A team of EPFL scientists has developed several antibody-based biosensors that have the potential to help healthcare centers in developing countries or even patients in their own homes keep track of drug concentration in the blood.
Being able to monitor drug concentration in the blood of a patient is an important aspect of any pharmaceutical treatment. However, this requires equipment and facilities that are often missing from field healthcare in developing countries, but also inhibits the quality of life for patients elsewhere. EPFL scientists, working through their startup LUCENTIX, have now developed an antibody-linked biosensor that can track drug concentration in the blood by changing its color. The biosensor is incorporated into a full system that can be used in the field or by patients at home. The science behind it is published in Angewandte Chemie.
The lab of Kai Johnsson at EPFL is known for developing biosensors, and the research gave birth to the startup LUCENTIX, which has developed a biosensor that allows patients to easily measure drug concentrations in their system without need for complex lab systems.
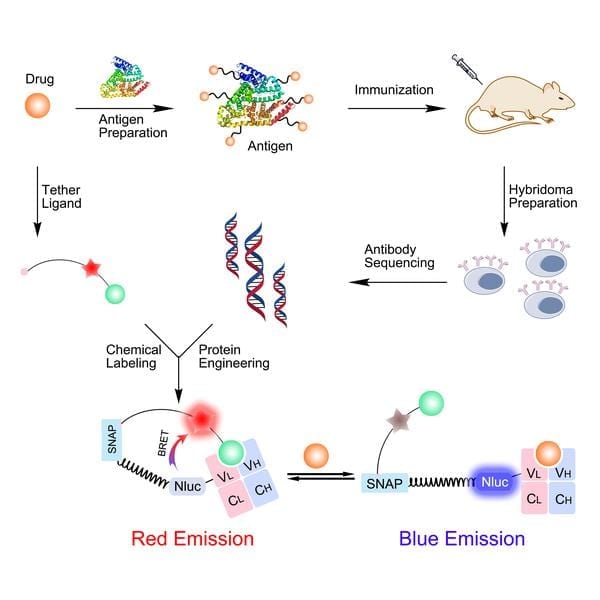
The biosensor is a molecule made up of three components: First, a protein that can bind the drug to be monitored. Second, the light-producing enzyme luciferase. And third, a “tagging” molecule called SNAP-tag, which carries a fluorescent ligand that the protein (the first component) recognizes and binds when no drug is present. This causes a reaction between the luciferase and the fluorescent molecule called “bioluminescent resonance energy transfer” (BRET), which produces a red light.
The recent innovation, carried out by postdoc Lin Xue, involves replacing the binding protein of the biosensor with part of an antibody that has been developed against the target drug. When the biosensor detects and binds the drug in the patient’s blood or saliva, the antibody “prefers” to bind this rather than the SNAP-tag’s fluorescent ligand. As the ligand is displaced, the BRET reaction is progressively disrupted, and now emits a blue light.
Antibodies are naturally able to identify and bind foreign molecules, turning our immune systems against potential infections. In addition, generating antibodies that can specifically identify small molecules like drugs is a routine procedure. This means that the monitoring system can be adapted to virtually an unlimited number of molecules, while patients can carry out the monitoring themselves at home and receive laboratory-level quality information. Current lab methods for doing this are complex and expensive, and reduce the quality of life for patients who have to often be confined within or close to hospitals.
Replacing the binding protein with an antibody establishes a general pipeline for the generation of biosensors that can identify a synthetic drug in a patient’s blood sample. As proof-of-principle, the EPFL scientists successfully tested the new biosensors against three drugs – methotrexate, theophylline, and quinine – in the lab. The next step will be to optimize the biosensor’s sensitivity so that it can accurately detect the nanomolar or even lower concentrations of drugs/biomolecules in clinical samples.
Learn more: Antibody biosensor offers unlimited point-of-care drug monitoring
[osd_subscribe categories=’drug-monitoring’ placeholder=’Email Address’ button_text=’Subscribe Now for any new posts on the topic “DRUG MONITORING’]
The Latest on: Point-of-care drug monitoring
[google_news title=”” keyword=”point-of-care drug monitoring” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- This Is a Very Weird Moment in the History of Drug Laws
If what you care about the most was a drop ... not just at some point having some transitory contact with the system. ezra klein There’s another argument that’s made in the Drug Policy Alliance ...
- Public officials seek greater oversight of prescription drug middlemen
Three publicly traded PBMs – CVS Caremark, Express Scripts and Optum RX – control about 80 percent of the U.S. PBM market, and the top six companies have over 95 percent, according to the Journal of ...
- Point of Care Diagnostics Market Report: Trends, Analysis, and $55.3 Billion Forecast by 2030
The global Point of Care Diagnostics Market witnessed significant growth in recent years, with a value of $29,478.63 million in 2020, expected to soar to $55,275.73 million by 2030, at a CAGR of 6.5% ...
- Utilization of Pharmacogenomics and Therapeutic Drug Monitoring for Opioid Pain Management
In this study, pharmacogenomics and therapeutic drug monitoring were used to evaluate ... Conclusion: These results suggest that patient care may be improved by genotyping and following ...
- Sibel Health gets Gates Foundation grant for maternal monitoring
The foundation that helped launch the Northwestern spinout for wireless monitoring of mothers and babies is chipping in another $17.5 million.
via Google News and Bing News