Though considered a promising large-scale energy storage device, the vanadium redox battery’s use has been limited by its inability to work well in a wide range of temperatures and its high cost. But new research indicates that modifying the battery’s electrolyte solution significantly improves its performance. So much so that the upgraded battery could improve the electric grid’s reliability and help connect more wind turbines and solar panels to the grid.
In a paper published by the journalAdvanced Energy Materials,researchers at the Department of Energy’s Pacific Northwest National Laboratory found that adding hydrochloric acid to the sulfuric acid typically used in vanadium batteries increased the batteries’ energy storage capacity by 70 percent and expanded the temperature range in which they operate.
“Our small adjustments greatly improve the vanadium redox battery,” said lead author and PNNL chemist Liyu Li. “And with just a little more work, the battery could potentially increase the use of wind, solar and other renewable power sources across the electric grid.”
Unlike traditional power, which is generated in a reliable, consistent stream of electricity by controlling how much coal is burned or water is sent through dam turbines, renewable power production depends on uncontrollable natural phenomena such as sunshine and wind. Storing electricity can help smooth out the intermittency of renewable power while also improving the reliability of the electric grid that transmits it. Vanadium batteries can hold on to renewable power until people turn on their lights and run their dishwashers. Other benefits of vanadium batteries include high efficiency and the ability to quickly generate power when it’s needed as well as sit idle for long periods of time without losing storage capacity.
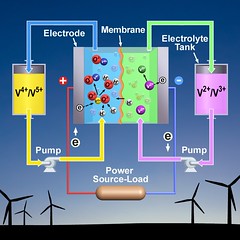
A vanadium battery is a type of flow battery, meaning it generates power by pumping liquid from external tanks to the battery’s central stack, or a chamber where the liquids are mixed. The tanks contain electrolytes, which are liquids that conduct electricity. One tank has the positively-charged vanadium ion V5+ floating in its electrolyte. And the other tank holds an electrolyte full of a different vanadium ion, V2+. When energy is needed, pumps move the ion-saturated electrolyte from both tanks into the stack, where a chemical reaction causes the ions to change their charge, creating electricity.
To charge the battery, electricity is sent to the vanadium battery’s stack. This causes another reaction that restores the original charge of vanadium ions. The electrical energy is converted into chemical energy stored in the vanadium ions. The electrolytes with their respective ions are pumped back into to their tanks, where they wait until electricity is needed and the cycle is started again.
A battery’s capacity to generate electricity is limited by how many ions it can pack into the electrolyte. Vanadium batteries traditionally use pure sulfuric acid for their electrolyte. But sulfuric acid can only absorb so many vanadium ions.