Credit: Salk Institute
The Salk approach, tested in a mouse model, points to cheaper and more effective treatments for humans
For most people with hemophilia B, whose bodies can’t properly form blood clots, constant injections to replenish their clotting factors are a way of life. But now, Salk researchers have demonstrated in mice that hemophilia B can be treated for life with one single injection containing disease-free liver cells that can produce their missing clotting factor. The finding, published in the journal Cell Reports on May 1, 2018, could drastically change what it means to be diagnosed with hemophilia B, and could pave the way toward similar treatments for other, related genetic disorders.
Hemophilia B is caused by defects in the gene for a protein called clotting factor IX (FIX). Hemophiliacs may make reduced amounts of the protein, or lack a functional version altogether, leading to life-threatening delays in blood clotting. Currently, patients are treated with injections—as often as a few times a week—containing FIX made in animal cells and then purified. But the approach is expensive, time-consuming and can become less effective over time.
Recently, Salk scientists developed a new approach, treating mice genetically engineered to have hemophilia B with strands of messenger RNA encoding the FIX gene. Like the standard treatment, however, this required repeat injections each time levels of the messenger RNA ran low. So the scientists wanted to try a more permanent approach: transplanting healthy liver cells, capable of producing FIX, into patients.
“The appeal of a cell-based approach is that you minimize the number of treatments that a patient needs,” says Suvasini Ramaswamy, a former Salk research associate in the Verma lab and first author of the new paper. “Rather than constant injections, you can do this in one shot.”
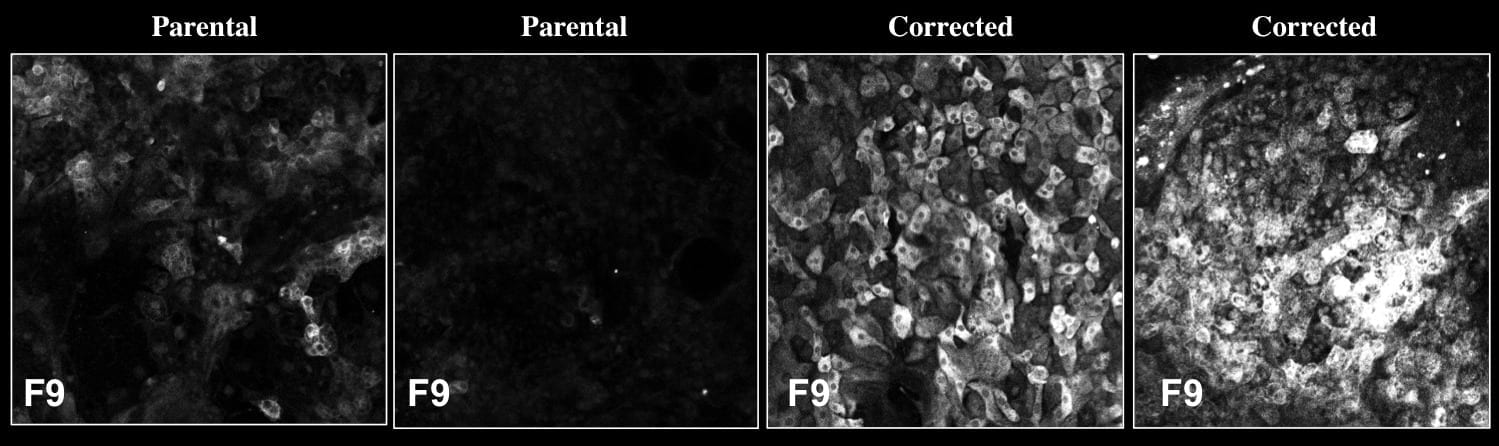
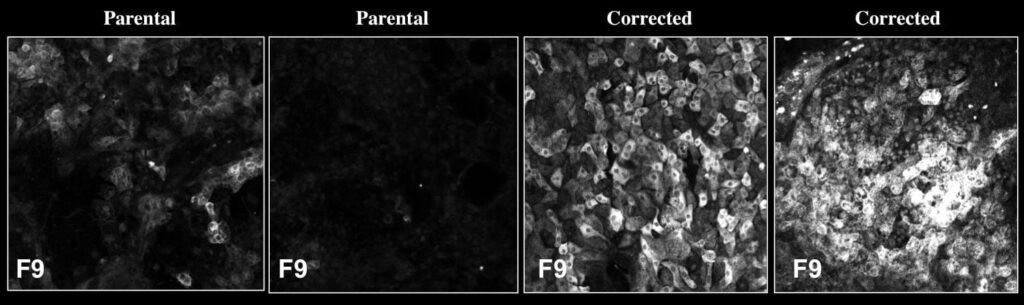
Since donor livers are often in short-supply, the researchers instead turned to stem-cell strategies to produce the healthy liver cells. They collected blood samples from two human patients with severe hemophilia B, who are unable to produce FIX. Then, in the lab, they reprogrammed the cells into induced pluripotent stem cells (iPSCs), which have the capability to turn into many other cell types, including liver. Using CRISPR/Cas9, a tool that can alter genes, they then repaired the mutations in each patient’s FIX gene. Finally, they coaxed those repaired cells to develop into liver precursor cells called hepatocyte-like cells (HLCs) and transplanted them into mice with hemophilia B.
Rather than perform surgery on hemophilic mice—a risky undertaking when their blood can’t always clot—the team transplanted the HLCs through the spleen to distribute the cells uniformly in the liver.
Not only did the new HLCs produce FIX, but they produced enough of the protein to allow the mice to form normal blood clots, and the cells continued to survive—and produce FIX—for at least a year after the transplantation.
In people with hemophilia, using their own cells to generate healthy HLCs, then transplanting them back into their bodies, could help avoid the immune complications that often accompany cell therapies. But more work is needed to translate the findings to the clinic.
“A lot of things have to happen before this can go into humans,” says Ramaswamy.
Already, she adds, the work demonstrates the value in combining stem-cell reprogramming and new gene-modifying approaches to treat genetic diseases.
Learn more: Single injection treats hemophilia B for life, in proof-of-concept study
The Latest on: Hemophilia B
[google_news title=”” keyword=”hemophilia B” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Hemophilia B
- Pfizer heralds “paradigm shift” in hemophilia B gene therapy with FDA approvalon April 30, 2024 at 7:43 am
Pfizer's groundbreaking $3.5 million gene therapy for Hemophilia B just got FDA approval. Here’s what it means for patients and the future of medicine ...
- Labcorp nets companion diagnostic approval for Pfizer's hemophilia B gene therapy Beqvezon April 29, 2024 at 10:56 am
The FDA approved its first gene therapy companion diagnostic test last June, developed through a collaboration between ARUP Laboatories and BioMarin, maker of the hemophilia A treatment Roctavian ...
- Pfizer wins approval for $3.5m hemophilia B gene therapyon April 29, 2024 at 3:21 am
The gene therapy is the third such product approved by the US Food and Drug Administration (FDA), following the November 2022 approval of CSL Behring’s Hemgenix (etranacogene dezaparvovec) – also for ...
- FDA Approves Companion Diagnostic for Hemophilia B Gene Therapy Beqvezon April 28, 2024 at 5:00 pm
Beqvez is indicated for the treatment of adults with moderate to severe hemophilia B who currently use factor IX prophylaxis therapy, or have current or historical life-threatening hemorrhage, or have ...
- FDA Approves Pfizer’s One-Time Gene Therapy BEQVEZ for Hemophilia Bon April 28, 2024 at 2:23 pm
The BEQVEZ gene therapy inserts a working copy of the Factor IX (FIX) gene that codes for a high-activity FIX variant.
- US Approves Gene Therapy Treatment For Hemophiliaon April 26, 2024 at 11:04 am
Pharmaceutical giant Pfizer has received US approval for a gene therapy against a form of hemophilia, a rare and inherited blood clotting disorder, the company said Friday.
- U.S. FDA Approves Pfizer’s BEQVEZ™ (fidanacogene elaparvovec-dzkt), a One-Time Gene Therapy for Adults with Hemophilia Bon April 26, 2024 at 7:45 am
Pfizer Inc. (NYSE: PFE) announced today that the U.S. Food and Drug Administration (FDA) has approved BEQVEZ™ (fidanacogene elaparvovec-dzkt) for the ...
- FDA Approves Second Gene Therapy for Hemophilia Bon April 26, 2024 at 7:38 am
Pfizer reported a list price of $3.5 million for its new gene therapy, Beqvez, for adults with this rare bleeding disorder.
- Pfizer scores FDA nod for hemophilia B gene therapy, will charge $3.5M per doseon April 26, 2024 at 7:19 am
Ten years after dipping its toe into the gene therapy pool in a licensing deal with Spark Therapeutics, Pfizer has gained an FDA approval for the acquired treatment. | Ten years after dipping its toe ...
via Bing News