CREDIT
Masaaki Sadakiyo / International Institute for Carbon-Neutral Energy Research, Kyushu University
Research team Kyushu University creates electrolytic flow cell to produce alcohols from carboxylic acids
Interest in renewable energy continues to burgeon. Many renewables, though, can be frustratingly intermittent – when the sun stops shining, or the wind stops blowing, the power flickers. The fluctuating supply can be partly smoothed-out by energy storage during peak production times. However, storing electricity is not without its challenges either.
Recently, a team at the International Institute for Carbon-Neutral Energy Research (I2CNER), within southern Japan’s Kyushu University, created a device to store energy in chemical form through continuous electrolysis.
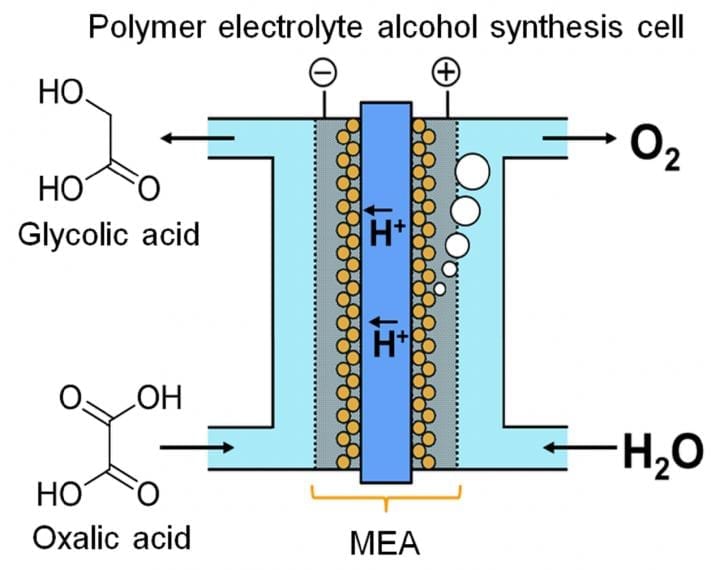
The researchers noted that glycolic acid (GC) has a much greater energy capacity than hydrogen, one of the more popular energy-storage chemicals. GC can be produced by four-electron reduction of oxalic acid (OX), a widely available carboxylic acid. As described in their publication in Scientific Reports, the team devised an electrolytic cell based on a novel membrane-electrode assembly. Sandwiched between two electrodes are an iridium oxide-based anode and a titanium dioxide (TiO2)-coated titanium (Ti) cathode, linked by a polymer membrane.
“Flow-type systems are very important for energy storage with liquid-phase reaction,” study lead author Masaaki Sadakiyo explains. “Most electrolyzers producing alcohols operate a batch process, which is not suitable for this purpose. In our device, by using a solid polymer electrolyte in direct contact with the electrodes, we can run the reaction as a continuous flow without addition of impurities (e.g. electrolytes). The OX solution can effectively be thought of as a flowable electron pool.”
Another key consideration is the cathode design. The cathodic reaction is catalyzed by anatase TiO2. To ensure a solid connection between catalyst and cathode, the team “grew” TiO2 directly on Ti in the form of a mesh or felt. Electron microscope images show the TiO2 as a wispy fuzz, clinging to the outside of the Ti rods like a coating of fresh snow. In fact, its job is to catalyze the electro-reduction of OX to GC. Meanwhile, at the anode, water is oxidized to oxygen.
The team found that the reaction accelerated at higher temperatures. However, turning the heat up too high encouraged an unwanted by-process – the conversion of water to hydrogen. The ideal balance between these two effects was at 60°C. At this temperature, the device could be further optimized by slowing the flow of reactants, while increasing the amount of surface area available for the reaction.
Interestingly, even the texture of the fuzzy TiO2 catalyst made a major difference. When TiO2 was prepared as a “felt,” by growing it on thinner and more densely packed Ti rods, the reaction occurred faster than on the “mesh” – probably because of the greater surface area. The felt also discouraged hydrogen production, by blanketing the Ti surface more snugly than the mesh, preventing the exposure of bare Ti.
“In the right conditions, our cell converts nearly 100% of OX, which we find very encouraging,” co-author Miho Yamauchi says. “We calculate that the maximum volumetric energy capacity of the GC solution is around 50 times that of hydrogen gas. To be clear, the energy efficiency, as opposed to capacity, still lags behind other technologies. However, this is a promising first step to a new method for storing excess current.”
Learn more: Exploring electrolysis for energy storage
The Latest on: Flow-type polymer electrolyte cell
[google_news title=”” keyword=”flow-type polymer electrolyte cell” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- New technology changes how proteins in individual cells are studiedon May 8, 2024 at 7:13 am
Researchers from Karolinska Institutet, together with Pixelgen Technologies, have developed and applied a technique that makes it possible to map proteins in individual cells in a completely new way.
- What do epithelial cells in urine mean?on April 30, 2024 at 5:00 pm
These cells cover a person’s skin and also occur along the surfaces of the digestive tract, the internal organs, and blood vessels. There are three types of ... of urine flow, and then use ...
- Sepion Advances EV Battery Safety with AI-Driven Non-Flammable Electrolyteon April 30, 2024 at 12:26 pm
Press Release Sepion Technologies, a leading developer of nanoengineered separator coatings and liquid electrolytes for faster charging, lower-cost, and energy-dense lithium-ion and lithium-metal ...
- Discovering exceptional oxide ion conductivity at lower temperatureson April 24, 2024 at 5:00 pm
Feb. 3, 2022 — A new high-temperature polymer fuel cell that operates at 80-160 degrees Celsius, with a higher-rated power density than state-of-the-art fuel cells, solves the longstanding ...
- Insulin Resistance – What You Need to Knowon April 21, 2024 at 5:00 pm
Insulin resistance is when cells in your muscles ... Insulin resistance and prediabetes can both lead to type 2 diabetes. So much glucose stays in your bloodstream, you will need medication ...
- Electromagnetically driven flow in unsupported electrolyte layers: lubrication theory and linear stability of annular flowon April 12, 2024 at 8:32 am
Department of Mathematics, Swinburne University of Technology, Hawthorn, Victoria 3122, Australia We consider a thin horizontal layer of a non-magnetic electrolyte ... type deformations of the free ...
- Flow Cytometry Work Cell from Hudsonon April 5, 2024 at 5:00 pm
The Automated Flow Cytometry work cell provides scientists with an automated sample acquisition system for cellular analysis research. This integrated platform combines the Guava EasyCyte Plus ...
- The Most Affordable Hydrogen-powered Car In 2024on March 12, 2024 at 4:09 pm
These are made from thin sets of catalyst plates (anode and cathode) with a Polymer Electrolyte Membrane sandwiched in between. The oxygen and hydrogen that flow into the fuel cell unit combine in ...
- DNBSEQ-G400: Flow cell system for genome sequencingon January 15, 2024 at 1:33 am
*means that “the product model names, DNBSEQ-G400 and MGISEQ-2000, are used in the selected oversea countries and China, respectively.” **means that “*Unless otherwise informed, StandardMPS ...
- Better Ion Transport Through Polymer Chemistry: Polymer Electrolytes and Ion-conducting Membraneson January 4, 2024 at 7:12 am
Electrochemical processes in devices like solar cells, fuel cells ... will zoom out to describe how mechanically tough polymer membranes with high ionic conductivity control the flow of electrolytes ...
via Google News and Bing News









