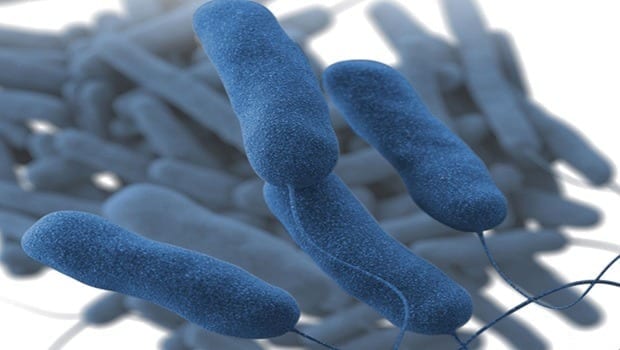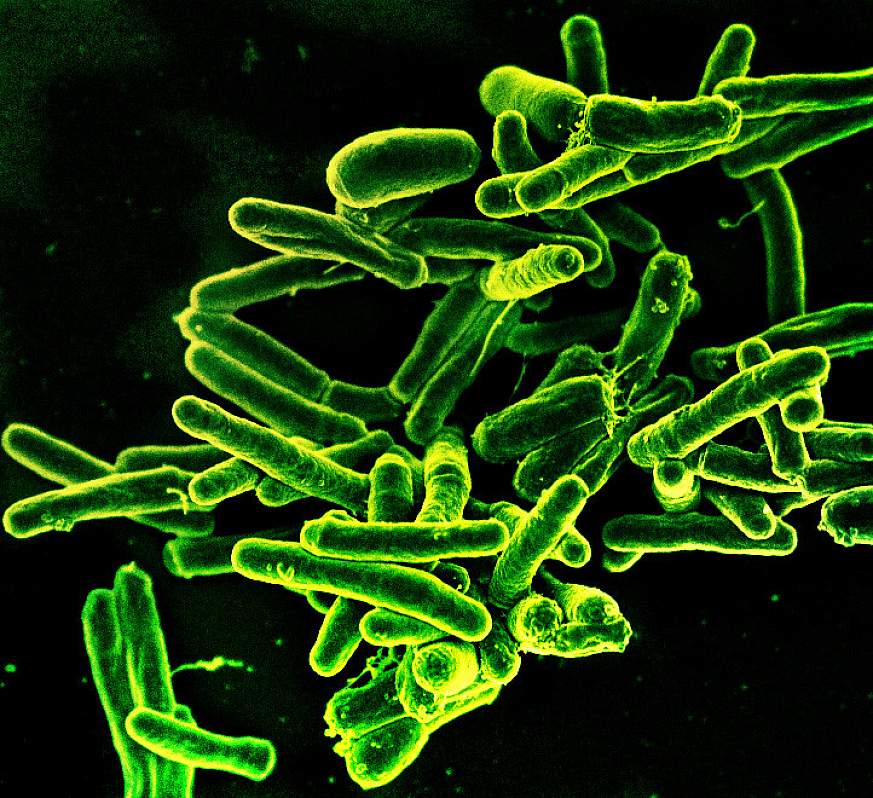
Scanning electron micrograph of Mycobacterium tuberculosis bacteria, which cause TB.NIAID
A clinical trial testing a freeze-dried, temperature-stable experimental tuberculosis (TB) vaccine in healthy adults found that it was safe and stimulated both antibodies and responses from the cellular arm of the immune system.
The Phase 1 trial was supported by the National Institute of Allergy and Infectious Diseases (NIAID), part of the National Institutes of Health. A non-temperature stable form of the candidate previously had been tested in several clinical trials. However, this was the first clinical trial of any subunit TB vaccine candidate in a temperature-stable (thermostable) form. Results are published in Nature Communications.
The experimental vaccine, ID93+GLA-SE, was developed by Christopher B. Fox, Ph.D., and scientists at the Access to Advanced Health Institute (formerly the Infectious Disease Research Institute) in Seattle. It is a recombinant subunit vaccine made from four proteins of Mycobacterium tuberculosis bacteria combined with GLA-SE, an immune-stimulating adjuvant. The freeze-dried formulation does not require refrigeration and is mixed with sterile water just prior to injection. Thermostable vaccines are desirable in settings where maintaining cold or frozen vaccines for long periods can be costly and difficult.
The current trial investigated whether administering temperature-stable vaccine containing both ID93 and GLA-SE in a single vial would be as effective at inducing an immune response as a regimen in which non-thermostable ID93 and liquid GLA-SE are held in two vials and combined prior to injection. A single-vial presentation of a thermostable vaccine would have clear advantages in ease of storage, transport and administration, the investigators note.
Daniel F. Hoft, M.D., Ph.D., director of the Saint Louis University Center for Vaccine Development, led the single-site trial at the university’s School of Medicine. Twenty-three participants received the thermostable single-vial regimen, while 22 participants received the two-vial, non-thermostable regimen. Both vaccine presentations were safe and well-tolerated. Recipients of the single-vialled thermostable vaccine had robust T-cell responses and produced higher levels of antibodies in the blood than those receiving the non-thermostable two-vial presentation.
The investigators note some limitations in this small trial. For example, no established correlates of protection define what immune responses are required for vaccine-induced protection from TB disease. Therefore, it is not possible to say whether the enhanced immune responses seen in the thermostable vaccine formulation would translate to improved protective vaccine efficacy. Nevertheless, they conclude, results of this trial demonstrate “a proof-of-concept that adjuvant-containing vaccines can be formulated in a freeze-dried single-vial presentation without detrimentally impacting clinical immunogenicity or safety characteristics.”
Original Article: Temperature-Stable TB Vaccine Safe, Prompts Immune Response in NIH-Supported Study
More from: National Institutes of Health | Saint Louis University
The Latest Updates from Bing News
Go deeper with Bing News on:
Tuberculosis vaccine
- Plan to set up multi-million shilling vaccine plant in top gear
Kenya will start construction of a multi-million-shilling vaccine manufacturing plant before the end of this year.
- Calls for vaccinating migrants grow amid TB and measles outbreaks in US
The Biden administration is no longer vaccinating migrants in custody at the southern border ahead of their release into the United States, according to the federal agency and a vaccine provider ...
- Three-month anti-TB vaccination drive begins in 12 districts in Andhra Pradesh
Three-month anti-tuberculosis vaccination drive for individuals aged 18+ in 12 districts in State, starting May 16.
- Animal studies lend support to an innovative HIV vaccine strategy — but there’s still a long way to go
Studies in mice and monkeys showed that immune cells could be shepherded to produce antibodies that block a broad swath of HIV strains — a first step toward a potential vaccine.
- Home-grown vaccine demonstrates 100% protection against tuberculosis
The Walter Sisulu and North-West universities unveiled the results of their preclinical trials for the combination vaccine against tuberculosis and COVID-19.
Go deeper with Bing News on:
TB vaccine
- Calls for vaccinating migrants grow amid TB and measles outbreaks in US
The Biden administration is no longer vaccinating migrants in custody at the southern border ahead of their release into the United States, according to the federal agency and a vaccine provider ...
- Home-grown vaccine demonstrates 100% protection against tuberculosis
The Walter Sisulu and North-West universities unveiled the results of their preclinical trials for the combination vaccine against tuberculosis and COVID-19.
- Ministry licenses vaccines against dengue, shingles and pneumococcal 23
Three new vaccines including a dengue fever vaccine, a vaccine against shingles and a pneumococcal 23 vaccine have been licensed by the Ministry of Health (MoH).
- Three-month anti-TB vaccination drive begins in 12 districts
Three-month anti-tuberculosis vaccination drive for individuals aged 18+ in 12 districts in State, starting May 16.
- TB vaccination programme at 508 centres in Krishna district from today
VIJAYAWADA: As a part of the programme to completely eliminate tuberculosis (TB) disease by 2025 in the State, District Medical and Health Officer Dr G Geetha Bai has conducted an awareness programme ...