CREDIT
Masaaki Sadakiyo / International Institute for Carbon-Neutral Energy Research, Kyushu University
Research team Kyushu University creates electrolytic flow cell to produce alcohols from carboxylic acids
Interest in renewable energy continues to burgeon. Many renewables, though, can be frustratingly intermittent – when the sun stops shining, or the wind stops blowing, the power flickers. The fluctuating supply can be partly smoothed-out by energy storage during peak production times. However, storing electricity is not without its challenges either.
Recently, a team at the International Institute for Carbon-Neutral Energy Research (I2CNER), within southern Japan’s Kyushu University, created a device to store energy in chemical form through continuous electrolysis.
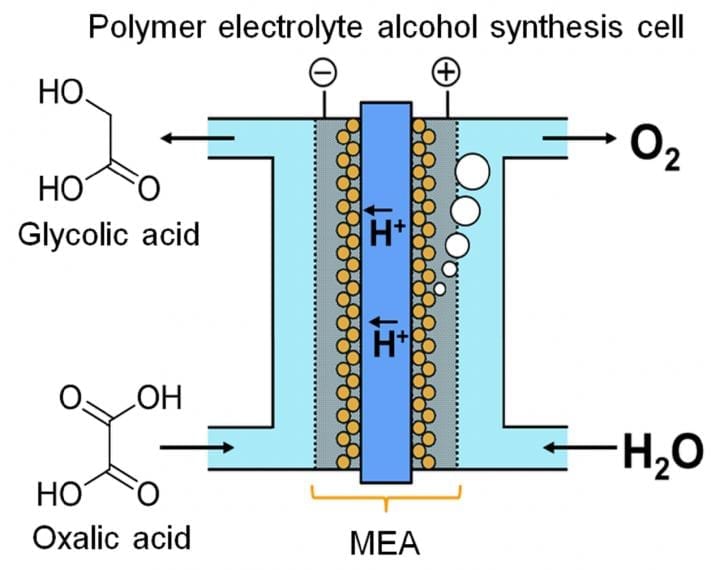
The researchers noted that glycolic acid (GC) has a much greater energy capacity than hydrogen, one of the more popular energy-storage chemicals. GC can be produced by four-electron reduction of oxalic acid (OX), a widely available carboxylic acid. As described in their publication in Scientific Reports, the team devised an electrolytic cell based on a novel membrane-electrode assembly. Sandwiched between two electrodes are an iridium oxide-based anode and a titanium dioxide (TiO2)-coated titanium (Ti) cathode, linked by a polymer membrane.
“Flow-type systems are very important for energy storage with liquid-phase reaction,” study lead author Masaaki Sadakiyo explains. “Most electrolyzers producing alcohols operate a batch process, which is not suitable for this purpose. In our device, by using a solid polymer electrolyte in direct contact with the electrodes, we can run the reaction as a continuous flow without addition of impurities (e.g. electrolytes). The OX solution can effectively be thought of as a flowable electron pool.”
Another key consideration is the cathode design. The cathodic reaction is catalyzed by anatase TiO2. To ensure a solid connection between catalyst and cathode, the team “grew” TiO2 directly on Ti in the form of a mesh or felt. Electron microscope images show the TiO2 as a wispy fuzz, clinging to the outside of the Ti rods like a coating of fresh snow. In fact, its job is to catalyze the electro-reduction of OX to GC. Meanwhile, at the anode, water is oxidized to oxygen.
The team found that the reaction accelerated at higher temperatures. However, turning the heat up too high encouraged an unwanted by-process – the conversion of water to hydrogen. The ideal balance between these two effects was at 60°C. At this temperature, the device could be further optimized by slowing the flow of reactants, while increasing the amount of surface area available for the reaction.
Interestingly, even the texture of the fuzzy TiO2 catalyst made a major difference. When TiO2 was prepared as a “felt,” by growing it on thinner and more densely packed Ti rods, the reaction occurred faster than on the “mesh” – probably because of the greater surface area. The felt also discouraged hydrogen production, by blanketing the Ti surface more snugly than the mesh, preventing the exposure of bare Ti.
“In the right conditions, our cell converts nearly 100% of OX, which we find very encouraging,” co-author Miho Yamauchi says. “We calculate that the maximum volumetric energy capacity of the GC solution is around 50 times that of hydrogen gas. To be clear, the energy efficiency, as opposed to capacity, still lags behind other technologies. However, this is a promising first step to a new method for storing excess current.”
Learn more: Exploring electrolysis for energy storage
The Latest on: Flow-type polymer electrolyte cell
[google_news title=”” keyword=”flow-type polymer electrolyte cell” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- Pathogenesis of Type 2 Diabeteson April 26, 2024 at 5:00 pm
The pathogenesis of type 2 diabetes ordinarily involves the development of insulin resistance associated with compensatory hyperinsulinemia, followed by progressive beta-cell impairment that ...
- Korean researchers build 8 kW solid oxide electrolysis cell that can produce 5.7 kg of hydrogen per dayon April 26, 2024 at 3:23 am
The Korea Institute of Energy Research has developed a solid oxide electrolysis cell stack that uses a special kind of separator plate to ensure proper flow of hydrogen and oxigen after water ...
- Batteries Newson April 24, 2024 at 5:00 pm
Revolutionary Method Developed for Mass-Producing Polymer Solid Electrolytes ... The team use water to replace organic electrolytes -- which enable the flow of ... Road to Better Performing ...
- Discovery of new vascular cell type may pave way for novel strategies to treat cardiovascular diseaseson April 18, 2024 at 12:46 pm
Cardiovascular diseases, including stroke and myocardial infarction, are the world's leading causes of mortality, accounting for over 18 million deaths a year.
- Outlook and Life Expectancy for Non-Small Cell Lung Carcinomaon April 9, 2024 at 5:00 pm
Non-small cell lung carcinoma (NSCLC), also called non-small cell lung cancer, is the most common type of lung cancer. “Non-small cell” refers to the way cancer cells look under a microscope.
- Quantitative Cell Profiling of Immune Cell Types From Whole Blood Sampleson April 7, 2024 at 5:00 pm
This application note explores an optimized RNA-sequencing workflow that reliably determines cell subset abundance and cell-type-specific gene expression profiles from bulk tissue or blood expression ...
- Accurate cell type annotation for single-cell chromatin accessibility data via contrastive learning and reference guidanceon April 7, 2024 at 5:00 pm
the importance of cell type annotation in scCAS data is on the rise due to its potential to capture the chromatin regulatory landscape that controls gene transcription in each cell type.
- New electrolyte for safer, more stable solid-state batterieson April 4, 2024 at 5:00 pm
Now, a group of researchers led by the Tokyo University of Science have discovered a stable, highly conductive electroyte material in the form of pyrochlore-type oxyfluoride. This innovation ...
- Upper urinary tract urothelial canceron March 28, 2024 at 10:05 am
Upper urinary tract urothelial cancer is a rare type ... cells line the renal pelvis, ureters, bladder and urethra. They can change shape and stretch. This means these parts of the body can expand to ...
- GPT-4 for identifying cell types in single cells matches and sometimes outperforms expert methodson March 24, 2024 at 5:00 pm
March 25, 2024-- GPT-4 can accurately interpret types of cells important for the analysis of single-cell RNA sequencing -- a sequencing process fundamental to interpreting cell types – with high ...
via Google News and Bing News









