Scientists at Trinity College, Dublin say they have made a major breakthrough in treatment for age-related macular degeneration (AMD) – the leading cause of blindness in people over 50.
Through their research, they have discovered that interleukin-18 (IL-18), a component of the immune system, can protect patients from vision loss, and it can be administered in a non-invasive way. Since current treatments for AMD involve periodically injecting medication directly into the eyeball, this could have significant implications for AMD therapy.
“[IL-18] has already entered into clinical trials [for cancer research], and it has a very good safety profile when administered intravenously. It was never injected into the eye,” study author Matthew Campbell, research assistant professor in genetics at Trinity, told FoxNews.com.
“So all the signs show this could be another tool in the arsenal against wet AMD.”
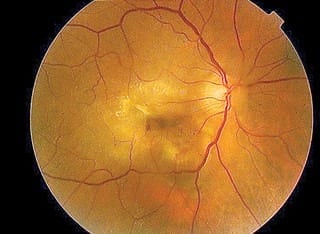
AMD has two major forms: “dry,” the more common form, and “wet,” which accounts for 90 percent of the severe vision loss caused by macular degeneration. When a patient suffers from wet AMD, the blood vessels grow into the retina, the layer of tissue lining the inner surface of the eye, and begin to leak blood or fluid – causing immediate retinal blindness. Retinal blindness can be especially challenging for patients, as the retina provides sharp, central vision for reading, driving and perceiving small details.
In order to combat wet AMD, patients m? patients must receive them on a monthly basis.
Campbell and his team say IL-18 can work just as effectively as these anti-VEGF injections, as it produces cells that prevent wet AMD.
The Latest on: Age-related macular degeneration
[google_news title=”” keyword=”Age-related macular degeneration” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Age-related macular degeneration
- Nanoscope Therapeutics Appoints New Chief Medical Officeron April 26, 2024 at 3:38 pm
Dallas-based Nanoscope said Dr. Allen C. Ho will help define the strategy of developing its gene mutation-agnostic therapies. Ho is attending surgeon and director of retina research at Wills Eye ...
- Science Corporation acquires retinal implant from Pixium Visionon April 26, 2024 at 6:39 am
S cience Corporation, a brain-computer interface technology company, has announced the acquisition of intellectual property and related assets for the Prima retinal implant from French bioelectronics ...
- Local group helps macular degeneration patients cope with vision losson April 26, 2024 at 3:07 am
To provide opportunity for encouragement, Phoenix-based Associated Retina Consultants, in coordination with the International Low Vision Support Group, has established a Macular Degeneration Support ...
- BCI maker Science acquires retinal implant from Pixium Visionon April 25, 2024 at 12:54 pm
Brain-computer interface developer Science Corporation acquired the IP and related assets for the Pixium Vision Prima implant.
- To Your Good Health: Over-the-counter eye drops safe to use with AMD medicationon April 25, 2024 at 7:15 am
I am a healthy 67-year-old female being treated with Eylea for wet age-related macular degeneration (AMD). I have just had my second round of Eyelea, and with ...
- People over State Pension age with eyesight issues could be due up to £434 each monthon April 24, 2024 at 11:32 pm
Attendance Allowance could help people who are severely short-sighted or have eye conditions including cataract, glaucoma or macular degeneration.
- BrightFocus Foundation Announces $10M in New Funding Across Brain and Vision Research, Celebrates Historic Diversity of Grant Award Recipientson April 24, 2024 at 11:00 pm
Global nonprofit BrightFocus Foundation announces $10 million in new Alzheimer’s disease, macular degeneration, and glaucoma research grant funding—age-related diseases with no cure that affect more ...
- High dose aflibercept may be promising for treatment of age-related macular degenerationon April 24, 2024 at 2:55 pm
Aflibercept 8 mg was non-inferior to 2 mg for best-corrected visual acuity at 48 weeks. 2. Ocular adverse events were comparable across treatment groups. Evidence Rating Level: 1 (Excellent) Study ...
- AMD STOPPER Offers New Hope to Sufferers of Age-Related Macular Degenerationon April 23, 2024 at 9:50 am
AMD Stopper Inc. is a newly organized company which holds the formulas, patent applications, and intellectual property rights to the formula developed by an eye care researcher to treat Age-Related ...
- Dry Age-related Macular Degeneration Seven Major Market to Exhibit Growth at a CAGR of 19.8% by 2034 | DelveInsighton April 22, 2024 at 2:31 pm
DelveInsight's Dry Age-related Macular Degeneration Market Insights report includes a comprehensive understanding of current treatment practices, dry age-related macular degeneration marketed drug, ...
via Bing News