NIH-funded study of people with type 1 diabetes shows safety, efficacy benefits of new system
A multicenter randomized clinical trial evaluating a new artificial pancreas system – which automatically monitors and regulates blood glucose levels – has found that the new system was more effective than existing treatments at controlling blood glucose levels in people with type 1 diabetes. The trial was primarily funded by the National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK), part of the National Institutes of Health.
The study showed that the system improved participants’ blood glucose control throughout the day and overnight. The latter is a common but serious challenge for children and adults with type 1 diabetes, since blood sugar can drop to dangerously low levels when a person is asleep. The research is published in the New England Journal of Medicine.
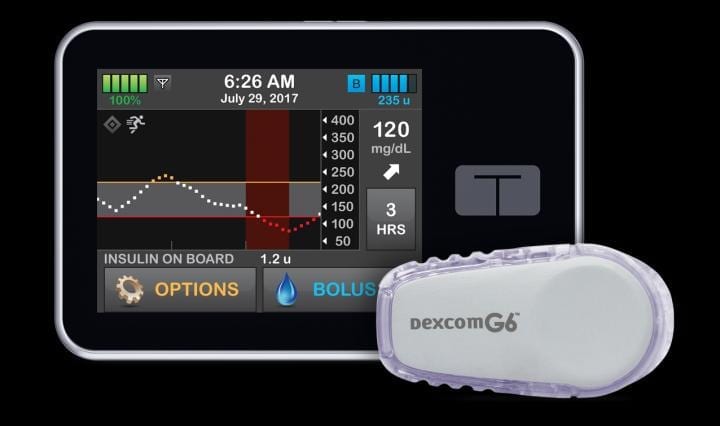
The artificial pancreas, also known as closed-loop control, is an “all-in-one” diabetes management system that tracks blood-sugar levels using a continuous glucose monitor (CGM) and automatically delivers the hormone insulin when needed using an insulin pump. The system replaces reliance on testing by fingerstick or CGM with separate delivery of insulin by multiple daily injections or a pump.
The International Diabetes Closed-Loop (iDCL) Study involves five separate artificial pancreas clinical protocols implemented by 10 research centers in the United States and Europe. This six-month study was the third phase in a series of trials. It was conducted with participants living their usual day-to-day lives, so the researchers could best understand how the system works in typical daily routines.
“Testing the safety and effectiveness of new technologies in real-world settings is critical to prove the usability of these systems by people with diabetes to achieve a better daily control of their blood glucose levels,” said Guillermo Arreaza-Rubín, M.D., director of NIDDK’s Diabetes Technology Program and project scientist for the study. “Earlier technologies have made the management of type 1 diabetes easier, and this research shows that this artificial pancreas system has the potential to improve health of people living with type 1 diabetes, while also potentially lifting much of the burden of care from those with the disease and their caregivers.”
This iDCL protocol enrolled 168 participants age 14 or older with type 1 diabetes. They were randomly assigned to use either the artificial pancreas system called Control-IQ or sensor-augmented pump (SAP) therapy with a CGM and insulin pump that did not automatically adjust insulin throughout the day. Participants had contact with study staff every two to four weeks to download and review device data. No remote monitoring of the systems was done, so that the study would reflect real-world use.
The researchers found that users of the artificial pancreas system significantly increased the amount of time with their blood glucose levels in the target range of 70 to 180 mg/dL by an average of 2.6 hours per day since beginning the trial, while the time in range in the SAP group remained unchanged over six months. Artificial pancreas users also showed improvements in time spent with high and low blood glucose, hemoglobin A1c, and other measurements related to diabetes control compared to the SAP group. High adherence to device use in both groups and 100% participant retention were important strengths of the study. During the study, no severe hypoglycemia events occurred in either group. Diabetic ketoacidosis occurred in one participant in the artificial pancreas group due to a problem with equipment that delivers insulin from the pump.
The Control-IQ technology was derived from a system originally developed at the University of Virginia, Charlottesville, (UVA) by a team led by Boris Kovatchev, Ph.D., director of the UVA Center for Diabetes Technology with funding support from NIDDK. In this system, the insulin pump is programmed with advanced control algorithms based on a mathematical model that uses the person’s glucose monitoring information to automatically adjust the insulin dose. Tandem Diabetes Care has submitted the results to the U.S. Food and Drug Administration for approval to market the Control-IQ system.
“This artificial pancreas system has several unique features that improve glucose control beyond what is achievable using traditional methods,” said Kovatchev. “In particular, there is a special safety module dedicated to prevention of hypoglycemia, and there is gradually intensified control overnight to achieve near-normal blood sugar levels every morning.”
Kovatchev was co-lead author of the study with colleagues Sue A. Brown, M.D., of UVA and Roy Beck, M.D., Ph.D., from the Jaeb Center for Health Research, Tampa, Florida, which was the coordinating center for the study.
“Artificial pancreas technology has tremendous potential to improve the day-to-day lives of people with type 1 diabetes,” said NIDDK Director Griffin P. Rodgers, M.D. “By making management of type 1 diabetes easier and more precise, this technology could reduce the daily burden of this disease, while also potentially reducing diabetes complications including eye, nerve and kidney diseases.”
Learn more: Artificial pancreas system better controls blood glucose levels than current technology
The Latest on: Artificial pancreas
[google_news title=”” keyword=”artificial pancreas” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Artificial pancreas
- Experimental Type 1 Diabetes Drug Shields Pancreas Cells from the Usual Crippling Immune System Attackon May 7, 2024 at 11:12 am
Scientists say an experimental antibody drug appears to prevent and reverse type 1 diabetes in mice—and often lengthen their lives.
- UVA study confirms AI provides efficiency when added to artificial pancreas regulating diabeteson May 2, 2024 at 1:06 pm
Adding advanced artificial intelligence to an artificial pancreas regulating type 1 diabetes is safe and improves the system’s efficiency.
- Study finds AI improves efficiency of artificial pancreas system for Type 1 diabeticson May 2, 2024 at 12:45 pm
UVA researchers compared an advanced experimental artificial pancreas system with an artificial pancreas algorithm incorporating AI.
- Adding AI to artificial pancreas enhances efficiency, study findson May 2, 2024 at 11:50 am
Adding advanced artificial intelligence to an artificial pancreas regulating type 1 diabetes is safe and improves the system's efficiency, a first-of-its-kind study from the University of Virginia ...
- Adding AI to artificial pancreas enhances efficiency, study findson May 1, 2024 at 5:00 pm
Adding advanced artificial intelligence to an artificial pancreas regulating type 1 diabetes is safe and improves the system’s efficiency, a first-of-its-kind study from the University of ...
- Artificial pancreas: NHS rolls out hybrid closed loop systems for people with type 1 diabeteson April 9, 2024 at 5:00 pm
This month, the NHS will start contacting people to offer them an artificial pancreas, a system which sees a glucose sensor under the skin work out how much insulin should be delivered via a pump.
- Artificial Pancreas Pipeline Report including Stages of Development, Segments, Region and Countries, Regulatory Path and Key Companies 2024on April 3, 2024 at 5:00 pm
Dublin, April 04, 2024 (GLOBE NEWSWIRE) -- The "Artificial Pancreas Pipeline Report including Stages of Development, Segments, Region and Countries, Regulatory Path and Key Companies, 2024 Update ...
- Game-Changing Artificial Pancreas Set to Improve Lives of Type 1 Diabetics in UKon April 2, 2024 at 11:38 am
The artificial pancreas device combines an under-the-skin sensor to measure blood glucose with existing insulin pump technology, seen above, to more seamlessly deliver insulin to users when needed.
- World-first artificial pancreas roll-out will see up to 150,000 diabetics get the life-changing gadgets on the NHSon April 1, 2024 at 4:52 pm
The NHS is giving up to 150,000 patients an artificial pancreas in what has been hailed a 'landmark moment' for type 1 diabetics. The world-first rollout will effectively spell the end of painful ...
- Diabetes patients to be offered artificial-pancreas technologyon March 31, 2024 at 5:00 pm
Tens of thousands of people with type 1 diabetes in England are to be offered a new technology, dubbed an artificial pancreas, to help manage the condition. The system uses a glucose sensor under ...
via Bing News