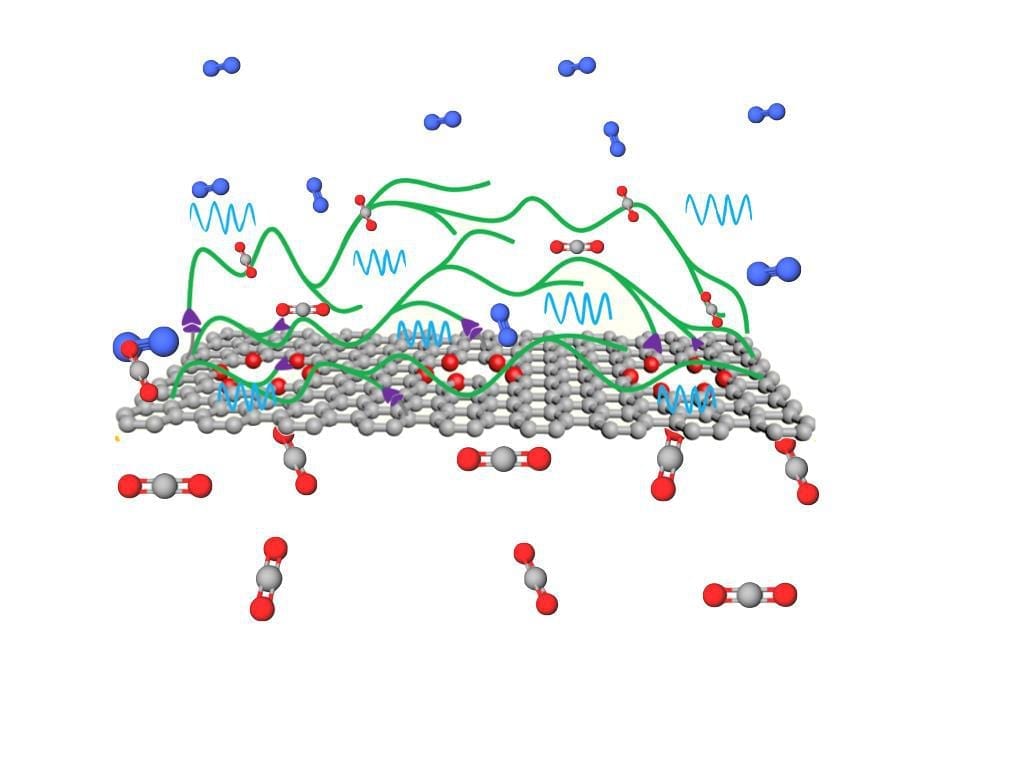
Mineralizing carbon dioxide underground is a potential carbon storage method.
(Illustration by Cortland Johnson | Pacific Northwest National Laboratory)
A new high-profile scientific review article in Nature Reviews Chemistry discusses how carbon dioxide (CO2) converts from a gas to a solid in ultrathin films of water on underground rock surfaces. These solid minerals, known as carbonates, are both stable and common.
“As global temperatures increase, so does the urgency to find ways to store carbon,” said Pacific Northwest National Laboratory (PNNL) Lab Fellow and coauthor Kevin Rosso. “By taking a critical look at our current understanding of carbon mineralization processes, we can find the essential-to-solve gaps for the next decade of work.”
Mineralization underground represents one way to keep CO2 locked away, unable to escape back into the air. But researchers first need to know how it happens before they can predict and control carbonate formation in realistic systems.
“Mitigating human emissions requires fundamentally understanding how to store carbon,” said PNNL chemist Quin Miller, co-lead author of the scientific review featured on the journal cover. “There is a pressing need to integrate simulations, theory, and experiments to explore mineral carbonation problems.”
Below the ground and in the water
Instead of emitting CO2 into the air, one option is to pump it into the ground. Putting CO2 deep underground theoretically sequesters the carbon away. However, gas leaks remain a concern. But if that CO2 gas could be pumped into rocks rich in metals like magnesium and iron, the CO2 can be transformed into stable and common carbonate minerals. PNNL’s Basalt Pilot Project at Wallula is a field site dedicated to studying CO2 storage in carbonates.
Although these subsurface environments are generally dominated by water, the conversion of gaseous carbon dioxide to solid carbonate can also occur when injected CO2 displaces that water, creating extremely thin films of residual water in contact with rocks. But these highly confined systems behave differently than CO2 in contact with a pool of water.
In thin films, the ratio of water and CO2 controls the reaction. Small amounts of metal leach out from the rocks, reacting both in the film and on the rock surface. This leads to the creation of new carbonate materials.
Previous work led by Miller, summarized in the review, showed that magnesium behaves similarly to calcium in thin water films. The nature of the water film plays a central role in how the system reacts.
Understanding how and when these carbonates form requires a combination of laboratory experiments and theoretical modeling studies. Laboratory work allows researchers to tune the ratio of water to CO2 and watch carbonates form in real time. Teams can see what specific chemicals are present at different points in time, providing essential information about reaction pathways.
However, laboratory-based work has its limits. Researchers cannot observe individual molecules or see how they interact. Chemistry models can fill in that gap by predicting how molecules move in exquisite detail, giving conceptual backbone to experiments. They also allow researchers to study mineralization in hard to experimentally access conditions.
“There are important synergies between models and laboratory or field studies,” said MJ Qomi, a professor at the University of California, Irvine and co-lead author of the article. “Experimental data grounds models in reality, while models provide a deeper level of insight into experiments.” Qomi has collaborated with the PNNL team for three years. He recently received a Department of Energy Early Career Research Program award to study carbonate mineralization in adsorbed water films.
From fundamental science to solutions
The team outlined key questions that need answering to make this form of carbon storage practical. Researchers must develop knowledge of how minerals react under different conditions, particularly in conditions that mimic real storage sites, including in ultrathin water films. This should all be done through an integrated combination of modeling and laboratory experiments.
Mineralization has the potential to keep carbon safely stored underground. Knowing how CO2 will react with different minerals can help make sure that what gets pumped underneath the surface stays there. The fundamental science insights from mineralization work can lead to practical CO2 storage systems. The Basalt Pilot Project represents an important study site that bridges small-scale basic science and large-scale research applications.
“This work combines a focus on fundamental geochemical insights with a goal of solving crucial problems,” said Miller. “Without prioritizing decarbonization technologies, the world will continue warming to a degree humanity cannot afford.”
Original Article: Converting Carbon Dioxide to Minerals Underground
More from: Pacific Northwest National Laboratory | University of California Irvine | University of Wyoming
The Latest Updates from Bing News
Go deeper with Bing News on:
Carbon mineralization
- Big Tree Announces Extension of Its Non-Brokered Private Placement
TORONTO, ON / ACCESSWIRE / May 17, 2024 / Big Tree Carbon Inc. (the "Company" or "Big Tree") (TSXV:BIGT) wishes to announce ...
- Morgan explains soil health basics at Soil Health U
Cristine Morgan, chief scientific officer at the Soil Health Institute reviewed the basics at Soil Health U and Trade Show earlier this year.
- Clearing The Air: Why Carbon Storage Must Be Part Of The Solution
When people talk about the climate crisis, they tend to agree on one primary solution: Cut carbon dioxide emissions.
- 20 FINALISTS ANNOUNCED IN XPRIZE CARBON REMOVAL RACING TO REVERSE CLIMATE CHANGE
M competition catalyzed the industry toward game-changing carbon negative solutions LOS ANGELES, May 8, 2024 PRNewswire -- XPRIZE, the world's leader in designing and operating large-scale incentive ...
- Vancouver-based Arca Named as Finalist in Prestigious XPRIZE Carbon Removal Competition
Arca, a leader in carbon mineralization technology, announced its recognition as a top 20 finalist in the prestigious XPRIZE Carbon Removal Competition.
Go deeper with Bing News on:
Carbon storage
- Overlooked coastal marine ecosystems can capture more carbon dioxide than previously thought, finds study
The ability of coastal ecosystems to capture and store carbon dioxide has been underestimated. The question is not just about seagrass meadows and mangrove forests, which have already attracted ...
- DOE falling short in assessing carbon capture risks, audit says
The Government Accountability Office found the department's practices may "undermine the likelihood of project success." ...
- New Barrow carbon storage facility predicted to pump £1.8 billion into economy
Use precise geolocation data and actively scan device characteristics for identification. This is done to store and access information on a device and to provide personalised ads and content, ad and ...
- Alaska Legislature passes carbon-storage bill with additional energy provisions
The Alaska Legislature has passed a bill that combines carbon storage, new regulation of natural gas storage, state financing for new Cook Inlet natural gas development and an expansion of the state’s ...
- Carbon capture technology faces uncertainty despite heavy investment
Carbon capture technology captures CO2 from the air to store underground, seeming like a perfect climate solution.