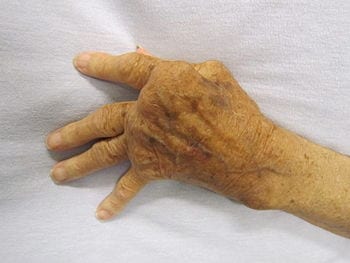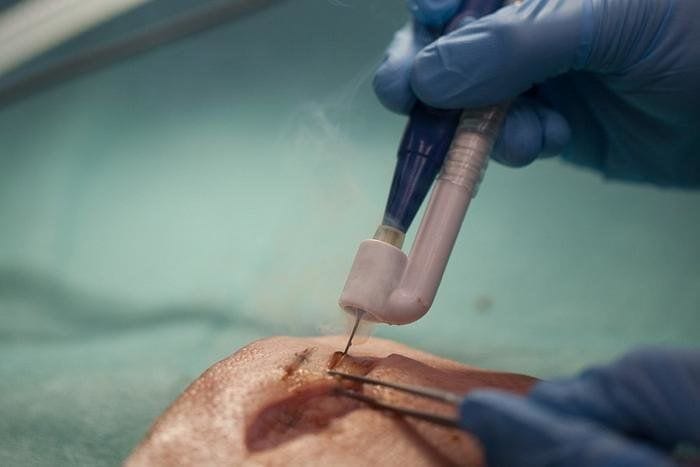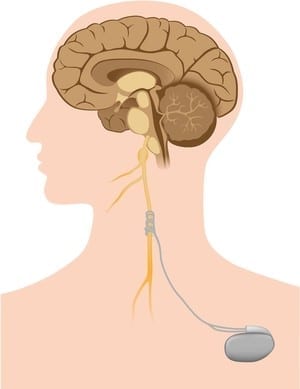
The results of a pilot study presented today at the Annual European Congress of Rheumatology (EULAR 2019) suggest that electro stimulation of one of the nerves connecting the brain to the body (the vagus nerve), could provide a novel treatment approach for patients with rheumatoid arthritis.
“This is a really exciting development. For many patients suffering from rheumatoid arthritis, current treatments don’t work, or aren’t tolerated,” said Professor Thomas Dörner, Chairperson of the Scientific Programme Committee, EULAR. “These results open the door to a novel approach to treating not only rheumatoid arthritis, but other chronic inflammatory diseases. This is certainly an area for further study.”
The vagus nerve is the longest and the most complex of the 12 pairs of cranial nerves that originate from the brain. The name ‘vagus’ comes from the latin word for ‘wandering’. This is because the vagus nerve wanders from the brain into the organs of the neck, chest and abdomen.
Recent advances in neuroscience and immunology have mapped circuits in the brain that regulate immune responses. In one of the circuits, the ‘inflammatory reflex’, signals are transmitted in the vagus nerve that inhibit the production of cytokines including tumor necrosis factor (TNF), an inflammatory molecule that is a major therapeutic target in rheumatoid arthritis. It is thought that, by stimulating the activity of this inflammatory reflex, innate immune responses can be modulated without abolishing them or producing significant immunosuppression.
In this pilot study, a novel miniaturised neurostimulator called a MicroRegulator was implanted into 14 patients with rheumatoid arthritis who had failed on at least two biologics or targeted oral therapies with different mechanisms of action. Patients were randomised to three groups who were either placebo, stimulated once daily, or stimulated four times a day for 12 weeks. At the end of the study, the patients who received once-daily stimulation were shown to have a better response than those on four-times-daily stimulation with two thirds meeting the EULAR good or moderate response criteria and a mean change in DAS28-CRP of -1.24. The mean change in DAS28-CRP* in the placebo group was 0.16.1
Cytokines (a broad and loose category of small proteins that are important in cell signalling) were also measured in the study with the actively stimulated groups showing a decrease of more than 30% in levels of Interleukin (IL) 1?, IL-6, and TNF-?. Implantation and stimulation were generally well tolerated with no device or treatment-related SAEs and two surgery-related adverse events that resolved without clinically significant effects.
“Our pilot study suggests this novel MicroRegulator device is well tolerated and reduces signs and symptoms of rheumatoid arthritis,” said Mark Genovese, M.D., James W. Raitt Endowed Professor of Medicine, Stanford University, Stanford, California, USA. “These data support the study of this device in a larger placebo-controlled study as a novel treatment approach for rheumatoid arthritis and possibly other chronic inflammatory diseases.”
This study follows a proof-of-concept study which used reprogrammed epilepsy stimulators on the vagus nerve to demonstrate reduced systemic inflammation and improved disease activity in 17 patients with rheumatoid arthritis.
The study included 14 patients with active rheumatoid arthritis who had had an insufficient response to more than two biological disease modifying anti-rheumatic drugs (bDMARDs) or JAK inhibitors with more than two modes of action. All patients remained on stable background of methotrexate. The first three patients were implanted and stimulated after three weeks, following safety review board approval, the remaining 11 patients were implanted and randomised to one minute of stimulation once-daily, one minute of stimulation four times daily, or one minute of placebo stimulation.
Learn more: Vagus nerve stimulation study shows significant reduction in rheumatoid arthritis symptoms
The Latest on: Electro stimulation
[google_news title=”” keyword=”electro stimulation” num_posts=”10″ blurb_length=”0″ show_thumb=”left”]
via Google News
The Latest on: Electro stimulation
- Soterix Medical Launches MxN-GO EEG System for Wire-free HD-tES and EEGon April 30, 2024 at 9:08 am
Soterix Medical, the pioneers in HD-tDCS/HD-tES, collaborated with mBrainTrain, the experts in research-grade mobile EEG, to create the ultimate solution for mobile hybrid stimulation/EEG studies.
- Neuromodulation to see market growth, pioneering technology for epilepsyon April 30, 2024 at 4:32 am
A DBS is a device that comprises a brain-oriented pacemaker (an implantable pulse generator) that sends electrical stimulation through surgically implanted electrodes to specific targets in the brain.
- Medtronic bags FDA approval for Inceptiv spinal cord stimulatoron April 29, 2024 at 3:42 am
The device is now cleared to enter the US market to treat chronic pain by outputting electrical pulses in rhythm with the user.
- Medtronic snags FDA approval for body-sensing, closed-loop spinal cord stimulatoron April 26, 2024 at 8:31 am
Medtronic has secured FDA approval for its first closed-loop spinal cord stimulator, designed to take in signals from the body and adjust its therapy automatically. | The Inceptiv implant for chronic ...
- FDA approves Medtronic’s Inceptiv closed-loop rechargeable spinal cord stimulatoron April 26, 2024 at 6:15 am
Inceptiv treats chronic pain and represents Medtronic’s first SCS device to offer a closed-loop feature. This feature senses each person’s unique biological signals, adjusting stimulation ...
- Global Functional Electrical Stimulation Industryon April 24, 2024 at 8:28 am
Global Functional Electrical Stimulation Industry projects a 4.1% CAGR to US$ 931.4 Million by 2033, driven by spinal cord injury prevalence ...
- Does Electrical Muscle Stimulation Really Supercharge Your Workouts?on April 23, 2024 at 1:32 pm
Electrical muscle stimulation (EMS) workouts are having a moment—not for the first time, and probably not for the last—based on their futuristic vibes and the fitness industry’s constant quest to sell ...
- Neuronoff hits first-in-human milestone for Injectrode neuromodon April 23, 2024 at 10:39 am
Neuronoff announced today that it completed the first-in-human clinical trial evaluating its flagship Injectrode neuromodulation product.
- NeurAxis Announces New Medical Policy Coverage in North Dakotaon April 23, 2024 at 6:37 am
The current medical treatments, which are off-label drugs, can often have serious side effects, and most lack scientific evidence of efficacy. “We are pleased to announce a new medical coverage policy ...
- Body20 now offering electro-muscle-stimulation physical training in Springon April 22, 2024 at 10:00 pm
The business focuses on providing customers customized workouts through physical training with electro-muscle-stimulation technology to create muscle contractions, according to the company’s website.
via Bing News