Researchers at Penn State have developed an innovative technology to regenerate functional neurons after brain injury and also in model systems used for research on Alzheimer’s disease.
The scientists have used supporting cells of the central nervous system, glial cells, to regenerate healthy, functional neurons, which are critical for transmitting signals in the brain.
Gong Chen, a professor of biology, the Verne M. Willaman Chair in Life Sciences at Penn State, and the leader of the research team, called the method a breakthrough in the long journey toward brain repair.
“This technology may be developed into a new therapeutic treatment for traumatic brain and spinal cord injuries, stroke, Alzheimer’s disease, Parkinson’s disease and other neurological disorders,” Chen said. The research was posted online Dec. 19 by the journal Cell Stem Cell.
When the brain is harmed by injury or disease, neurons often die or degenerate, but glial cells become more branched and numerous. These “reactive glial cells” initially build a defense system to prevent bacteria and toxins from invading healthy tissues, but this process eventually forms glial scars that limit the growth of healthy neurons.
“A brain-injury site is like a car-crash site,” Chen explained. “Reactive glial cells are like police vehicles, ambulances and fire trucks immediately rushing in to help — but these rescue vehicles can cause problems if too many of them get stuck at the scene. The problem with reactive glial cells is that they often stay at the injury site, forming a glial scar and preventing neurons from growing back into the injured areas.”
So several years ago, Chen’s lab tested new ways to transform glial scar tissue back to normal neural tissue.
“There are more reactive glial cells and fewer functional neurons in the injury site,” Chen said, “so we hypothesized that we might be able to convert glial cells in the scar into functional neurons at the site of injury in the brain. This research was inspired by the Nobel Prize-winning technology of induced pluripotent stem cells (iPSCs) developed in Shinya Yamanaka’s group, which showed how to reprogram skin cells into stem cells.”
Chen and his team began by studying how reactive glial cells respond to a specific protein, NeuroD1, which is known to be important in the formation of nerve cells in the hippocampus area of adult brains. They hypothesized that expressing NeuroD1 protein into the reactive glial cells at the injury site might help to generate new neurons — just as it does in the hippocampus. To test this hypothesis, his team infected reactive glial cells with a retrovirus that specifies the genetic code for the NeuroD1 protein.
“The retrovirus we used is replication-deficient and thus cannot kill infected cells like other viruses found in the wild,” Chen said. “More importantly, a retrovirus can infect only dividing cells such as reactive glial cells, but it does not affect neurons, which makes it ideal for therapeutic use with minimal side effect on normal brain functions.”
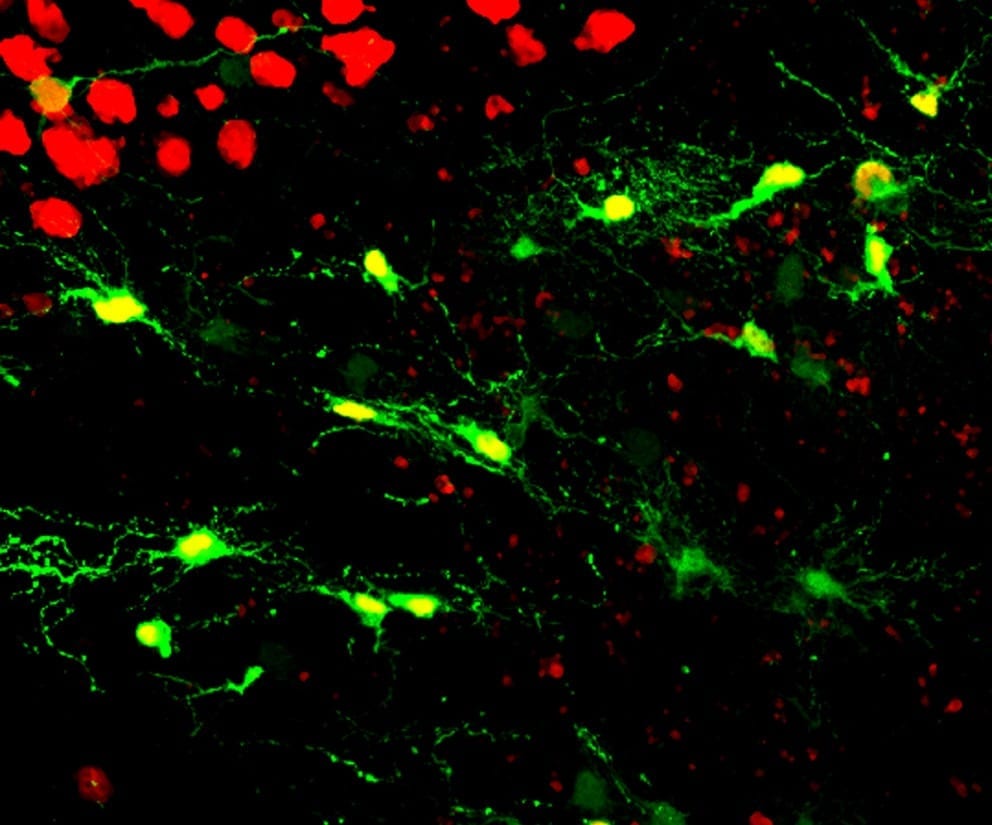
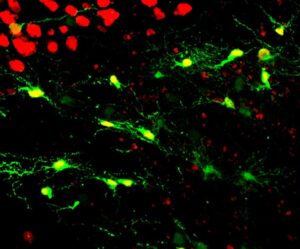
In a first test, Chen and his team investigated whether reactive glial cells can be converted into functional neurons after injecting NeuroD1 retrovirus into the cortex area of adult mice. The scientists found that two types of reactive glial cells — star-shaped astroglial cells and NG2 glial cells — were reprogrammed into neurons within one week after being infected with the NeuroD1 retrovirus.
“Interestingly, the reactive astroglial cells were reprogrammed into excitatory neurons, whereas the NG2 cells were reprogrammed into both excitatory and inhibitory neurons, making it possible to achieve an excitation-inhibition balance in the brain after reprogramming,” Chen said.
His lab also performed electrophysiological tests, which demonstrated that the new neurons converted by the NeuroD1 retrovirus could receive neurotransmitter signals from other nerve cells, suggesting that the newly converted neurons had successfully integrated into local neural circuits.
In a second test, Chen and his team used a transgenic-mouse model for Alzheimer’s disease, and demonstrated that reactive glial cells in the mouse’s diseased brain also can be converted into functional neurons. Furthermore, the team demonstrated that even in 14-month-old mice with Alzheimer’s disease — an age roughly equivalent to 60 years old for humans — injection of the NeuroD1 retrovirus into a mouse cortex can still induce a large number of newborn neurons reprogrammed from reactive glial cells.
“Therefore, the conversion technology that we have demonstrated in the brains of mice potentially may be used to regenerate functional neurons in people with Alzheimer’s disease,” Chen said.
The Latest on: Neuron regeneration
[google_news title=”” keyword=”Neuron regeneration” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Neuron regeneration
- Transformative Discovery Could Solve Billion-Dollar Problem of Poorly Managed Wound Healingon May 7, 2024 at 6:35 pm
Scientists discover a crucial protein that enhances wound healing and muscle regeneration, a process often impaired by conditions such as diabetes and aging. Researchers have discovered an essential ...
- Former Salisbury men’s lacrosse goalie Johnny Rodriguez vows to ‘fight like hell’ against ALS diagnosison May 7, 2024 at 10:30 am
Gambrills resident and Mount Saint Joseph graduate Johnny Rodriguez has accepted his ALS diagnosis as a challenge.
- Potential Treatment Strategies for Enhancing Neuroplasticity and Regeneration After Ischemic Strokeon May 6, 2024 at 4:59 pm
Rehabilitative training can enhance this reorganization. A critical period of increased neuronal plasticity might exist after stroke. This critical period may be determined by the balance between ...
- Red light therapy for repairing spinal cord injury passes milestoneon May 6, 2024 at 6:43 am
Patients with spinal cord injury (SCI) could benefit from a future treatment to repair nerve connections using red and near-infrared light.
- Deciphering Proteins and their Functions in the Regenerating Retinaon April 18, 2024 at 5:00 pm
In fact, axon regeneration is not determined by the presence or absence of a certain molecule. It is rather a complex pathophysiological process involving neuronal and non-neuronal changes ...
- Early developmental spinal cord extracellular matrix can promote neural regenerationon April 3, 2024 at 7:16 am
However, achieving satisfactory repair outcomes remains challenging, making nerve regeneration after SCI ... migration, and neuronal differentiation of NPCs, as well as promoted axon growth ...
- ‘New way of thinking’: Chinese researchers unveil unique strategy for tissue regeneration, functionon April 2, 2024 at 2:23 am
Tissue regeneration involves a complicated coordination ... and promoted the growth of neurons in the area. This boost in neuron growth allowed for more functional injury repair, allowing the ...
- Fenglian Xu, Ph.D.on March 4, 2024 at 12:06 am
Neuronal outgrowth, regeneration, synapse formation and plasticity are pivotal not only for normal brain development and function, but also for brain repair after injury or degeneration. The primary ...
- New biomaterial implants for spinal cord neuronal regenerationon April 28, 2021 at 5:18 am
The NeuroStimSpinal project is developing new a graphene-based and decellularised biomaterial that can foster neuronal regeneration, laying down the foundation for innovative approaches in spinal cord ...
- Priming axon regenerationon March 18, 2019 at 11:00 am
In healthy neurons, myelin is thought to provide cues essential for cell maintenance; when neurons are damaged, however, these signals appear to be misread, preventing neuronal regeneration.
via Bing News