A team of researchers has developed an ultrasound-based system that can non-invasively and remotely control genetic processes in live immune T cells so that they recognize and kill cancer cells.
There is a critical need to non-invasively and remotely manipulate cells at a distance, particularly for translational applications in animals and humans, researchers said.
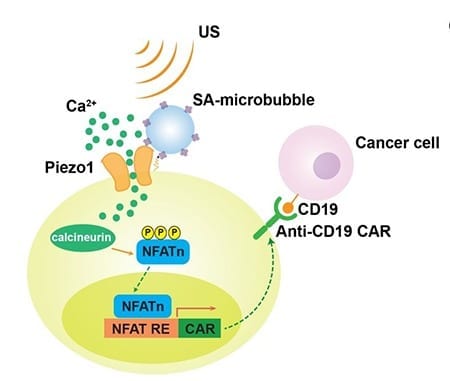
The team developed an innovative approach to use mechanogenetics—a field of science that focuses on how physical forces and changes in the mechanical properties of cells and tissues influence gene expression—for the remote control of gene and cell activations. Researchers used ultrasound to mechanically perturb T cells, and then converted the mechanical signals into genetic control of cells.
In this study, researchers show how their remote-controlled mechanogenetics system can be used to engineer chimeric antigen receptor (CAR)-expressing T cells that can target and kill cancer cells. The engineered CAR-T cells have mechano-sensors and genetic transducing modules that can be remotely activated by ultrasound via microbubble amplification.
“CAR-T cell therapy is becoming a paradigm-shifting therapeutic approach for cancer treatment,” said bioengineering professor Peter Yingxiao Wang at the University of California San Diego. “However, major challenges remain before CAR-based immunotherapy can become widely adopted. For instance, the non-specific targeting of CAR-T cells against nonmalignant tissues can be life-threatening. This work could ultimately lead to an unprecedented precision and efficiency in CAR-T cell immunotherapy against solid tumors, while minimizing off-tumor toxicities.”
The team brings together the laboratories of professors Wang and Shu Chien, both bioengineering professors at the Jacobs School of Engineering and the Institute of Engineering in Medicine at UC San Diego, in collaboration with professors Kirk Shung of the University of Southern California and Michel Sadelain at Memorial Sloan Kettering Cancer Center in New York. Researchers present their findings in the Jan. 15 issue of the Proceedings of the National Academy of Sciences, with UC San Diego Ph.D. candidate Yijia Pan as the first author.
Researchers found that microbubbles conjugated to streptavidin can be coupled to the surface of a cell, where mechanosensitive Piezo1 ion channels are expressed. Upon exposure to ultrasound waves, microbubbles vibrate and mechanically stimulate Piezo1 ion channels to let calcium ions inside the cell. This triggers downstream pathways, including calcineurin activation, NFAT dephoshorylation and translocation into the nucleus. The nucleus-translocated NFAT can bind to upstream response elements of genetic transducing modules to initiate gene expression of chimeric antigen receptor (CAR) for the recognition and killing of target cancer cells.
Learn more: Researchers Develop a Remote-Controlled Cancer Immunotherapy System
The Latest on: CAR-T cell therapy
[google_news title=”” keyword=”CAR-T cell therapy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]- First FDA-approved cellular therapy for metastatic melanoma available in South Floridaon May 1, 2024 at 9:04 pm
Patients in South Florida with metastatic melanoma will soon have access to the first cellular therapy for this advanced form of skin cancer, following its recent approval by the Food and Drug ...
- Cell Therapy Manufacturing: Time to Move Faston April 30, 2024 at 9:54 am
The more quickly manufacturing and release obstructions can be cleared, the sooner lifesaving treatments can be administered.
- T-Cell Exhaustion in CLL: Allogeneic CAR T Trial Reaching Patients With Unmet Needon April 29, 2024 at 5:59 pm
"Off the shelf" CAR T-cell therapies could offer a solution in chronic lymphocytic leukemia, where T-cell exhaustion creates treatment challenges. This article will appear in the May 2024 issue of ...
- Do cutting-edge CAR-T-cell therapies cause cancer? What the data sayon April 29, 2024 at 5:00 pm
Regulators have identified around 30 cases of cancer linked to this blockbuster treatment. But is CAR T to blame? The hunt is on for answers.
- CAR T-Cell Therapy Market to Reach Valuation of USD 22.2 Billion By 2032: Astute Analyticaon April 29, 2024 at 7:30 am
Soaring blood cancer rates fuel the CAR-T cell therapy market, a personalized weapon against aggressive cancers. Despite high costs, research breakthroughs and growing approvals promise broader access ...
- Cell and gene therapy companies trip at scalability hurdleon April 29, 2024 at 3:09 am
Experts hold scalability challenges and high costs accountable for market failures within the cell and gene therapy landscape.
- Biomarkers Linked to Cell Therapy Response for Bone Marrow Tumors Identifiedon April 29, 2024 at 2:09 am
In a recent clinical study, researchers identified several biomarkers that are associated with the response to CAR T cell therapy in multiple myeloma, a malignant tumour disease in the bone marrow.
- Biomarkers identified for successful treatment of bone marrow tumorson April 26, 2024 at 8:09 am
CAR T cell therapy has proven effective in treating various hematological cancers. However, not all patients respond equally well to treatment. In a clinical study, researchers from the University of ...
- CD7 CAR T-Cell Therapy, Stem-Cell Transplant Beneficial for CD7-Positive Tumorson April 25, 2024 at 11:30 pm
For patients with relapsed or refractory CD7-positive leukemia or lymphoma, sequential CD7 chimeric antigen receptor (CAR) T-cell therapy followed by haploidentical hematopoietic stem-cell ...
- New Therapy Destroys Deadly Brain Tumor in Dayson April 25, 2024 at 6:30 am
A groundbreaking new therapy destroys deadly brain tumors within days. The new treatment, CAR T-cell therapy, helps treat glioblastoma.
via Google News and Bing News