Battery leverages both iron and oxygen to drive more lithium ions
On paper, it doesn’t seem like Christopher Wolverton’s super lithium-rich battery should work. For one, the novel battery uses iron, an inexpensive metal that has notoriously failed in batteries. And in another difficult feat, the battery leverages oxygen to help drive the chemical reaction, which researchers previously believed would cause the battery to become unstable.
But not only does the battery work, it does so incredibly well.
Teaming up with researchers at Argonne National Laboratory, Wolverton’s group developed a rechargeable lithium-iron-oxide battery that can cycle more lithium ions than its common lithium-cobalt-oxide counterpart.
The result is a much higher capacity battery that could enable smart phones and battery-powered automobiles to last much longer.

“Our computational prediction of this battery reaction is very exciting, but without experimental confirmation, there would be a lot of skeptics,” said Wolverton, professor of materials science and engineering in Northwestern’s McCormick School of Engineering. “The fact that it actually works is remarkable.”
Supported by the US Department of Energy’s Energy Frontier Research Center program, the research was recently published in Nature Energy. Zhenpeng Yao, a PhD student in Wolverton’s laboratory, and Chun Zhan, a postdoctoral fellow at Argonne, served as the paper’s first authors. Wolverton and Yao led the computational development, and Argonne led the experimental component of the research.
Lithium-ion batteries work by shuttling lithium ions back and forth between the anode and the cathode. When the battery is charged, the ions move back to the anode, where they are stored. The cathode is made from a compound that comprises lithium ions, a transition metal, and oxygen. The transition metal, which is typically cobalt, effectively stores and releases electrical energy when lithium ions move from the anode to the cathode and back. The capacity of the cathode is then limited by the number of electrons in the transition metal that can participate in the reaction.
“In the conventional case, the transition metal is doing the reaction,” Wolverton said. “Because there is only one lithium ion per one cobalt, that limits of how much charge can be stored. What’s worse is that current batteries in your cell phone or laptop typically only use half of the lithium in the cathode.”
The lithium-cobalt-oxide battery has been on the market for 20 years, but researchers have long searched for a less expensive, higher capacity replacement. Wolverton’s team has improved upon the common lithium-cobalt-oxide battery by leveraging two strategies: replacing cobalt with iron, and forcing oxygen to participate in the reaction process.
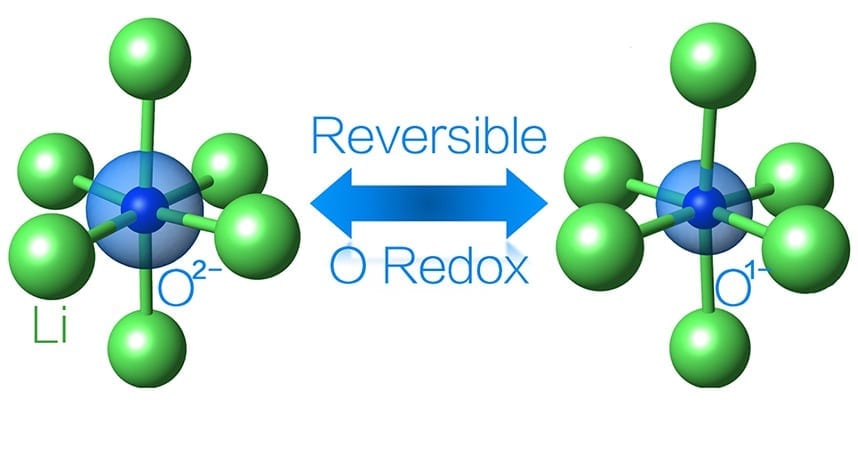
If the oxygen could also store and release electrical energy, the battery would have the higher capacity to store and use more lithium. Although other research groups have attempted this strategy in the past, few have made it work.
“The problem previously was that often, if you tried to get oxygen to participate in the reaction, the compound would become unstable,” Yao said. “Oxygen would be released from the battery, making the reaction irreversible.”
Through computational calculations, Wolverton and Yao discovered a formulation that works reversibly. First, they replaced cobalt with iron, which is advantageous because it’s among the cheapest elements on the periodic table. Second, by using computation, they discovered the right balance of lithium, iron, and oxygen ions to allow the oxygen and iron to simultaneously drive a reversible reaction without allowing oxygen gas to escape.
“Not only does the battery have an interesting chemistry because we’re getting electrons from both the metal and oxygen, but we’re using iron,” Wolverton said. “That has the potential to make a better battery that is also cheap.”
And perhaps even more importantly, the fully rechargeable battery starts with four lithium ions, instead of one. The current reaction can reversibly exploit one of these lithium ions, significantly increasing the capacity beyond today’s batteries. But the potential to cycle all four back and forth by using both iron and oxygen to drive the reaction is tantalizing.
“Four lithium ions for each metal — that would change everything,” Wolverton said. “That means that your phone could last eight times longer or your car could drive eight times farther. If battery-powered cars can compete with or exceed gasoline-powered cars in terms of range and cost, that will change the world.”
Wolverton has filed a provisional patent for the battery with Northwestern’s Innovation and New Ventures Office. Next, he and his team plan to explore other compounds where this strategy could work.
Learn more: New Lithium-Rich Battery Could Last Much Longer
The Latest on: Super lithium-rich battery
[google_news title=”” keyword=”super lithium-rich battery” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- The best TVs for console gaming (2024)on May 1, 2024 at 9:29 am
Back in the 1980s, the best TV for console gaming was literally any TV. Graphics were what they were, and there weren’t televisions on the market yet that catered to console gamers. Basically, if you ...
- Woodside Energy: Climate Becomes An Issueon April 30, 2024 at 8:05 am
Woodside is determined to dramatically expand its oil & gas production in Asia at a time when LNG expansion is being questioned. Read more on WDS stock here.
- Scientists design super-battery made with cheap, readily affordable chemical element, Na — Salt-based cell has surprisingly good energy density and charges in secondson April 29, 2024 at 9:50 pm
Researchers at the Korea Advanced Institute of Science and Technology (KAIST) have developed a high-performance, hybrid sodium-ion battery that charges rapidly and offers impressive energy ...
- Memorial Day sales 2024: Everything we know, including early deals you can shop nowon April 29, 2024 at 2:02 pm
The holiday weekend runs Friday, May 24 through Monday, May 27 — here are some early savings you can score right now.
- Uncovering The Hidden Value In American Lithiumon April 29, 2024 at 12:02 pm
American Lithium's shares appear undervalued given its three projects' economic assessments. Read more about AMLI stock here.
- The 30 best Walmart deals to shop this week — save up to 80% on Mother's Day gifts, gardening supplies and moreon April 26, 2024 at 2:23 pm
Some major deals on board: a Dyson stick vac for just $300, an HP laptop for $240 off and a powerful tower fan at a nearly 40%-plus markdown.
- New Geothermal Technology Could Expand Clean Power Generationon April 24, 2024 at 6:00 am
Long confined to regions with volcanic activity, geothermal promises to become a much more versatile energy source thanks to new technologies ...
- Chinese battery developer unveils new tech with 1,300-mile range that could revolutionize EVs: 'An important piece of the puzzle'on April 24, 2024 at 3:45 am
"It's easy to see what all the hype is about." Chinese battery developer unveils new tech with 1,300-mile range that could revolutionize EVs: 'An important piece of the puzzle' first appeared on The ...
- The Mercedes G-Wagen, the ultimate off-road status symbol, goes electricon April 23, 2024 at 11:31 pm
For those who prefer to grind gravel and bound over boulders while surrounded by luxurious napa leather — and can’t imagine emitting even a single molecule of carbon dioxide while doing so — the fully ...
- Boeing adds a new outside firmon April 22, 2024 at 3:25 pm
Boeing retained The Doerrer Group’s Paul Doerrer on March 7 to lobby on issues related to commercial planes, according to a disclosure filing.
via Google News and Bing News









