Stanford scientists have created a copper-based catalyst that produces large quantities of ethanol from carbon monoxide gas at room temperature.
Stanford University scientists have found a new, highly efficient way to produce liquid ethanol from carbon monoxide gas. This promising discovery could provide an eco-friendly alternative to conventional ethanol production from corn and other crops, say the scientists. Their results are published in the April 9 advanced online edition of the journal Nature.
“We have discovered the first metal catalyst that can produce appreciable amounts of ethanol from carbon monoxide at room temperature and pressure – a notoriously difficult electrochemical reaction,” said Matthew Kanan, an assistant professor of chemistry at Stanford and coauthor of the Naturestudy.
Most ethanol today is produced at high-temperature fermentation facilities that chemically convert corn, sugarcane and other plants into liquid fuel. But growing crops for biofuel requires thousands of acres of land and vast quantities of fertilizer and water. In some parts of the United States, it takes more than 800 gallons of water to grow a bushel of corn, which, in turn, yields about 3 gallons of ethanol.
The new technique developed by Kanan and Stanford graduate student Christina Li requires no fermentation and, if scaled up, could help address many of the land- and water-use issues surrounding ethanol production today. “Our study demonstrates the feasibility of making ethanol by electrocatalysis,” Kanan said. “But we have a lot more work to do to make a device that is practical.”
Novel electrodes
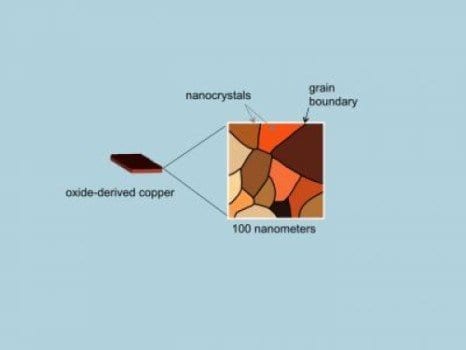
Two years ago, Kanan and Li created a novel electrode made of a material they called oxide-derived copper. They used the term “oxide-derived” because the metallic electrode was produced from copper oxide.
“Conventional copper electrodes consist of individual nanoparticles that just sit on top of each other,” Kanan said. “Oxide-derived copper, on the other hand, is made of copper nanocrystals that are all linked together in a continuous network with well-defined grain boundaries. The process of transforming copper oxide into metallic copper creates the network of nanocrystals.”
For the Nature study, Kanan and Li built an electrochemical cell – a device consisting of two electrodes placed in water saturated with carbon monoxide gas. When a voltage is applied across the electrodes of a conventional cell, a current flows and water is converted to oxygen gas at one electrode (the anode) and hydrogen gas at the other electrode (the cathode). The challenge was to find a cathode that would reduce carbon monoxide to ethanol instead of reducing water to hydrogen.
“Most materials are incapable of reducing carbon monoxide and exclusively react with water,” Kanan said. “Copper is the only exception, but conventional copper is very inefficient.”
In the Nature experiment, Kanan and Li used a cathode made of oxide-derived copper. When a small voltage was applied, the results were dramatic.
“The oxide-derived copper produced ethanol and acetate with 57 percent faradaic efficiency,” Kanan said. “That means 57 percent of the electric current went into producing these two compounds from carbon monoxide. We’re excited because this represents a more than 10-fold increase in efficiency over conventional copper catalysts. Our models suggest that the nanocrystalline network in the oxide-derived copper was critical for achieving these results.”
Carbon neutral
The Stanford team has begun looking for ways to create other fuels and improve the overall efficiency of the process. “In this experiment, ethanol was the major product,” Kanan said. “Propanol would actually be a higher energy-density fuel than ethanol, but right now there is no efficient way to produce it.”
In the experiment, Kanan and Li found that a slightly altered oxide-derived copper catalyst produced propanol with 10 percent efficiency. The team is working to improve the yield for propanol by further tuning the catalyst’s structure.
Ultimately, Kanan would like to see a scaled-up version of the catalytic cell powered by electricity from the sun, wind or other renewable resource.
For the process to be carbon neutral, scientists will have to find a new way to make carbon monoxide from renewable energy instead of fossil fuel, the primary source today. Kanan envisions taking carbon dioxide (CO2) from the atmosphere to produce carbon monoxide, which, in turn, would be fed to a copper catalyst to make liquid fuel. The CO2 that is released into the atmosphere during fuel combustion would be re-used to make more carbon monoxide and more fuel – a closed-loop, emissions-free process.
“Technology already exists for converting CO2 to carbon monoxide, but the missing piece was the efficient conversion of carbon monoxide to a useful fuel that’s liquid, easy to store and nontoxic,” Kanan said. “Prior to our study, there was a sense that no catalyst could efficiently reduce carbon monoxide to a liquid. We have a solution to this problem that’s made of copper, which is cheap and abundant. We hope our results inspire other people to work on our system or develop a new catalyst that converts carbon monoxide to fuel.”
The Latest on: Ethanol production
[google_news title=”” keyword=”Ethanol production” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Ethanol production
- USDA Raises Ethanol Industry Corn Use Forecast -- OPISon May 10, 2024 at 11:32 am
U.S. ethanol plants will use more corn than had been projected earlier in the current marketing year and maintain that level in the next marketing ...
- Harnessing breadfruit starch for bioethanol productionon May 10, 2024 at 11:00 am
In a bid to address the growing demand for renewable energy, a team of scientists has turned to an unlikely source—the humble breadfruit. A recent study published in the Journal of Bioresources and ...
- One-step biomass-to-ethanol conversion tech advanceson May 10, 2024 at 6:15 am
New Hampshire-based startup Terragia Biofuel has secured funding to proceed with commercializing its comparatively low-cost technology for the conversion of cellulosic biomass into ethanol and other ...
- ISMA seeks policy support to meet India's 20% ethanol blending targeton May 10, 2024 at 1:13 am
The drop in Indian cane production due to the global climate phenomenon El Niño led to a halt in ethanol production from syrup and BHM from mid-December 2023.
- ISMA moots strategic policy interventions to meet 20% ethanol blending target by 2030on May 9, 2024 at 8:57 am
To address the challenges faced by the sugar industry and meet the 20 per cent ethanol blending target by 2030, the Indian Sugar and Bio-Energy Manufacturers Association (ISMA) has stressed the need ...
- Dextrose… with a 40% lower carbon footprint? Green Plains gears up to launch ‘truly disruptive’ tech at ethanol plant in Iowaon May 8, 2024 at 5:56 pm
Green Plains will be able to produce dextrose with significantly lower capital and carbon intensity vs the standard wet milling process.
- Ethanol supply gets tighter, production downon May 8, 2024 at 3:56 pm
U.S. ethanol stocks have fallen for five weeks in a row. The U.S. Energy Information Administration says the domestic ethanol supply dropped to an 18-week low at 24.2 million barrels, a decrease of 1.
- Can corn ethanol really help decarbonize US air travel?on May 5, 2024 at 8:30 pm
The White House has set a goal of increasing U.S. production of SAF to at least 3 billion gallons ... And, most likely, producers will turn to using ethanol — a biofuel that’s most commonly made from ...
- Ethanol Market To Reach USD 142.02 Billion By 2031 Driven By Surging Demand For Sustainable Fuel Alternativeson May 5, 2024 at 6:11 am
( MENAFN - Evertise Digital) - The SNS Insider report sheds light on the promising future of the Ethanol Market . The report estimates that the market size, valued at USD 91.15 Billion in 2023, ...
- March Corn-For-Ethanol Use Rises More Than Expectedon May 1, 2024 at 5:30 pm
Corn-for-ethanol use totaled 468.8 million bu. during March, up 23.3 million bu. (5.1%) from February and 32.7 million bu. (7.5%) more than last year. Traders expected corn ethanol use of 464.5 ...
via Bing News








![[Right: Artist's concept of nanometer-size metallic wires and metallic particles embedded in semiconductors, as grown by Dr. Hong Lu. Credit: Peter Allen, UCSB]](https://innovationtoronto.com/wp-content/uploads/2014/04/gossard-lu-nanowires-900.jpg)