There’s hope for patients with myotonic dystrophy. A new small molecule developed by researchers at the University of Illinois has been shown to break up the protein-RNA clusters that cause the disease in living human cells, an important first step toward developing a pharmaceutical treatment for the as-yet untreatable disease.
Steven C. Zimmerman, the Roger Adams Professor of Chemistry at the U. of I., led the group in developing and demonstrating the compound. The National Institutes of Health supported the work published in the journal ACS Chemical Biology.
Myotonic dystrophy type 1 is the most common form of muscular dystrophy in adults, affecting one in 8,000 people in North America. It causes progressive weakness as the muscles deteriorate over time. There is no treatment available for the disease; though a few measures can help ease some symptoms, nothing can halt their inevitable progression.
“This is a disease that currently doesn’t have any treatment, so we have a huge interest in finding therapeutic agents,” said graduate student Amin Haghighat Jahromi, the first author of the paper.
Myotonic dystrophy type 1, called DM1 for short, is caused by a mutation to one gene. In a healthy person, one small segment of the gene – a DNA sequence of CTG – is repeated a few times. In someone with DM1, the sequence is repeated more than 50 times, even up to thousands of repeats. The sequence is transcribed into RNA over and over, like a skipping record stuck in a loop. The repetitive RNA binds to the protein MBNL1, which is essential for regulating protein balance in cells. The RNA traps the MBNL1 protein in aggregates within the cell’s nucleus.
“The RNA is functioning in an abnormal way, and unfortunately, it’s toxic,” Zimmerman said. “MNBL regulates a process called alternative splicing that controls how much of different proteins are made. Affected cells make the proteins, just not at the right levels, so all the levels are imbalanced. There are more than 100 proteins that are affected.”
The Illinois group developed a small molecule that could infiltrate the nucleus and bind to the RNA, forcing it to let go of MBNL1 so the protein can do its job. The molecule is small and water-soluble so it can cross the membrane into the cell, which has been a challenge for researchers attempting to use methods with larger molecules. It specifically targets only the repeating RNA sequence so as not to interfere with other cellular functions.
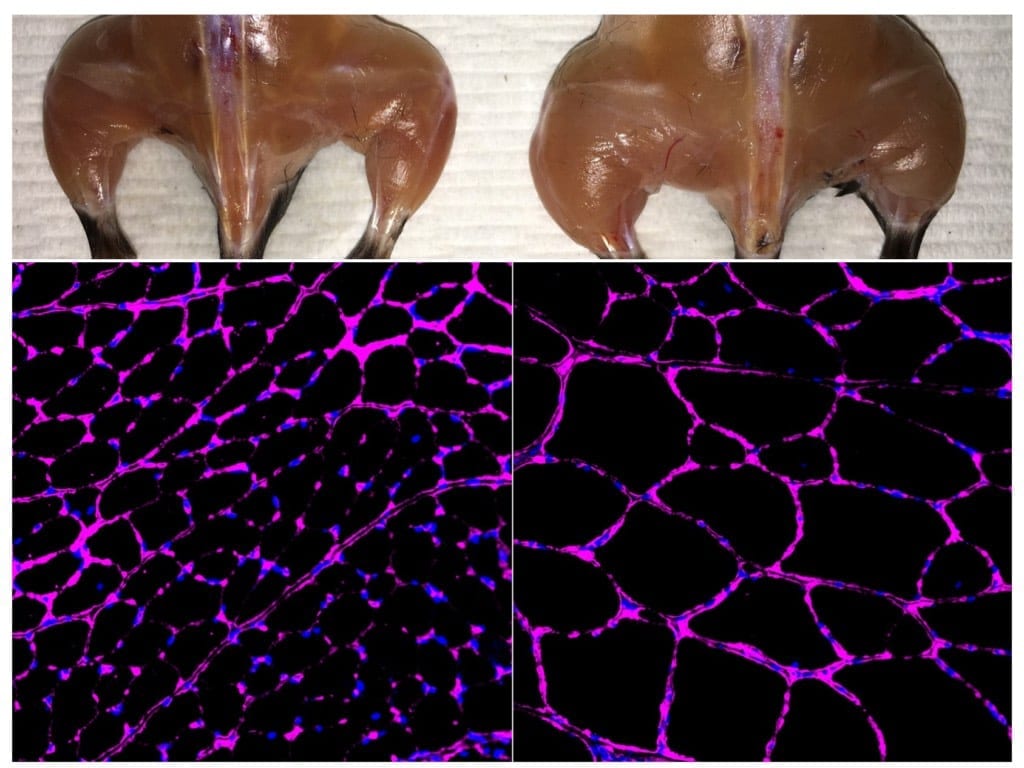
The researchers administered the molecule to live cells that have the disease features of DM1. Using advanced microscopy methods, they were able to watch the cells over time to see how they responded to the molecule. In only a few hours, they saw the clusters within the nucleus break up and were able to measure that the MBNL1 protein had increased its regulatory activity.
“This is the first study that gives direct evidence for the function of the compound,” Jahromi said. “We track how the cell is changing upon treatment with the compound and see the effect directly.”
Next, the researchers plan to begin collaborating with other groups to test their molecule in fruit flies and mice. Although the molecule will need many rounds of testing for toxicity, efficacy, metabolism and possible side effects before human trials can begin, finding a molecule that works in living cells is an important first step toward making a drug that could treat myotonic dystrophy.
The Latest Bing News on:
Muscular dystrophy treatment
- Patients With DMD Receiving Eteplirsen Show Prolonged Survival, Study Findson April 25, 2024 at 2:03 pm
The study provides evidence of survival benefits among patients with Duchenne muscular dystrophy (DMD) receiving eteplirsen compared with the natural history of the condition.
- DeWine: Ohio first state in the nation to screen newborn babies for Duchenne Muscular Dystrophyon April 24, 2024 at 9:00 pm
Ohio Gov. Mike DeWine on Monday announced that Ohio will become the first state in the nation to begin screening all newborn babies for Duchenne Muscular Dystrophy. The provision was included in HR 33 ...
- Researchers uncover SNUPN gene responsible for a new muscular disorderon April 24, 2024 at 8:03 am
A study, published in Nature Communications, sheds light on a newly identified subtype of muscular dystrophy, revealing an unsuspected role of SNUPN gene in muscle cell function.
- Capricor Therapeutics Announces Positive Type-B Meeting with FDA for CAP-1002 Program for Duchenne Muscular Dystrophyon April 24, 2024 at 8:00 am
Company Aligned with FDA on Demonstration of Non-Clinical Comparability; Allowing for Immediate Use of San Diego Manufacturing Facility--FDA ...
- Ohio sets the trend in screening newborns for rare disease. What to know about DMDon April 24, 2024 at 3:58 am
Ohio Gov. Mike DeWine announced the state will be the first in the nation to screen newborns for the rare disease Duchenne Muscular Dystrophy.
- Koç University Researchers Uncover SNUPN Gene Responsible for a New Muscular Disorderon April 23, 2024 at 11:53 pm
Koç University researchers have uncovered a new muscular disorder linked to SNUPN deficiency. Published in Nature Communications, this groundbreaking ...
- My son's smile gives me hope for Duchenne muscular dystrophy. A new law could help otherson April 23, 2024 at 5:01 am
Opinion: Newborn testing can give families a head start on giving a child with the degenerative muscular disease a shot at the best outcome in life.
- Transitioning to adulthood with Duchenne muscular dystrophyon April 22, 2024 at 8:35 am
Transitioning to being an adult with Duchenne muscular dystrophy can involve changing doctors, more independent living, and going to college.
- An FDA pathway can accelerate innovation for Duchenne muscular dystrophyon April 22, 2024 at 1:31 am
Few treatments are available for people with Duchenne muscular dystrophy. The FDA's accelerated approval program can help change that.
- 2-year EDG-5506 treatment helps preserve motor function in BMDon April 18, 2024 at 8:54 am
Oral treatment with EDG-5506 continued to stabilize muscle function after two years in men with Becker MD taking part in a Phase 1 trial.
The Latest Google Headlines on:
Muscular dystrophy treatment
[google_news title=”” keyword=”muscular dystrophy treatment” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
The Latest Bing News on:
Muscular dystrophy
- Midland Fire Fighters host the Annual Fill the Boot campaignon April 26, 2024 at 11:23 pm
that’s because MFD kicked off its annual fill the Boot campaign. Midland firefighters and volunteers are walking along the streets to fundraise money and their goal is to fill up this boot to help the ...
- These 15 One-of-a-Kind Watches Won’t Appear at the OnlyWatch Auction. We Asked the Brands Why.on April 26, 2024 at 9:00 am
We asked 15 brands why their unique timepieces for OnlyWatch will not appear at the rescheduled auction in Geneva next month, and what the fate of those watches might be.
- Patients With DMD Receiving Eteplirsen Show Prolonged Survival, Study Findson April 25, 2024 at 2:03 pm
The study provides evidence of survival benefits among patients with Duchenne muscular dystrophy (DMD) receiving eteplirsen compared with the natural history of the condition.
- Muscular Dystrophy UK Earns PIF TICK Accreditationon April 25, 2024 at 7:48 am
Muscular Dystrophy UK, the leading charity for more than 110,000 children and adults in the UK living with one of over 60 muscle wasting and weakening ...
- DeWine: Ohio first state in the nation to screen newborn babies for Duchenne Muscular Dystrophyon April 24, 2024 at 9:00 pm
Ohio Gov. Mike DeWine on Monday announced that Ohio will become the first state in the nation to begin screening all newborn babies for Duchenne Muscular Dystrophy. The provision was included in HR 33 ...
- Researchers uncover SNUPN gene responsible for a new muscular disorderon April 24, 2024 at 8:03 am
A study, published in Nature Communications, sheds light on a newly identified subtype of muscular dystrophy, revealing an unsuspected role of SNUPN gene in muscle cell function.
- Gov. DeWine announces Ohio will be first state to screen newborns for DMDon April 24, 2024 at 7:23 am
Gov. Mike DeWine announced Tuesday that the state of Ohio will become the first in the United States to screen all newborn babies for Duchenne muscular dystrophy.
- Edgewise gets EU orphan drug status for muscular dystrophy drugon April 23, 2024 at 7:29 am
Edgewise Therapeutics (EWTX) has received orphan drug designation from EU regulators for sevasemten in the treatment of Becker and Duchenne muscular dystrophies. Read more here.
- Duchenne UK and Parent Project Muscular Dystrophy Award $500,000 to Evaluate Safety and Tolerability of Muscle Progenitor Cells in Phase 1 Trialon April 23, 2024 at 7:01 am
Parent Project Muscular Dystrophy (PPMD), a US nonprofit organization, and the UK charity Duchenne UK, two leading organizations dedicated to ending Duchenne muscular dystrophy (Duchenne), are excited ...
- My son's smile gives me hope for Duchenne muscular dystrophy. A new law could help otherson April 23, 2024 at 5:01 am
Opinion: Newborn testing can give families a head start on giving a child with the degenerative muscular disease a shot at the best outcome in life.
The Latest Google Headlines on:
Muscular dystrophy
[google_news title=”” keyword=”muscular dystrophy” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]