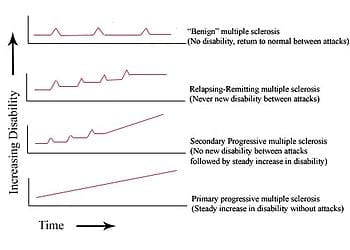
Scientists at The New York Stem Cell Foundation (NYSCF) Research Institute are one step closer to creating a viable cell replacement therapy for multiple sclerosis from a patient’s own cells.
For the first time, NYSCF scientists generated induced pluripotent stem (iPS) cells lines from skin samples of patients with primary progressive multiple sclerosis and further, they developed an accelerated protocol to induce these stem cells into becoming oligodendrocytes, the myelin-forming cells of the central nervous system implicated in multiple sclerosis and many other diseases.
Existing protocols for producing oligodendrocytes had taken almost half a year to produce, limiting the ability of researchers to conduct their research. This study has cut that time approximately in half, making the ability to utilize these cells in research much more feasible.
Stem cell lines and oligodendrocytes allow researchers to “turn back the clock” and observe how multiple sclerosis develops and progresses, potentially revealing the onset of the disease at a cellular level long before any symptoms are displayed. The improved protocol for deriving oligodendrocyte cells will also provide a platform for disease modeling, drug screening, and for replacing the damaged cells in the brain with healthy cells generated using this method.
“We are so close to finding new treatments and even cures for MS. The enhanced ability to derive the cells implicated in the disease will undoubtedly accelerate research for MS and many other diseases,” said Susan L. Solomon, NYSCF Chief Executive Officer.
Take me to the complete story . . .
The Latest on: Cell replacement therapy
[google_news title=”” keyword=”Cell replacement therapy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Cell replacement therapy
- U.S. FDA Approves Pfizer’s BEQVEZ™ (fidanacogene elaparvovec-dzkt), a One-Time Gene Therapy for Adults with Hemophilia Bon April 26, 2024 at 7:45 am
Pfizer Inc. (NYSE: PFE) announced today that the U.S. Food and Drug Administration (FDA) has approved BEQVEZ™ (fidanacogene elaparvovec-dzkt) for the ...
- FDA approves Beqvez gene therapy for adults with hemophilia Bon April 26, 2024 at 6:39 am
Beqvez is indicated for those on preventive treatment with a factor IX replacement therapy or have had life-threatening bleeding episodes.
- Therapeutic potential of human umbilical cord-derived mesenchymal stem cells in Crohn’s diseaseon April 25, 2024 at 6:00 am
Whether human umbilical cord-derived mesenchymal stem cells (hUC-MSCs) could heal luminal ulcers in Crohn’s disease.
- Cell Therapy Startup Energized via Smarter Operationson April 23, 2024 at 5:00 pm
A British-based cell therapy company is using smart operations to streamline its viral vector processing. According to Paul S. Carter, senior director, manufacturing science and technology (MSAT), at ...
- Cellares, BMS announce $380M cell therapy manufacturing agreementon April 23, 2024 at 8:28 am
Our collaboration ... and our collective expertise furthers our mission to accelerate access to life-saving cell therapies for patients globally," according to Fabian Gerlinghaus, co-founder and CEO, ...
- Early Data Indicate Cell Therapies Could ‘Reset the Clock’ in Parkinson’son April 21, 2024 at 9:03 pm
A one-time treatment for Parkinson’s disease could be a ‘market changer,’ experts told BioSpace, adding that cell therapies could limit the adverse effects seen with current drugs.
- How myeloid cell replacement could help treat autoimmune encephalomyelitison April 19, 2024 at 5:40 am
Autoimmune conditions such as multiple sclerosis (MS) are disorders of the immune system associated in this case with demyelination of the central nervous system (CNS). The term demyelination ...
- First Parkinson's Patient Gets Aspen Neuroscience's Autologous Cell Replacement Therapyon April 18, 2024 at 8:20 am
NEW YORK – Aspen Neuroscience on Wednesday said it has dosed the first patient in a first-in-human trial of ANPD001, its autologous neuronal cell replacement therapy for moderate-to-severe Parkinson's ...
- The four common medicines that could help prolong your lifeon April 17, 2024 at 9:24 am
Thanks to medical, scientific and social progress, a child born in the UK today has a greater than 50 per cent chance of living into their 90s. Might one day we be able to enhance our longevity even ...
- Philly-area biotech is worried about Congress’ crackdown on Chinese manufacturerson April 17, 2024 at 7:27 am
WuXi AppTec, one of the biggest biotech employers in Philadelphia, is facing intense scrutiny from the U.S. government, pushing local companies to seek alternatives.
via Bing News