard work, ...
- What are prion diseases? Hunters died of fatal disorder after eating tainted deer meat, researchers sayon April 27, 2024 at 9:34 am
A recent report published in the medical journal Neurology suggests that two hunters contracted Creutzfeldt-Jakob disease, a fatal neurological disorder, after eating venison obtained from deer ...
- Soft robotic nerve cuffs could revolutionize treatment of neurological conditionson April 26, 2024 at 5:49 pm
Researchers have developed tiny, flexible devices that can wrap around individual nerve fibres without damaging them.
- How Texas MS 150 brings in funds to fight the disease better than everon April 26, 2024 at 4:05 pm
Multiple sclerosis (MS) can be a devastating diagnosis that affects about a million people in the United States. The good news is there’s been more progress in MS than in any other neurological ...
- CDC: Deer meat didn't cause hunters' deaths; concerns about chronic wasting disease remainon April 26, 2024 at 1:26 pm
There have been no cases of chronic wasting disease in people to date, the CDC says. But it is still investigating the potential risks.
- Five Things to Know about Functional Neurological Disorderon April 26, 2024 at 10:28 am
Functional neurological disorder describes a pattern of symptoms with no clear physical cause. FND is considered a subtype of conversion disorder and is believed to be an extreme reaction to stress.
- What It’s Like to Live With Neurologic Lyme Disease: A Patient Perspectiveon April 26, 2024 at 6:24 am
Diagnosing patients with neurologic Lyme disease can take upwards of 2 years, which Maria Arini Lopez discovered first-hand in the spring of 2021.
- Contaminated deer meat may have caused 2 hunters' deadly brain diseaseson April 25, 2024 at 11:28 am
A new report calls for further study into a possible link between chronic wasting disease in deer and a rare and deadly brain disease in humans.
- Chronic wasting disease feared in deaths of 2 hunters who ate deer meaton April 24, 2024 at 3:14 am
Chronic wasting disease has not yet been shown to be transmissible from animals to humans. However, the researchers are concerned about possible connections.
via Google News and Bing News