Photo: Melanie Gonick
Energy storage device could deliver more power than current versions of this technology.
Energy storage devices called supercapacitors have become a hot area of research, in part because they can be charged rapidly and deliver intense bursts of power. However, all supercapacitors currently use components made of carbon, which require high temperatures and harsh chemicals to produce.
Now researchers at MIT and elsewhere have for the first time developed a supercapacitor that uses no conductive carbon at all, and that could potentially produce more power than existing versions of this technology.
The team’s findings are being reported in the journal Nature Materials, in a paper by Mircea Dinc?, an MIT associate professor of chemistry; Yang Shao-Horn, the W.M. Keck Professor of Energy; and four others.
“We’ve found an entirely new class of materials for supercapacitors,” Dinc? says.
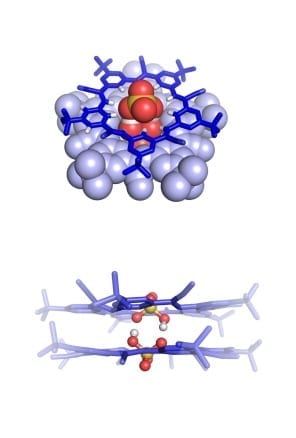
Dinc? and his team have been exploring for years a class of materials called metal-organic frameworks, or MOFs, which are extremely porous, sponge-like structures. These materials have an extraordinarily large surface area for their size, much greater than the carbon materials do. That is an essential characteristic for supercapacitors, whose performance depends on their surface area. But MOFs have a major drawback for such applications: They are not very electrically conductive, which is also an essential property for a material used in a capacitor.
“One of our long-term goals was to make these materials electrically conductive,” Dinc? says, even though doing so “was thought to be extremely difficult, if not impossible.” But the material did exhibit another needed characteristic for such electrodes, which is that it conducts ions (atoms or molecules that carry a net electric charge) very well.
“All double-layer supercapacitors today are made from carbon,” Dinc? says. “They use carbon nanotubes, graphene, activated carbon, all shapes and forms, but nothing else besides carbon. So this is the first noncarbon, electrical double-layer supercapacitor.”
One advantage of the material used in these experiments, technically known as Ni3(hexaiminotriphenylene)2, is that it can be made under much less harsh conditions than those needed for the carbon-based materials, which require very high temperatures above 800 degrees Celsius and strong reagent chemicals for pretreatment.
The team says supercapacitors, with their ability to store relatively large amounts of power, could play an important role in making renewable energy sources practical for widespread deployment. They could provide grid-scale storage that could help match usage times with generation times, for example, or be used in electric vehicles and other applications.
The new devices produced by the team, even without any optimization of their characteristics, already match or exceed the performance of existing carbon-based versions in key parameters, such as their ability to withstand large numbers of charge/discharge cycles. Tests showed they lost less than 10 percent of their performance after 10,000 cycles, which is comparable to existing commercial supercapacitors.
But that’s likely just the beginning, Dinc? says. MOFs are a large class of materials whose characteristics can be tuned to a great extent by varying their chemical structure. Work on optimizing their molecular configurations to provide the most desirable attributes for this specific application is likely to lead to variations that could outperform any existing materials. “We have a new material to work with, and we haven’t optimized it at all,” he says. “It’s completely tunable, and that’s what’s exciting.”
While there has been much research on MOFs, most of it has been directed at uses that take advantage of the materials’ record porosity, such as for storage of gases. “Our lab’s discovery of highly electrically conductive MOFs opened up a whole new category of applications,” Dinc? says. Besides the new supercapacitor uses, the conductive MOFs could be useful for making electrochromic windows, which can be darkened with the flip of a switch, and chemoresistive sensors, which could be useful for detecting trace amounts of chemicals for medical or security applications.
While the MOF material has advantages in the simplicity and potentially low cost of manufacturing, the materials used to make it are more expensive than conventional carbon-based materials, Dinc? says. “Carbon is dirt cheap. It’s hard to find anything cheaper.” But even if the material ends up being more expensive, if its performance is significantly better than that of carbon-based materials, it could find useful applications, he says.
This discovery is “very significant, from both a scientific and applications point of view,” says Alexandru Vlad, a professor of chemistry at the Catholic University of Louvain in Belgium, who was not involved in this research. He adds that “the supercapacitor field was (but will not be anymore) dominated by activated carbons,” because of their very high surface area and conductivity. But now, “here is the breakthrough provided by Dinca et al.: They could design a MOF with high surface area and high electrical conductivity, and thus completely challenge the supercapacitor value chain! There is essentially no more need of carbons for this highly demanded technology.”
And a key advantage of that, he explains, is that “this work shows only the tip of the iceberg. With carbons we know pretty much everything, and the developments over the past years were modest and slow. But the MOF used by Dinca is one of the lowest-surface-area MOFs known, and some of these materials can reach up to three times more [surface area] than carbons. The capacity would then be astonishingly high, probably close to that of batteries, but with the power performance [the ability to deliver high power output] of supercapacitors.”
Learn more: New kind of supercapacitor made without carbon
The Latest on: Supercapacitor
[google_news title=”” keyword=”supercapacitor” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Supercapacitor
- Lamborghini Countach Drags the Ferrari 812, SF90, and Hyundai Ioniq 5 N; Guess Who Won?on May 17, 2024 at 9:56 am
However, the Lamborghini’s is seconded by a tiny electric motor that ups the 769-hp naturally-aspirated output with an extra 33 hp fed from a supercapacitor. That’s 803 total horsepower sent to all ...
- IndyCar’s hybrid engine extension to debut in Julyon May 14, 2024 at 7:51 pm
A fter five years of delays and setbacks, the IndyCar Series will make the transition to hybridization at the July 5-7 race weekend at the Mid-Ohio Sports Car Course.
- IndyCar Hybrid Era Begins At Mid-Ohio In July’s Honda Indy 200on May 14, 2024 at 11:00 am
Delayed by a half-season, IndyCar officials announced on May 14 the hybrid assist unit will be on all engines beginning with the July 7 Honda Indy 200 at Mid-Ohio.
- Forget chopped liver: South Korean scientists convert chicken fat into energy storage deviceson May 14, 2024 at 12:57 am
Chicken fat is what Jews used for many years to improve their chopped liver for a Shabbat meal – until they learned that it contains a ton of cholesterol .
- Chicken fat supercapacitors could store green energy of the futureon May 13, 2024 at 8:20 am
The soot from burning off rendered chicken fat can be made into supercapacitors. Deposit Photos SHARE. As vital as it is for society to transition to renewable energy, it’s not ...
- Supercapacitor made of chicken with the smell of fried onions — scientists have turned chicken fat into an energy storage deviceon May 11, 2024 at 11:32 am
Some materials for energy storage (such as supercapacitors) can be expensive and pollute the environment. Producing alternative energy storage from th ...
- Repurposed chicken fat offers efficient green energy storage alternativeon May 11, 2024 at 2:17 am
Researchers have developed a method to turn chicken fat into carbon electrodes for supercapacitors in energy storage.
- Scientists convert chicken fat into energy storage deviceson May 10, 2024 at 9:26 am
Who knew leftover chicken fat could one day be used to store energy? Researchers have found a way to create carbon-based nanostructures from the grease, and they used it in an electrode for a ...
- Scientists design super-battery made with cheap, readily affordable chemical element, Na — Salt-based cell has surprisingly good energy density and charges in secondson April 29, 2024 at 10:06 pm
Developing the hybrid battery hinged upon improving the energy storage rate of battery-type anodes and boosting the relatively low capacity of supercapacitor-type cathode materials. The research team ...
via Bing News