Berkeley Lab advance is first demonstration of efficient, light-powered production of fuel via artificial photosynthesis
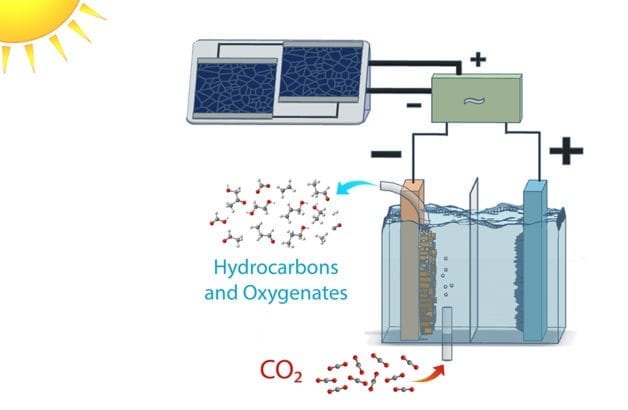
Scientists at the Department of Energy’s Lawrence Berkeley National Laboratory (Berkeley Lab) have harnessed the power of photosynthesis to convert carbon dioxide into fuels and alcohols at efficiencies far greater than plants. The achievement marks a significant milestone in the effort to move toward sustainable sources of fuel.
Many systems have successfully reduced carbon dioxide to chemical and fuel precursors, such as carbon monoxide or a mix of carbon monoxide and hydrogen known as syngas. This new work, described in a study published in the journal Energy and Environmental Science, is the first to successfully demonstrate the approach of going from carbon dioxide directly to target products, namely ethanol and ethylene, at energy conversion efficiencies rivaling natural counterparts.
The researchers did this by optimizing each component of a photovoltaic-electrochemical system to reduce voltage loss, and creating new materials when existing ones did not suffice.
“This is an exciting development,” said study principal investigator Joel Ager, a Berkeley Lab scientist with joint appointments in the Materials Sciences and the Chemical Sciences divisions. “As rising atmospheric CO2 levels change Earth’s climate, the need to develop sustainable sources of power has become increasingly urgent. Our work here shows that we have a plausible path to making fuels directly from sunlight.”
That sun-to-fuel path is among the key goals of the Joint Center for Artificial Photosynthesis(JCAP), a DOE Energy Innovation Hub established in 2010 to advance solar fuel research. The study was conducted at JCAP’s Berkeley Lab campus.
The initial focus of JCAP research was tackling the efficient splitting of water in the photosynthesis process. Having largely achieved that task using several types of devices, JCAP scientists doing solar-driven carbon dioxide reduction began setting their sights on achieving efficiencies similar to those demonstrated for water splitting, considered by many to be the next big challenge in artificial photosynthesis.
Another research group at Berkeley Lab is tackling this challenge by focusing on a specific component in a photovoltaic-electrochemical system. In a study published today, they describe a new catalyst that can achieve carbon dioxide to multicarbon conversion using record-low inputs of energy.
Not just for noon
For this JCAP study, researchers engineered a complete system to work at different times of day, not just at a light energy level of 1-sun illumination, which is equivalent to the peak of brightness at high noon on a sunny day. They varied the brightness of the light source to show that the system remained efficient even in low light conditions.
When the researchers coupled the electrodes to silicon photovoltaic cells, they achieved solar conversion efficiencies of 3 to 4 percent for 0.35 to 1-sun illumination. Changing the configuration to a high-performance, tandem solar cell connected in tandem yielded a conversion efficiency to hydrocarbons and oxygenates exceeding 5 percent at 1-sun illumination.
At left is a surface view of a bimetallic copper-silver nanocoral cathode taken from a scanning electron micrograph. To the right is an energy-dispersive X-ray image of the cathode with the copper (in pink/red) and silver (in green) highlighted. (Credit: Gurudayal/Berkeley Lab)
“We did a little dance in the lab when we reached 5 percent,” said Ager, who also holds an appointment as an adjunct professor at UC Berkeley’s Materials Science and Engineering Department.
Among the new components developed by the researchers are a copper-silver nanocoral cathode, which reduces the carbon dioxide to hydrocarbons and oxygenates, and an iridium oxide nanotube anode, which oxidizes the water and creates oxygen.
“The nice feature of the nanocoral is that, like plants, it can make the target products over a wide range of conditions, and it is very stable,” said Ager.
The researchers characterized the materials at the National Center for Electron Microscopy at the Molecular Foundry, a DOE Office of Science User Facility at Berkeley Lab. The results helped them understand how the metals functioned in the bimetallic cathode. Specifically, they learned that silver aids in the reduction of carbon dioxide to carbon monoxide, while the copper picks up from there to reduce carbon monoxide further to hydrocarbons and alcohols.
Seeking better, low-energy breakups
Because carbon dioxide is a stubbornly stable molecule, breaking it up typically involves a significant input of energy.
“Reducing CO2 to a hydrocarbon end product like ethanol or ethylene can take up to 5 volts, start to finish,” said study lead author Gurudayal, postdoctoral fellow at Berkeley Lab. “Our system reduced that by half while maintaining the selectivity of products.”
Notably, the electrodes operated well in water, a neutral pH environment.
“Research groups working on anodes mostly do so using alkaline conditions since anodes typically require a high pH environment, which is not ideal for the solubility of CO2,” said Gurudayal. “It is very difficult to find an anode that works in neutral conditions.”
The researchers customized the anode by growing the iridium oxide nanotubes on a zinc oxide surface to create a more uniform surface area to better support chemical reactions.
“By working through each step so carefully, these researchers demonstrated a level of performance and efficiency that people did not think was possible at this point,” said Berkeley Lab chemist Frances Houle, JCAP deputy director for Science and Research Integration, who was not part of the study. “This is a big step forward in the design of devices for efficient CO2 reduction and testing of new materials, and it provides a clear framework for the future advancement of fully integrated solar-driven CO2-reduction devices.”
Learn more: Solar-to-Fuel System Recycles CO2 to Make Ethanol and Ethylene
The Latest on: Solar fuel
[google_news title=”” keyword=”solar fuel” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- 12 Stocks to Play Growing Energy Demand, From Oil and Gas to Nuclear and Solaron May 17, 2024 at 11:35 am
Energy has been at the center of some of the biggest stories in the past year, from the Israel-Hamas war to the enormous electricity consumption of artificial-intelligence applications. Investing ...
- Sun, sustainability, and silicon: A double dose of solar fuel researchon May 17, 2024 at 11:17 am
The race is on to develop a new generation of liquid fuels that are activated by sunlight, and Yale researchers are helping to lead the way.
- Sun, sustainability, and silicon: A double dose of Yale solar fuel researchon May 17, 2024 at 7:15 am
In the first study, led by Wang’s lab at Yale and the lab of Tianquan Lian at Emory University, researchers constructed an electrode consisting of an array of silicon micropillars, coated with a layer ...
- Climate Change Is Making Rooftop Solar More Valuable, Research Showson May 17, 2024 at 6:00 am
In a warmer world, it will take more energy to cool people off. Rooftop solar could step in to fill the energy gap.
- Satellite Image Shows Plans For Vast New Solar Farm in USon May 17, 2024 at 4:07 am
The proposed 5,200 acre site would be Wyoming's largest solar facility, generating enough electricity to power roughly 73,000 homes.
- Free Solar for Farmers: Climate Law Gives Rich Incentiveson May 17, 2024 at 2:30 am
The law now offers a particularly sweet deal: a combination of subsidies that can cover or even exceed a project’s costs.
- Smelting Steel Without Fossil Fuels: Solar Power Shatters the 1,000°C Barrier for Industrial Heatingon May 17, 2024 at 1:14 am
Swiss researchers have developed a solar energy method using synthetic quartz to achieve temperatures above 1,000°C for industrial processes, potentially replacing fossil fuels in the production of ...
- Breakthrough solar power technology could replace fossil fuels in heavy manufacturingon May 15, 2024 at 5:04 pm
A potentially groundbreaking solar-powered device has achieved temperatures over 1,000C, raising hopes for using green energy to run some of the most fossil fuel-intensive manufacturing processes on ...
- Scientists generate heat over 1,000 degrees Celsius with solar power instead of fossil fuelon May 15, 2024 at 10:45 am
Instead of burning fossil fuels to smelt steel and cook cement, researchers in Switzerland want to use heat from the sun. The proof-of-concept study, published May 15 in the journal Device, uses ...
- Are solar panels worth it? The cost and environmental benefits of going solaron May 15, 2024 at 9:04 am
Are solar panels worth it? Find out what you can expect to pay and how much you’ll save so you can decide if solar is right for your home.
via Google News and Bing News












