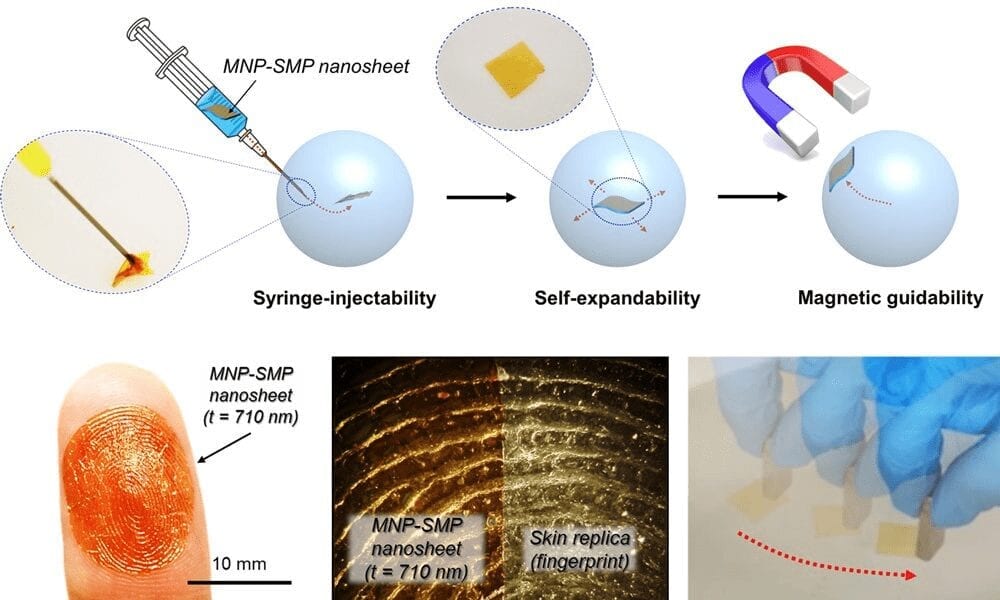
Ultrathin films are able to be injected using minimally-invasive syringe needles and can be used as a platform to deliver molecular and cellular drugs. The use of shape-memory polymer also enables unprecedented temperature-dependent control to allow for adhesion and removal of the nanosheets on biological surfaces.
Syringe-injectable biomaterials, medical devices and engineered tissues have attracted great attention as minimally-invasive implants for diagnosis, therapy and regenerative medicine. Free-standing polymeric ultrathin films, commonly referred to as polymeric nanosheets, are one of the commonly used platforms for syringe-injectable biomedical devices because of their flexibility and conformability.
These nanosheets are less than 1 micrometer in thickness, which is thinner than a strand of hair that is usually about 100 micrometers wide. They are a promising platform for drug delivery through needle-injection. Despite recent development in nanosheets technologies using polylactic acid (PLA) and polylactic-co-glycolic acid (PLGA), polymeric nanosheets are yet to overcome several technical challenges to serve as an effective injectable platform: namely, (1) limitation in the size of nanosheets injectable through medical needles, (2) suboptimal mechanical robustness (e.g., ripping during injection), and (3) limited control over shape recovery and motion control after injection.
To overcome these limitations, researchers from Digital Manufacturing and Design (DManD) Centre at the Singapore University of Technology and Design (SUTD) developed nanosheets using polyurethane-based shape-memory polymer (SMP) and magnetic nanoparticles (MNP) to demonstrate unprecedented capabilities to handle nanosheets. SMP offers two unique mechanical characteristics – a large change in the Young’s moduli by the change in temperature, and shape-memory effect (SME) to recover the memorised shape.
In addition, the researchers demonstrated that the fabricated SMP nanosheets can be rendered magnetic with MNP to perform non-contact motion control using an external magnetic field. Specifically, the following four capabilities were demonstrated by using the 710 nm thick nanosheet with the glass transition temperature (Tg) of 25?C: (1) syringe-injectability through the medical needles, (2) self-expandability after ejection, (3) conformability and removability on the biological surfaces, and (4) guidability in an external magnetic field. These capabilities enable in vivo practical applications as a syringe-injectable platform.
As an added advantage, the change of the modulus by temperature offers a unique capability to control the adhesion and removal of the MNP-SMP nanosheet on the biological surfaces. This would have been difficult to achieve using conventional nanosheets having a constant modulus and has not been demonstrated previously.
Envisaging the syringe-injectable delivery of molecular drugs or cellular constructs into internal organs, the researchers added the MNP-SMP nanosheets with an additional layer of PLGA, which is best known as a biomaterial used for drug delivery, to extend the functionality as a carrier of molecular and cellular drugs. This can be done without compromising the demonstrated capabilities. SMP and MNP offered the same capabilities to the nanosheets containing an additional layer of PLGA, suggesting the vast potential of the developed nanosheets for drug and cell delivery.
“The MNP-SMP nanosheets can be further functionalized by loading or printing drugs, cells and electric circuits on the surface by integrating emerging printing technologies such as inkjet printing, 3D printing and bioprinting,” said Dr Kento Yamagishi from SUTD, the lead author of the paper.
“The MNP-SMP nanosheets will contribute to the development of advanced syringe-injectable medical devices as a platform to deliver drugs and cells to the specific site or lesion in the body for minimally invasive diagnosis and therapy,” added Principal Investigator, Assistant Professor Michinao Hashimoto from SUTD.
The Latest on: Syringe-injectable medical devices
[google_news title=”” keyword=”syringe-injectable medical devices” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Syringe-injectable medical devices
- How the medical syringe became a tool of control when police restrain peopleon April 26, 2024 at 5:32 am
An investigation led by The Associated Press found at least 94 people died after they were given sedatives and restrained by police from 2012 through 2021. About half of the ...
- Global Intradermal Injection Industryon April 26, 2024 at 5:14 am
Global Intradermal Injection Industry forecasted to registering 10.8% CAGR, and projected to reach US$ 10,814.47 Million by 2032.
- Medical Devices Newson April 25, 2024 at 6:36 am
Ranked by W-2s issued by Bay Area offices in 2023 Showcase your company news with guaranteed exposure both in print and online Gather your employees for an afternoon of games as we honor the Best ...
- LSU researchers develop syringe device for enhanced stem cell therapyon April 24, 2024 at 6:00 am
LSU researchers led by veterinary medicine professor Mandi Lopez have introduced a groundbreaking syringe device that could revolutionize tendon injury treatment.
- Plastics industry veterans launch Polymer Medicalon April 22, 2024 at 1:51 pm
Founded by Benjamin Harp and Tom Rybicki, Polymer Medical provides contract injection-molding and assembly for healthcare and biosciences.
- Counterfeit Botox Is Making People Sick—Here’s How to Spot an Unlicensed Injectoron April 19, 2024 at 11:37 am
The FDA and the CDC are investigating claims of counterfeit Botox in nonmedical settings. Experts share everything you need to know.
- I Tried Skinvive, the New "Skincare in a Syringe"on April 18, 2024 at 11:58 am
Before trying the treatment on myself, I wanted to hear about it from the experts. I sat down for a video call with Saami Khalifian, M.D., double board-certified cosmetic dermatologist and surgeon and ...
- Global Smart Syringes Market Size To Exceed USD 20.5 Billion By 2033 | CAGR Of 8.22%on April 17, 2024 at 1:01 pm
The Global Smart Syringes Market Size was Valued at USD 9.3 Billion in 2023 and the Worldwide Smart Syringes Market Size is Expected to Reach USD 20.5 Billion by 2033, according to a research report ...
- Halozyme Therapeutics gets grant for injection device with safety features for medicament administrationon April 15, 2024 at 6:16 am
Discover the innovative injection device patented by Halozyme Therapeutics Inc, ensuring safe and effective medication delivery to patients. Explore the unique features and components designed for ...
via Bing News