Sometimes it only takes a quick jolt of electricity to get a swarm of cells moving in the right direction.
Researchers at UC Berkeley found that an electrical current can be used to orchestrate the flow of a group of cells, an achievement that could establish the basis for more controlled forms of tissue engineering and for potential applications such as “smart bandages” that use electrical stimulation to help heal wounds.
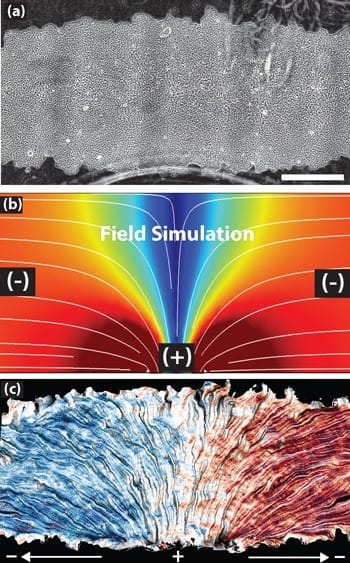
In the experiments, described in a study published this week in the journal Nature Materials, the researchers used single layers of epithelial cells, the type of cells that bind together to form robust sheathes in skin, kidneys, cornea and other organs. They found that by applying an electric current of about five volts per centimeter, they could encourage cells to migrate along the direct current electric field.
They were able to make the cells swarm left or right, to diverge or converge and to make collective U-turns. They also created elaborate shapes, such as a triceratops and the UC Berkeley Cal bear mascot, to explore how the population and configuration of cell sheets affect migration.
Directing herds vs. individuals
“This is the first data showing that direct current fields can be used to deliberately guide migration of a sheet of epithelial cells,” said study lead author Daniel Cohen, who did this work as a student in the UC Berkeley-UC San Francisco Joint Graduate Program in Bioengineering. “There are many natural systems whose properties and behaviors arise from interactions across large numbers of individual parts – sand dunes, flocks of birds, schools of fish, and even the cells in our tissues. Just as a few sheepdogs exert enormous control over the herding behavior of sheep, we might be able to similarly herd biological cells for tissue engineering.”
Galvanotaxis – the use of electricity to direct cell movement – had been previously demonstrated for individual cells, but how it influences the collective motion of cells was still unclear.
“The ability to govern the movement of a mass of cells has great utility as a scientific tool in tissue engineering,” said study senior author Michel Maharbiz, UC Berkeley associate professor of electrical engineering and computer sciences. “Instead of manipulating one cell at a time, we could develop a few simple design rules that would provide a global cue to control a collection of cells.”
The work was borne from a project, led by Maharbiz, to develop electronic nanomaterials for medical use that was funded by the National Science Foundation’s Emerging Frontiers in Research and Innovation program. The researchers collaborated with W. James Nelson, professor of molecular and cellular physiology at Stanford University and one of the world’s top experts in cell-to-cell adhesion. Cohen is now a postdoctoral research fellow in Nelson’s lab.
Possible wound healing applications
With our bodies full of flowing ions and salt solutions, it is no surprise that electrical signals play a big role in our physiology, from neural transmissions to muscle stimulation.
“The electrical phenomenon we are exploring is distinct in that the current produced is providing a cue for cells to migrate,” said Maharbiz.
The study authors are exploring the role of bioelectrical signals in the wound healing process, building upon the discovery in 1843 that an injury to the body creates a change in the electrical field at the wound site. By mapping the changes in the electrical field when an injury occurs and as it heals, the researchers may be able to develop technology to help speed and improve the repair process.
“These data clearly demonstrate that the kind of cellular control we would need for a smart bandage might be possible, and the next part of our work will focus on adapting this technology for use in actual injuries,” said Cohen.
The Latest on: Tissue engineering
[google_news title=”” keyword=”Tissue engineering” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Tissue engineering
- Two UTA faculty honored for outstanding researchon April 30, 2024 at 11:50 am
The University of Texas at Arlington is honoring two faculty for their outstanding contributions to research.
- New experimental evidence unlocks a puzzle in vascular tissue engineeringon April 25, 2024 at 9:25 am
Angiogenesis is a process of forming hierarchical vascular networks in living tissues. Its complexity makes the controlled generation of blood vessels in laboratory conditions a highly challenging ...
- Mini Colons Mimic Healthy, Tumor Tissue In Colorectal Cancer Developmenton April 25, 2024 at 12:01 am
The mini colons replicate both the physical structure and cellular diversity in the colon in both healthy and diseased states.
- Tissue Engineering and Regeneration Market [2028]: Navigating Opportunities and Challengeson April 24, 2024 at 3:11 am
USD 14.57 billion in 2022 and is poised for substantial growth, projecting a Compound Annual Growth Rate (CAGR) of 8.35% through 2028.
- Tissue Engineering News and Researchon April 23, 2024 at 5:00 pm
The field of bone tissue engineering (BTE) was a promising avenue for addressing bone injuries and defects by constructing artificial scaffolds with bionic functionalities. The promising results ...
- Will Heart Valve Tissue Engineering Change the World?on April 14, 2024 at 5:00 pm
Nat Clin Pract Cardiovasc Med. 2005;2(2):60-61. Several strategies for heart valve tissue engineering are evolving. They include the use of decellularized allogenic or xenogenic valve matrices ...
- Optimizing tissue engineering in rotator cuff repairon March 18, 2024 at 10:00 am
Advances in tissue engineering have demonstrated that mesenchymal stem cells (MSCs) and growth factors (GFs) enhance the regeneration of rotator cuff tendon-to-bone insertion in animal models ...
- Advancing tissue engineering with shape memory hydrogelson March 13, 2024 at 7:55 am
On seeding the hydrogel with mouse embryo fibroblast (NIH/3T3) cells, the researchers observed a direct correlation between the elastic modulus of the hydrogel and the number of adhered cells. In ...
- Cardiovascular Reparative Medicine and Tissue Engineeringon February 20, 2024 at 2:49 pm
Cardiovascular Reparative Medicine and Tissue Engineering (CRMTE) aims to develop future technologies and therapeutic strategies that will serve as treatment for cardiovascular disease. CRMTE includes ...
- TissueFab® Sacrificial bioink for tissue engineering applicationson November 14, 2023 at 4:00 am
Polaxomer is a thermos-responsive hydrogel that has been employed in bioprinting and tissue engineering as a mold, track patterning, and sacrificial material. Due to the nature of micellar-packing ...
via Bing News










