via Tel Aviv University
Novel RNA technology – Boosting personalized cancer care
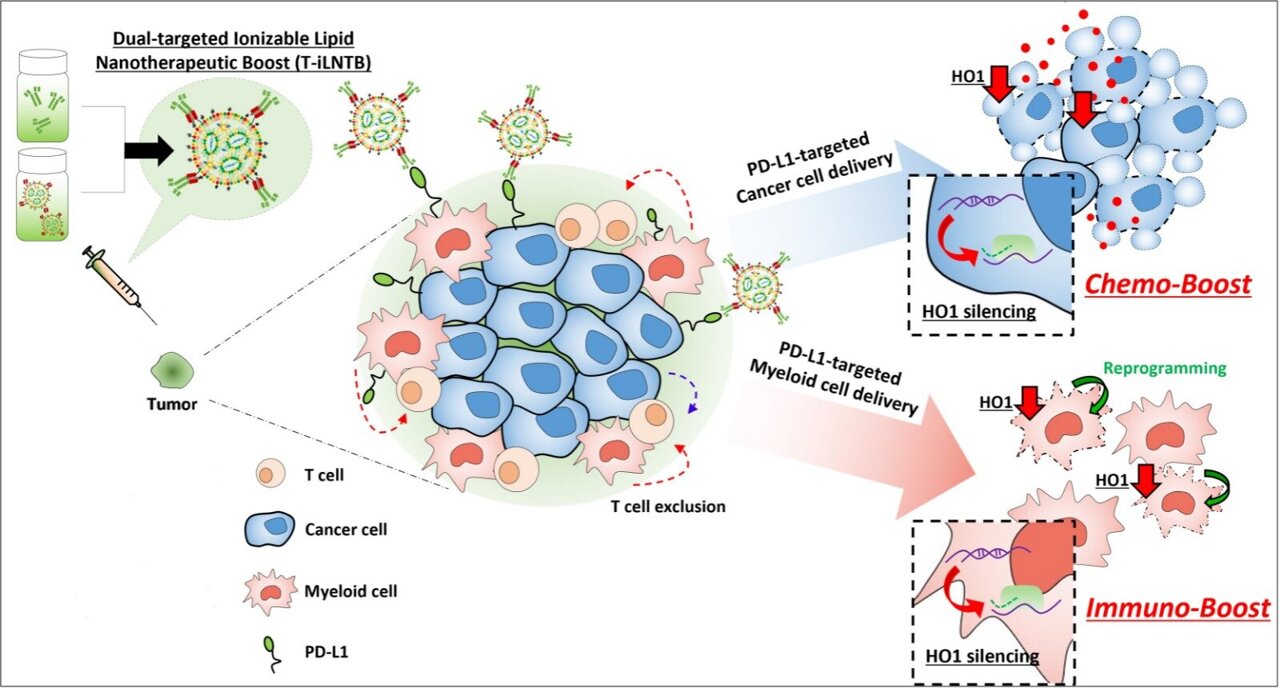
Researchers from Tel Aviv University proved that a drug delivery system based on lipid nanoparticles can utilize RNA to overcome resistance to both chemotherapy and immunotherapy in cancer treatments. The study opens a new path to a personalized and precisely targeted battle against cancer. The results were published in the scientific journal Advanced Materials.
The study was led by TAU Vice President for R&D Prof. Dan Peer, Head of the Laboratory of Precision Nanomedicine at the Shmunis School of Biomedicine and Cancer Research, Wise Faculty of Life Sciences, and a member of the Roman Abramovich Center for Nanoscience and Nanotechnology, together with post-doctoral researcher Dr. Seok-Beom Yong of South Korea. The study was funded via an ERC grant from the European Union and a research scholarship from the Korean government.
Chemo-immunotherapy, which combines chemotherapy with immunotherapy, is considered the most advanced standard of care for various types of cancer. While chemotherapy destroys cancer cells, immunotherapy encourages the cells of the immune system to identify and attack the remaining cancer cells. However, many patients fail to respond to chemo-immunotherapy, which means that the treatment is not sufficiently targeted. Prof. Peer and his team are the first in the world to prove the feasibility of a drug delivery system based on lipid nanoparticles that release their load only at the specifically targeted cells – cancer cells for chemotherapy and immune cells for immunotherapy.
“In our system a single nanoparticle is capable of operating in two different arenas,” explains Prof. Peer. “It increases the sensitivity of cancer cells resistant to chemotherapy, while also reinvigorating immune cells and increasing their sensitivity to cancer cells. Thus, with one precisely targeted nanoparticle we provide two different treatments, at very different sites. We tested this system in two types of lab models – one for metastasized melanoma, and the other for a local solid tumor. In both populations we observed positive effects of our delivery system.”
The new development by Prof. Peer’s team builds from another recent discovery: an enzyme called HO1 is used by cancer cells for both resisting chemotherapy and concealing themselves from the immune system. Silencing HO1 in the tumor is thus considered an optimal strategy in clinical research, but so far, all attempts to silence the enzyme led to severe side effects.
“Chemo-resistant tumors pose a significant challenge in our endless battle against cancer,” says Prof. Peer. “We aim to silence the enzyme HO1 which enables tumors to develop resistance to chemotherapy, and to conceal themselves from the immune system. But existing methods for silencing HO1 resemble using an F-16 fighter jet to blast a tiny ant. Our new nanodrug knows how to precisely target the cancer cells, silence the enzyme, and expose the tumor to chemotherapy, without causing any damage to surrounding healthy cells. Afterwards, the same nanoparticle goes on to the T-cells of the immune system and reprograms them to identify cancer cells. Active, highly aggressive tumors are able to conceal themselves from the immune system, and we restore the immune cells’ ability to recognize the cancer as a foreign body and attack it.”
“This is the first instance of a single drug based on an RNA-loaded nanoparticle doing two very different, even opposite jobs,” adds Prof. Peer. “This is only an initial study, but it has enormous potential in the ongoing fight against cancer.”
Original Article: The nanodrug that attacks the cancer twice A single nanoparticle does two jobs: enhancing the effectiveness of chemotherapy and reinvigorating the immune system
More from: Tel Aviv University
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Personalized cancer care
- The New Frontier: How Smartphone Data Can Revolutionize Cancer Care
Patient-generated "passive data," collected from smartphones, offers an alternative – continuous, real-time insights into the health and behavior of those living with ...
- Intermountain McKay-Dee Hospital brings comprehensive cancer care close to home for residents of northern Utah
OGDEN, Utah (ABC4 Utah) – Intermountain McKay-Dee Hospital is bringing cancer care close to home for residents of Northern Utah with a new state-of-the-art, comprehensive, and personalized cancer ...
- Advancing genomic medicine: National Cancer Center Japan's role in personalized cancer treatment
In a significant stride towards personalized cancer care, the National Cancer Center Japan (NCCJ) has marked a decade of progress in cancer genomic medicine (CGM).
- What happens after a cancer diagnosis? 3 things to be aware of
Princess Kate’s news raised questions about what happens after a diagnosis. Here’s what we can learn. “Cancer is a very emotional term in our society, and for good reason,” said Dr. Ruben Mesa, the ...
- With younger women getting breast cancer, national panel lowers mammogram screening age to 40
Breast cancer cases are increasing among younger women, which means it now makes sense to start regular screening younger, a federal task force says.
Go deeper with Google Headlines on:
Personalized cancer care
[google_news title=”” keyword=”personalized cancer care” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
RNA technology
- Startup that raised over $100M gets FDA approval for cancer test
St. Louis gastrointestinal health startup Geneoscopy has received U.S. Food and Drug Administration approval for the screening test it has developed to detect colorectal cancer. Geneoscopy, with the ...
- FDA Oks ColoSense, a Stool Test for Early Colorectal Cancer Detection
The FDA has approved ColoSense, a stool screening test with 93% accuracy for colon cancer in average-risk adults over the age of 45. This noninvasive option may boost early detection rates.
- FDA approves Geneoscopy’s ColoSense test for CRC screening
The ColoSense test is to be used with the ColoSense Collection Kit, Test Kit, Software, and specific instruments.
- FDA Approves ColoSense™ – Geneoscopy's Noninvasive Multi-target Stool RNA (mt-sRNA) Colorectal Cancer Screening Test
Geneoscopy, Inc. announced that the U.S. Food and Drug Administration (FDA) approved its noninvasive colorectal cancer screening test, ColoSense.
- Targeted DNA RNA Sequencing Market To Reach USD 51.3 Billion By 2032 | DataHorizzon Research
In 2023, the targeted DNA RNA sequencing market experienced growth, primarily driven by the cost-saving advantages offered by this technology. In contrast to non-targeted approaches, where complete ...
Go deeper with Google Headlines on:
RNA technology
[google_news title=”” keyword=”RNA technology” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]