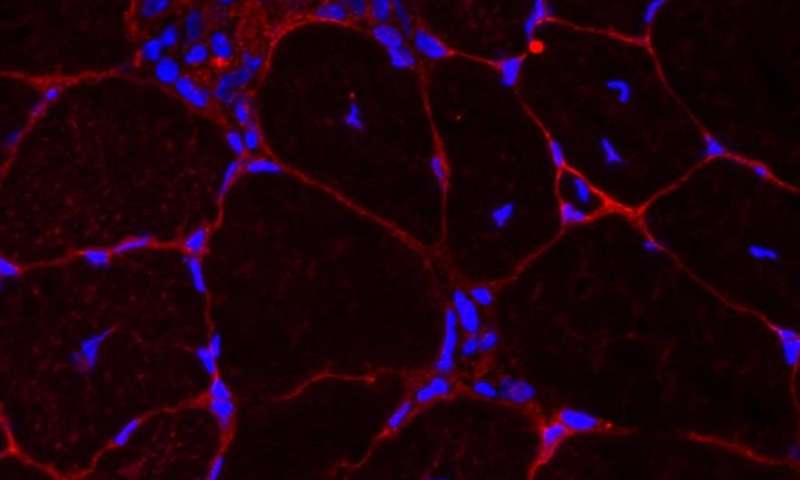
A detail of the muscle tissue the researchers are replicating with their novel 3D material.
RMIT biomedical engineers have successfully produced a 3D material that mimics nature to transform cells into muscle.
The researchers have for the first time incorporated the natural processes of embryonic development to build a material that can more naturally communicate with stem cells for effective tissue repair.
The breakthrough offers hope to people suffering from injuries where their muscles cannot repair themselves due to trauma or disease, and our rapidly ageing population.
Led by Dr Richard Williams from RMIT University, the researchers have combined lab-made peptides with natural proteins and polymers to create a 3D “hydrogel scaffold”.
The scaffold uses natural mechanisms to incorporate signals found in the natural developmental environment to support and engineer stem cells into muscle fibre.
“Our stem cells are at their best during development, where a scaffold drives them to produce all our tissues and organs; yet as people grow and age, they lose this ability and get a build-up of undesirable structures,” Williams said.
“So there is a real need for ways to repair and replace parts of the body as they wear out or become damaged, particularly as our population is now living longer.
“Essentially, we make a scaffold that sends healthy signals to the cells to reorganise themselves to start to grow the small fibres that make up a healthy muscle.
“It’s like travelling back in time when our potential for tissue development and repair was at its best.”
Williams said it is a “simple process” to deliver complex structures inspired by the way nature pieces structures together during development, starting with only two cells.
“We have developed a simple, cheap, yet powerful toolkit that encourages different environments to let different tissues grow,” he said.
“The material properties of natural tissues, like muscles, are really complex and extremely difficult to mimic synthetically.
“Researchers have been producing ‘organising’ signals for years to understand how cells work but only in a flat, or 2D, environment.
“We have found a way to use the proteins present during embryonic development in a 3D scaffold, and use this extra dimension to potentially grow muscle.
“Our research shows that combining a hydrogel with these small proteins is easy and effective. In fact, just 24 to 72 hours after seeding the cells into the tissue, there was a noticeable reorganisation of individual cells into the multi-cellular structures that go on to make up functioning muscle.”
The RMIT researchers are now exploring how they can bioprint muscle with the stem cells already embedded to put nerves, blood vessels and muscles together into lab grown “spare parts”.
The research has been carried out at RMIT University in Melbourne Australia, in collaboration with St Vincent’s Hospital Melbourne, Deakin University and the Australian National University.
The scaffold has been developed in Melbourne’s BioFab3D@AMCD lab – Australia’s first robotics and biomedical engineering centre embedded within a hospital.
The researchers’ findings are published in Biomacromolecules.
How the 3D scaffold works
During embryonic development, stem cells exist within a specific and unique scaffold that allows them to organise into complex structures, such as tissues.
As we age the scaffold changes and these signals are less effective or even wrong, and prevent tissue repair from occurring as it should.
The researchers have been able to replicate the developmental process by producing a 3D material that sends the right signals to stem cells to help replace muscle fibres and repair damage.
The researchers’ method is based on a hydrogel scaffold – a delicate web formed from water and proteins. The scaffold is decorated with the proteins, and provides the structure and signals that the cells need to make muscles grow.
Using the simple, yet effective process, the researchers can take the signals and use them to tell the cells to behave in a certain way.
Learn more: Spare parts from small parts: novel scaffolds to grow muscle
The Latest on: Tissue repair
[google_news title=”” keyword=”Tissue repair” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Tissue repair
- New experimental evidence unlocks a puzzle in vascular tissue engineeringon April 25, 2024 at 9:25 am
Angiogenesis is a process of forming hierarchical vascular networks in living tissues. Its complexity makes the controlled generation of blood vessels in laboratory conditions a highly challenging ...
- 3D Printed Silk Bio-Ink Shows Potential for Knee Meniscus Repairon April 24, 2024 at 7:00 am
Researchers have developed a silk-based bio-ink to 3D print a meniscus for better knee repair and regeneration.
- Tissue Regeneration and Organ Repair: Science or Science Fiction?on April 20, 2024 at 5:00 pm
At the moment, independently of all ethical consideration, substitution of body parts, different from the first attempts at tissue repair, remains a science fiction scenario. Presentations held ...
- Organ regeneration startup names veteran CEO as new leaderon April 18, 2024 at 3:30 am
The new chief executive of Morphoceuticals will chart the company's path into a relatively new and unknown field: Organ regeneration and tissue repair.
- TELA Bio launches OviTex IHR for inguinal hernia repair in USon April 16, 2024 at 4:19 am
Medical technology company TELA Bio has announced the commercial launch of OviTex IHR (Inguinal Hernia Repair) Reinforced Tissue Matrix, a product tailored for laparoscopic and robotic-assisted ...
- Tissue Repair Techniques of the Future: Options for Articular Cartilage Injuryon March 30, 2024 at 5:01 pm
Techvest, LLC's first annual conference on Tissue Repair, Replacement and Regeneration was held October 27-28, 1999 in New York, NY, and featured 50 presenting companies and eight lectures by ...
- Multiple biomaterials for immediate implant placement tissue repair: Current status and future perspectiveson March 29, 2024 at 11:21 am
More information: Xiaoqi Su et al, Multiple biomaterials for immediate implant placement tissue repair: Current status and future perspectives, MedComm – Biomaterials and Applications (2024).
- Sensory neurons play pivotal role in orchestrating tissue repair and regenerationon March 26, 2024 at 5:00 pm
Researchers from Monash University and Osaka University have unveiled a groundbreaking discovery regarding the pivotal role of sensory neurons in orchestrating tissue repair and regeneration ...
- Soft Tissue Repair Market revenue to exceed USD 17 Billion by 2036, says Research Nesteron March 11, 2024 at 5:07 am
New York, March 11, 2024 (GLOBE NEWSWIRE) -- The global soft tissue repair market size is predicted to expand at ~7% CAGR between 2024 and 2036. The market is projected to garner a revenue of USD ...
via Bing News