Stanford scientists have developed inexpensive silicon-based electrodes that dramatically improve the charge storage capacity of lithium-ion batteries.
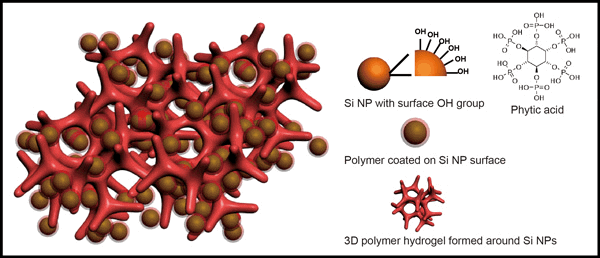
Stanford University scientists have dramatically improved the performance of lithium-ion batteries by creating novel electrodes made of silicon and conducting polymer hydrogel, a spongy substance similar to the material used in soft contact lenses and other household products.
Writing in the June 4 edition of the journal Nature Communications, the scientists describe a new technique for producing low-cost, silicon-based batteries with potential applications for a wide range of electrical devices.
“Developing rechargeable lithium-ion batteries with high energy density and long cycle life is of critical importance to address the ever-increasing energy storage needs for portable electronics, electric vehicles and other technologies,” said study co-author Zhenan Bao, professor of chemical engineering at Stanford.
To find a practical, inexpensive material that increases the storage capacity of lithium-ion batteries, Bao and her Stanford colleagues turned to silicon – an abundant, environmentally benign element with promising electronic properties.
“We’ve been trying to develop silicon-based electrodes for high-capacity lithium-ion batteries for several years,” said study co-author Yi Cui, associate professor of materials science and engineering at Stanford. “Silicon has 10 times the charge storage capacity of carbon, the conventional material used in lithium-ion electrodes. The problem is that silicon expands and breaks.”
Studies have shown that silicon particles can undergo a 400 percent volume expansion when combined with lithium. When the battery is charged or discharged, the bloated particles tend to fracture and lose electrical contact. To overcome these technical constraints, the Stanford team used a fabrication technique called in situ synthesis polymerization that coats the silicon nanoparticles within the conducting hydrogel.
A new way
This technique allowed the scientists to create a stable lithium-ion battery that retained a high storage capacity through 5,000 cycles of charging and discharging. “We attribute the exceptional electrochemical stability of the battery to the unique nanoscale architecture of the silicon-composite electrode,” Bao said.
Using a scanning electron microscope, the scientists discovered that the porous hydrogel matrix is riddled with empty spaces that allow the silicon nanoparticles to expand when lithium is inserted. This matrix also forms a three-dimensional network that creates an electronically conducting pathway during charging and discharging.
“It turns out that hydrogel has binding sites that latch onto silicon particles really well and at the same time provide channels for the fast transport of electrons and lithium ions,” explained Cui, a principal investigator with the Stanford Institute for Materials and Energy Sciences at the SLAC National Accelerator Laboratory. “That makes a very powerful combination.”
A simple mixture of hydrogel and silicon proved far less effective than the in situ synthesis polymerization technique. “Making the hydrogel first and then mixing it with the silicon particles did not work well,” Bao said. “It required an additional step that actually reduced the battery’s performance. With our technique, each silicon nanoparticle is encapsulated within a conductive polymer surface coating and is connected to the hydrogel framework. That improves the battery’s overall stability.”
Addressing the fire issue
Hydrogel primarily consists of water, which can cause lithium-ion batteries to ignite – a potential problem that the research team had to address. “We utilized the three-dimensional network property of the hydrogel in the electrode, but in the final production phase, the water was removed,” Bao said. “You don’t want water inside a lithium-ion battery.”
Although a number of technical issues remain, Cui is optimistic about potential commercial applications of the new technique to create electrodes made of silicon and other materials.
The Latest Bing News on:
Lithium-ion battery performance
- Breakthrough in Sodium Battery Chemistry Promises Lower Costson April 27, 2024 at 12:00 pm
A new mass synthesis process for sodium-containing sulfides could make all-solid-state sodium batteries more affordable and safer than lithium-ion batteries.
- Lithium Miners News For The Month Of April 2024on April 26, 2024 at 10:56 am
Welcome to the April 2024 edition of the lithium miner news. The past month saw lithium prices flat and a very busy month of good news for the lithium producers. We also saw the start of the Beijing ...
- Novel Anode Material Boosts Lithium-Ion Battery Performanceon April 25, 2024 at 9:16 am
Dr. Han Joong Tark and student researcher Lee Do Geun at Korea Electrotechnology Research Institute's Nano Hybrid Technology Research Center have devised a novel manufacturing method for ...
- Sodium-ion battery could charge in several secondson April 25, 2024 at 2:18 am
Researchers at the Korea Advanced Institute of Science and Technology (KAIST) have identified a high-energy, high-power hybrid sodium-ion battery capable of charging in just a few seconds. The system ...
- Asahi Kasei to Construct a Lithium-ion Battery Separator Plant in Canadaon April 24, 2024 at 5:00 pm
Asahi Kasei plans to construct an integrated plant in Ontario, Canada for base film manufacturing and coating of Hipore™ wet-process LIB separator.
- Single-walled carbon nanotubes doped with 'nitrogen' enhance the performance of secondary battery anodeon April 24, 2024 at 9:27 am
Researchers have developed a new manufacturing technique for "silicon/nitrogen-doped carbon composite anode materials." These materials aim to enhance the capacity and stability of lithium-ion battery ...
- How Clemson University research could affect your access to lithium ion batterieson April 24, 2024 at 4:06 am
While the availability of lithium iode batteries is expected to wane, researchers are looking to extend that supply by ...
- A Cheaper and More Sustainable Lithium Battery: How LiDFOB Could Change Everythingon April 23, 2024 at 10:19 pm
Scientists have developed an ultralow-concentration electrolyte for lithium-ion batteries. Lithium salts enhance the power of batteries but also increase their cost. A new electrolyte with a very low ...
- Charging ahead: The quest for clean energy and safer battery storageon April 22, 2024 at 10:33 am
This Earth Day, we looked at researchers pursuing safer alternatives to facilitate the implementation of energy storage solutions.
- An ultralow-concentration electrolyte for lithium-ion batterieson April 22, 2024 at 6:05 am
Lithium salts make batteries powerful but expensive. An ultralow-concentration electrolyte based on the lithium salt LiDFOB may be a more economical and more sustainable alternative. Cells using these ...
The Latest Google Headlines on:
Lithium-ion battery performance
[google_news title=”” keyword=”lithium-ion battery performance” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
The Latest Bing News on:
Lithium-ion batteries
- Breakthrough in Sodium Battery Chemistry Promises Lower Costson April 27, 2024 at 12:00 pm
A new mass synthesis process for sodium-containing sulfides could make all-solid-state sodium batteries more affordable and safer than lithium-ion batteries.
- Why are lithium ion batteries a hazard? What to know after Columbus LIB fire spurs evacuationon April 19, 2024 at 3:28 am
A Columbus tractor trailer fire caused by lithium batteries on Thursday has us asking: Why do lithium batteries explode? And can you recycle them?
The Latest Google Headlines on:
Lithium-ion batteries
[google_news title=”” keyword=”lithium-ion batteries” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]