via Phys.org
Few people who have undergone nasopharyngeal swabs for coronavirus testing would describe it as a pleasant experience. The procedure involves sticking a long swab up the nose to collect a sample from the back of the nose and throat, which is then analyzed for SARS-CoV-2 RNA by the reverse-transcription polymerase chain reaction (RT-PCR). Now, researchers reporting in ACS Nano have developed a prototype device that non-invasively detected COVID-19 in the exhaled breath of infected patients.
In addition to being uncomfortable, the current gold standard for COVID-19 testing requires RT-PCR, a time-consuming laboratory procedure. Because of backlogs, obtaining a result can take several days. To reduce transmission and mortality rates, healthcare systems need quick, inexpensive and easy-to-use tests. Hossam Haick, Hu Liu, Yueyin Pan and colleagues wanted to develop a nanomaterial-based sensor that could detect COVID-19 in exhaled breath, similar to a breathalyzer test for alcohol intoxication. Previous studies have shown that viruses and the cells they infect emit volatile organic compounds (VOCs) that can be exhaled in the breath.
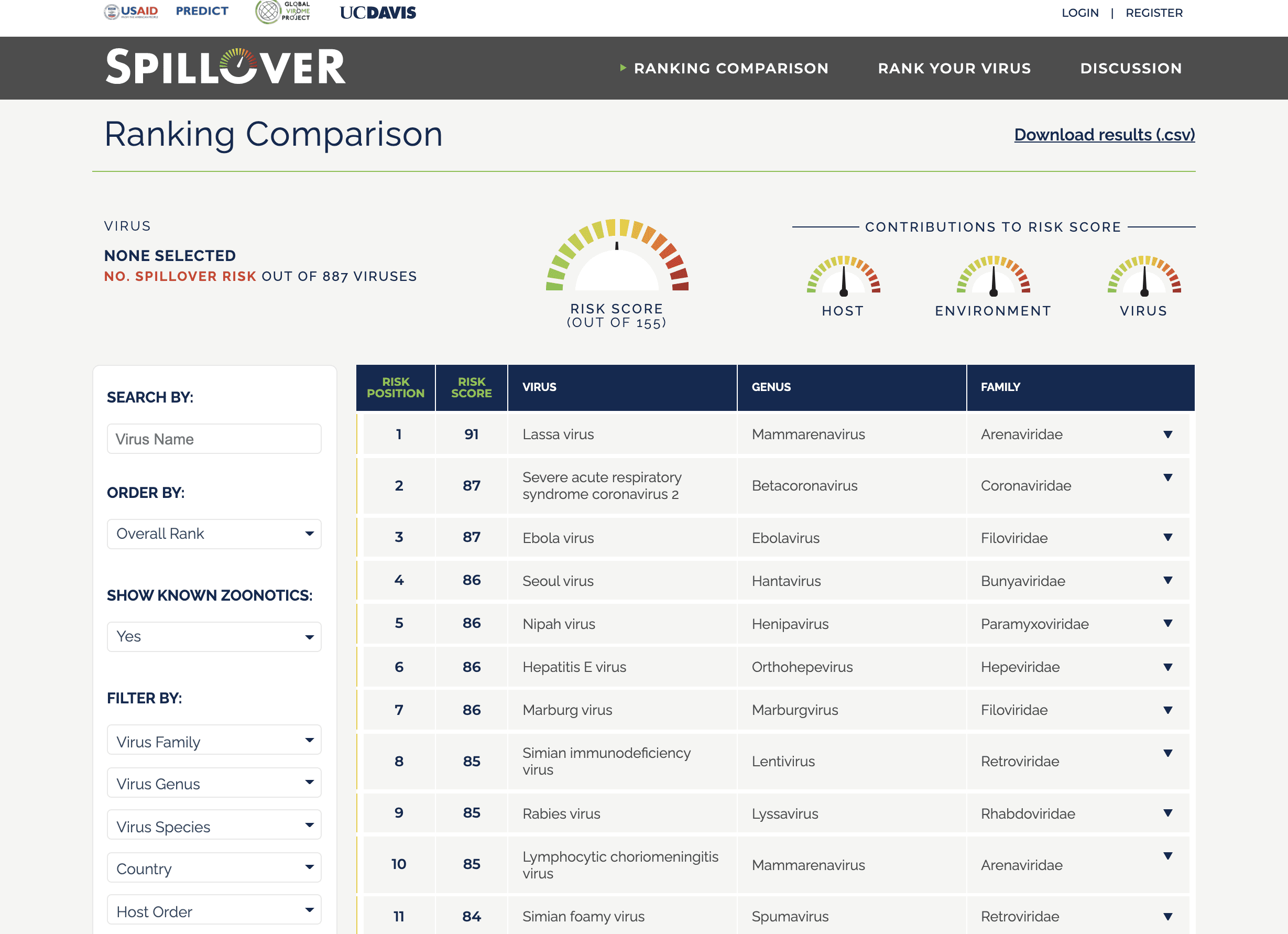
The researchers made an array of gold nanoparticles linked to molecules that are sensitive to various VOCs. When VOCs interact with the molecules on a nanoparticle, the electrical resistance changes. The researchers trained the sensor to detect COVID-19 by using machine learning to compare the pattern of electrical resistance signals obtained from the breath of 49 confirmed COVID-19 patients with those from 58 healthy controls and 33 non-COVID lung infection patients in Wuhan, China. Each study participant blew into the device for 2-3 seconds from a distance of 1–2 cm. Once machine learning identified a potential COVID-19 signature, the team tested the accuracy of the device on a subset of participants. In the test set, the device showed 76% accuracy in distinguishing COVID-19 cases from controls and 95% accuracy in discriminating COVID-19 cases from lung infections. The sensor could also distinguish, with 88% accuracy, between sick and recovered COVID-19 patients. Although the test needs to be validated in more patients, it could be useful for screening large populations to determine which individuals need further testing, the researchers say.
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Covid-19 breathalyzer test
- AstraZeneca vaccine: From its origin to side effects, all you need to know
Manufactured first in 2021, the AstraZeneca vaccine, known in India as Covishield, has been facing scrutiny over the years, with many countries banning it back in 2021 itself ...
- Ten-minute breath test to monitor antibiotic concentrations
Test would be quicker and non-invasive but more research needed **ECCMID has now changed name to ESCMID Global, please credit ESCMID Global Congress in all future stories** Exhaled breath may be very ...
- Expert Doctors: If You Notice This Symptom, It Could Be the New COVID Variant
It's dubbed KP.2, and experts say it's taking over as the most prominent strain. Here's what makes KP.2 different from other COVID variants.
- Quiz: Can you pass our 9 question test on the latest theories of COVID-19 transmission
The World Health Organization has issued a report updating terminology and explanations regarding the spread of the novel coronavirus. See if you're up on the latest vocab.
- JN.1 COVID Variant Symptoms vs. Allergy Symptoms
The JN.1 COVID variant has slightly different symptoms than other variants, as well as some differences when compared to seasonal allergies. Here's what doctors want you to know.
Go deeper with Google Headlines on:
Covid-19 breathalyzer test
[google_news title=”” keyword=”Covid-19 breathalyzer test” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
COVID-19 testing
- Scientist Who Sequenced COVID-19 Virus Allowed to Return to Lab After Protest
YouTube / Associated PressThe first Chinese scientist to publish a genomic sequence of the COVID-19 virus—without permission of government health authorities—says he has been allowed to return to his ...
- Cancer Testing Product Market Rising to Greatness Anticipating Market Size’s New Heights
Evolving Cancer Testing Product Market: Pre and Post COVID-19 Strategy The Cancer Testing Product market has exhibited notable growth dynamics. Commencing at $$ Million USD in 2016, it escalated to $$ ...
- Chinese scientist who published COVID-19 virus sequence allowed back in his lab after sit-in protest
The first scientist to publish a sequence of the COVID-19 virus in China says he has been allowed back into his lab after days of protest ...
- COVID-19 outbreak at Blind River’s North Shore Health Network
COVID-19 has been confirmed on the acute care unit of the North Shore Health Network (NSHN), prompting Algoma Public Health to declare an outbreak at the Blind River site.
- ESCMID Presenters Make Case for Saliva Samples in Molecular Infectious Disease Testing
As demonstrated during the COVID-19 pandemic and in newer research, saliva holds promise as an alternative sample type for PCR-based infectious disease testing.
Go deeper with Google Headlines on:
COVID-19 testing
[google_news title=”” keyword=”COVID-19 testing” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]