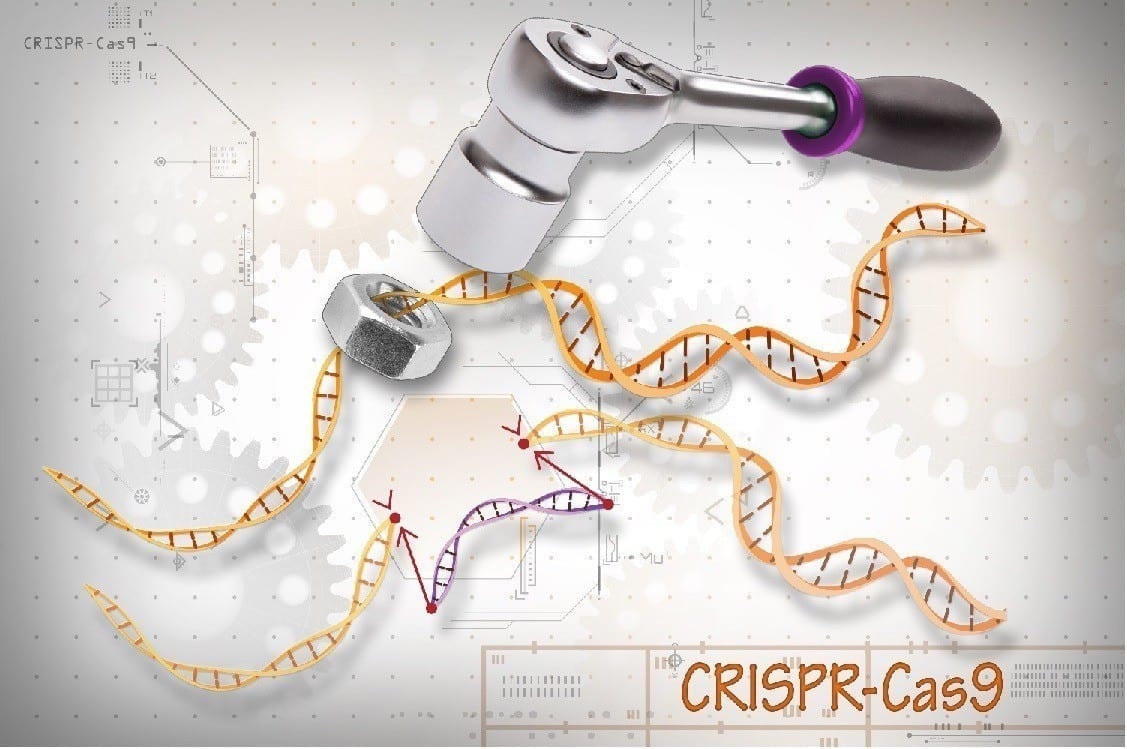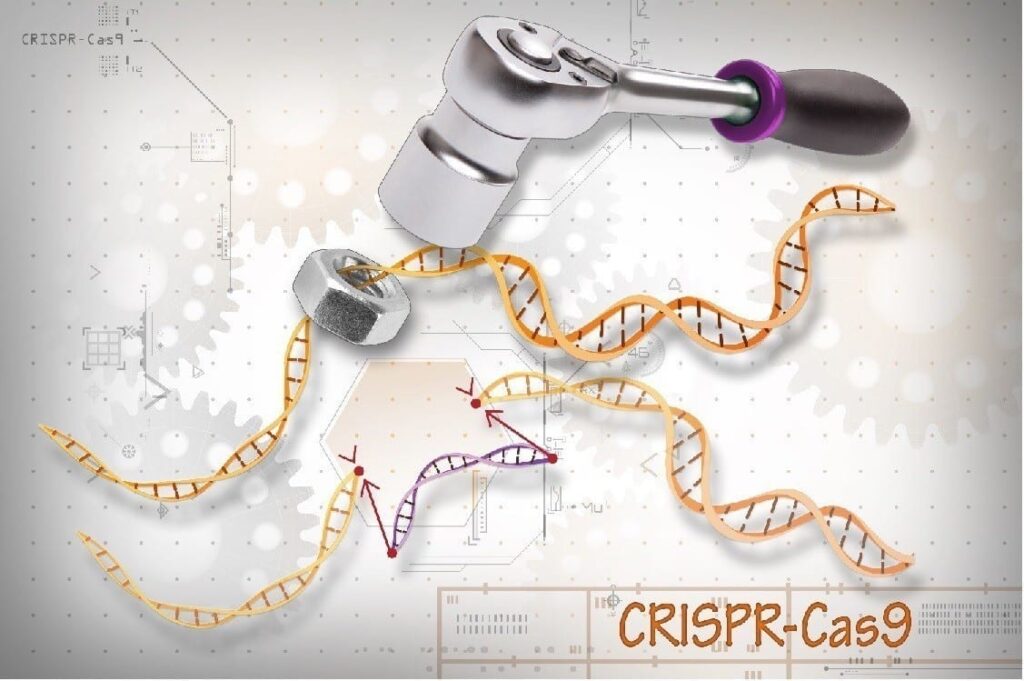
Scientists for the first time have used CRISPR gene editing to halt the progression of Duchenne muscular dystrophy (DMD) in a large mammal, according to a study by UT Southwestern that provides a strong indication that a lifesaving treatment may be in the pipeline.
The research published in Science documents unprecedented improvement in the muscle fibers of dogs with DMD – the most common fatal genetic disease in children, caused by a mutation that inhibits the production of dystrophin, a protein critical for muscle function.
Researchers used a single-cut gene-editing technique to restore dystrophin in muscle and heart tissue by up to 92 percent of normal levels. Scientists have estimated a 15 percent threshold is needed to significantly help patients.
“Children with DMD often die either because their heart loses the strength to pump, or their diaphragm becomes too weak to breathe,” said Dr. Eric Olson, Director of UT Southwestern’s Hamon Center for Regenerative Science and Medicine. “This encouraging level of dystrophin expression would hopefully prevent that from happening.”
DMD, which affects one in 5,000 boys, leads to muscle and heart failure, and premature death by the early 30s. Patients are forced into wheelchairs as their muscles degenerate and eventually onto respirators as their diaphragms weaken. No effective treatment exists, though scientists have known for decades that a defect in the dystrophingene causes the condition.
The Science study establishes the proof-of-concept for single-cut gene editing in dystrophic muscle and represents a major step toward a clinical trial. Already Dr. Olson’s team has corrected DMD mutations in mice and human cells by making single cuts at strategic points of the mutated DNA.
The latest research applied the same technique in four dogs that shared the type of mutation most commonly seen in DMD patients. Scientists used a harmless virus called adeno-associated virus (AAV) to deliver CRISPR gene-editing components to exon 51, one of the 79 exons that comprise the dystrophin gene.
CRISPR edited the exon, and within several weeks the missing protein was restored in muscle tissue throughout the body, including 92 percent correction in the heart and 58 percent in the diaphragm, the main muscle needed for breathing.
“Our strategy is different from other therapeutic approaches for DMD because it edits the mutation that causes the disease and restores normal expression of the repaired dystrophin,” said Dr. Leonela Amoasii, lead author of the study and Assistant Instructor of Molecular Biology in Dr. Olson’s lab. “But we have more to do before we can use this clinically.”
The lab will next conduct longer-term studies to measure whether the dystrophin levels remain stable and to ensure the gene edits do not have adverse side effects.
Dr. Olson hopes the next step beyond dogs is a clinical trial, which would be among several that UT Southwestern’s gene therapy center aims to launch in the coming years to address numerous deadly childhood diseases.
In the meantime, Dr. Olson’s recent work has spawned a biotechnology company, Exonics Therapeutics Inc., which is working to further optimize and bring this technology to the clinic. Exonics intends to extend the approach to additional DMD mutations, as well as other neuromuscular diseases. Exonics has licensed the technology from UT Southwestern.
Learn more: CRISPR halts Duchenne muscular dystrophy progression in dogs
The Latest on: Duchenne muscular dystrophy
[google_news title=”” keyword=”Duchenne muscular dystrophy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Duchenne muscular dystrophy
- Study Highlights Significant Increases in Utilization, Spending on DMD Drugs in Medicaidon May 16, 2024 at 5:00 pm
The findings add to recent research on the growing utilization, expenditure, and prices of Duchenne muscular dystrophy (DMD) therapies in the current landscape, an area health care policy could ...
- Israel develops worm-based research method on muscular dystrophyon May 16, 2024 at 12:11 pm
Israeli researchers at the Bar Ilan University (BIU) have developed a platform for modeling deadly human muscular system diseases in worms, the BIU said in a statement on Thursday.
- Jar of Hope staging unique race to save kidson May 15, 2024 at 7:49 am
STATEN ISLAND, N.Y. — Jim Raffone, founder/CEO of JAR of Hope ( https://www.jarofhope.org/ ), will play host to a unique half-marathon to save children with a rare and fatal disease.
- Solid Biosciences Provides First Quarter 2024 Business Update and Financial Resultson May 15, 2024 at 5:30 am
Received Rare Pediatric Disease and Orphan Drug Designation for Duchenne musculardystrophy (Duchenne) gene therapy candidate SGT-003 with ...
- Edgewise: Intriguing Drug Candidate, But Consider Selling After Recent Runon May 10, 2024 at 10:49 am
Edgewise reveals strong cash position and progressing clinical-stage programs, sparking a bull run. Find out why this is a good time to sell EWTX stock.
- Duchenne Muscular Dystrophyon May 9, 2024 at 4:59 pm
Rome, MD, MPH, examined how the FDA’s accelerated approval process has moved 5 genetically targeted treatments for Duchenne muscular dystrophy (DMD) through its pipeline despite limited evidence ...
- ASGCT 2024: Kate Therapeutics Unveils Gene Therapy Platform and Product Pipeline Progresson May 9, 2024 at 7:00 am
KateTx debuted less than a year ago with a $51 million Series A round co-led by Westlake Village BioPartners and Versant Ventures.
- Young boy dies in trial for Pfizer Duchenne gene therapyon May 7, 2024 at 2:57 pm
A boy with Duchenne muscular dystrophy died after receiving a Pfizer experimental gene therapy. The company is pausing its trial in older boys.
- Pfizer reports patient death in Duchenne gene therapy studyon May 7, 2024 at 2:49 pm
A young patient died due to cardiac arrest after receiving Pfizer's experimental gene therapy being tested in a mid-stage trial for a muscle-wasting disorder called Duchenne muscular dystrophy(DMD), ...
- Patient dies in Pfizer study of Duchenne gene therapyon May 7, 2024 at 2:39 pm
Pfizer said the patient, a young boy who was treated earlier last year, had died suddenly. The company is working with trial researchers to investigate further.
via Bing News