Associate Professor Ki-Bum Lee has developed patent-pending technology that may overcome one of the critical barriers to harnessing the full therapeutic potential of stem cells.
One of the major challenges facing researchers interested in regenerating cells and growing new tissue to treat debilitating injuries and diseases such as Parkinson’s disease, heart disease, and spinal cord trauma, is creating an easy, effective, and non-toxic methodology to control differentiation into specific cell lineages. Lee and colleagues at Rutgers and Kyoto University in Japan have invented a platform they call NanoScript, an important breakthrough for researchers in the area of gene expression. Gene expression is the way information encoded in a gene is used to direct the assembly of a protein molecule, which is integral to the process of tissue development through stem cell therapeutics.
Stem cells hold great promise for a wide range of medical therapeutics as they have the ability to grow tissue throughout the body. In many tissues, stem cells have an almost limitless ability to divide and replenish other cells, serving as an internal repair system.
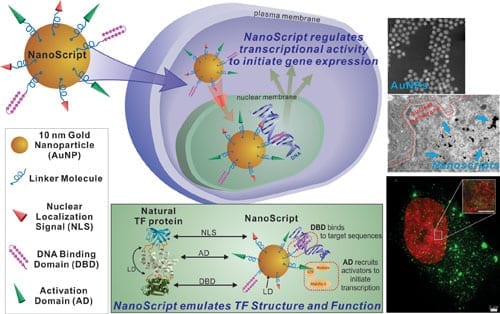
Transcription factor (TF) proteins are master regulators of gene expression. TF proteins play a pivotal role in regulating stem cell differentiation. Although some have tried to make synthetic molecules that perform the functions of natural transcription factors, NanoScript is the first nanomaterial TF protein that can interact with endogenous DNA. ACS Nano, a publication of the American Chemical Society (ACS), has published Lee’s research on NanoScript. The research is supported by a grant from the National Institutes of Health (NIH).
“Our motivation was to develop a highly robust, efficient nanoparticle-based platform that can regulate gene expression and eventually stem cell differentiation,” said Lee, who leads a Rutgers research group primarily focused on developing and integrating nanotechnology with chemical biology to modulate signaling pathways in cancer and stem cells. “Because NanoScript is a functional replica of TF proteins and a tunable gene-regulating platform, it has great potential to do exactly that. The field of stem cell biology now has another platform to regulate differentiation while the field of nanotechnology has demonstrated for the first time that we can regulate gene expression at the transcriptional level.”
NanoScript was constructed by tethering functional peptides and small molecules called synthetic transcription factors, which mimic the individual TF domains, onto gold nanoparticles.
“NanoScript localizes within the nucleus and initiates transcription of a reporter plasmid by up to 30-fold,” said Sahishnu Patel, Rutgers Chemistry graduate student and co-author of the ACS Nano publication. “NanoScript can effectively transcribe targeted genes on endogenous DNA in a nonviral manner.”
The Latest on: Stem Cell Therapeutics
[google_news title=”” keyword=”Stem Cell Therapeutics” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Stem Cell Therapeutics
- Revolutionary Leap in Biotherapy: Century Therapeutics' Ground-Breaking Technology Proves Promisingon May 12, 2024 at 12:00 am
Emerging from the recent ASGCT 27th Annual Meeting, Century Therapeutics (NASDAQ: IPSC) has caught the attention of the medical world. The company revealed its innovative Allo-Evasion™ platform, ...
- 24 hours with ‘RHONY’ alum Kelly Bensimon: Stem cell facials, smoothies and a luxe $13M real estate touron May 11, 2024 at 4:43 pm
From catching up over coffee at her favorite hotel to luxurious facials from her go-to stem cell specialist to sipping trendy smoothies, the model-turned-mogul gave us an inside look at life in ...
- Toddler born deaf can hear after gene therapy trial breakthrough her parents call "mind-blowing"on May 10, 2024 at 1:34 pm
The parents of a U.K. toddler say it's "absolutely mind-blowing" to see their daughter, enrolled in a gene therapy trial, hear for the first time.
- Century Therapeutics Presents Preclinical Data Highlighting Advances in iPSC Platform Technology and Programs at 2024 ASGCT Annual Meetingon May 10, 2024 at 5:59 am
In vitro data showcases CNTY-101’s ability to induce CD19-specific cytolysis of B-cells and potential to treat B-cell driven autoimmune diseases ...
- Deaf Baby Girl Hears for First Time in 'Mind-Blowing' Gene Therapy Trialon May 9, 2024 at 11:47 am
Hearing loss affects about 1.5 billion people, while congenital deafness—hearing loss present at birth—affects approximately 1.7 out of every 1,000 children born in the U.S. The OTOF-related deafness ...
- Fate Therapeutics Announces First Lupus Patient Treated in Phase 1 Autoimmunity Study of Off-the-shelf FT819 CAR T-cell Programon May 9, 2024 at 7:29 am
Pre-treatment Sample of Patient’s Blood Showed Rapid and Potent Depletion of CD19+ B Cells in Ex Vivo Cytotoxicity Assay with FT819Translational ...
- Lisata’s Autologous T Cell Therapy Is Safe but Fails to Slow T1D Disease Progression, Trial Findson May 9, 2024 at 6:34 am
An autologous and personalized regulatory T cell therapy is safe in patients with type 1 diabetes, but does not help preserve β-cell function.
- As Ozzy Osbourne announces stem cell therapy, experts urge caution, highlight riskson May 9, 2024 at 2:00 am
As rock legend Ozzy Osbourne has turned to stem cell therapy, some experts caution that the treatment is not for everyone. A doctor weighed in on the potential risks.
- Researchers enhance CAR-T cells to target solid tumorson May 8, 2024 at 8:54 pm
Researchers at the National Cancer Institute-designated Montefiore Einstein Comprehensive Cancer Center (MECCC) have shown that a breakthrough therapy for treating blood cancers can be adapted to ...
- Stem Cell Therapy Improves Post-Stroke Motor Functionon May 7, 2024 at 5:00 pm
The open-label, dose-escalation treatment (2.5, 5, 10, and 20 million cells) included tacrolimus immunosuppression for 8 weeks. Physical therapy was encouraged but not required. The final patient will ...
via Bing News