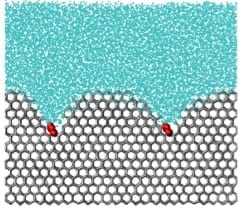
PHOTO CREDIT: University of Utah
A simulation of an ice inhibiting molecule. The molecule, in red, is like a weight on the surface of the ice crystal, curving it and preventing further ice crystal growth.
Cell therapies hold great promise for revolutionizing the treatment of cancers and autoimmune diseases. But this multibillion-dollar industry requires long-term storage of cells at super-cold cryogenic conditions, while ensuring they’ll continue to function upon thawing. However, these cold temperatures trigger the formation and growth of ice, which can pierce and tear apart cells. Research published in the Journal of the American Chemical Society by University of Utah chemists Pavithra Naullage and Valeria Molinero provides the foundation to design efficient polymers that can prevent the growth of ice that damages cells.
Nature’s antifreeze
Current strategies to cryopreserve cells and organs involve bathing them with large amounts of dimethyl sulfoxide, a toxic chemical that messes up ice formation but stresses the cells, decreasing their odds for survival.
Nature, however, has found a way to keep organisms alive under extreme cold conditions: antifreeze proteins. Fish, insects and other cold-blooded organisms have evolved potent antifreeze glycoproteins that bind to ice crystallites and halt them from growing and damaging cells.
The growing area of cell-based therapeutics demands the development of potent inhibitors of ice recrystallization that can compete in activity with natural antifreeze glycoproteins but do not have the cost and toxicity of dimethyl sulfoxide. This demand has propelled the synthesis of polymers that mimic the action of antifreeze glycoproteins. But the most potent synthetic ice recrystallization inhibitor found to date, polyvinyl alcohol (PVA), is orders of magnitude less potent than natural glycoproteins.
“Efforts to identify stronger inhibitors for ice growth seem to have stalled, as there is not yet a molecular understanding of the factors that limits the ice recrystallization inhibition efficiency of polymers,” Molinero says.
A hidden polymer design variable
How do molecules prevent ice crystals from getting bigger? Molecules that bind strongly to ice pin its surface—like stones on a pillow—making the ice front develop a curved surface around the molecules. This curvature destabilizes the ice crystal, halting its growth. Molecules that stay bound to ice for times longer than the time it takes to grow ice crystals succeed in preventing further growth and recrystallization.
Molinero and Naullage used large-scale molecular simulations to elucidate the molecular underpinnings of how flexibility, length and functionalization of polymers control their binding to ice and their efficiency to prevent ice growth. Their study shows that the bound time of the molecules at the ice surface is controlled by the strength of their ice binding coupled with the length of the polymer and how fast they propagate on the ice surface.
“We found that the efficiency of flexible polymers in halting ice growth is limited by the slow propagation of their binding to ice,” Molinero says.
The study dissects the various factors that control the binding of flexible polymers to ice and that account for the gap in potency of PVA and natural antifreeze glycoproteins. In a nutshell, each block of antifreeze glycoproteins binds more strongly to ice than PVA does, and are also favored by their secondary molecular structure that segregates the binding and non-binding blocks to allow them to attach faster to ice to stop its growth.
“To our knowledge, this work is first to identity the time of propagation of binding as a key variable in the design of efficient ice-binding flexible polymers,” Naullage says. “Our study sets the stage for the de novo design of flexible polymers that can meet or even surpass the efficiency of antifreeze glycoproteins and make an impact in biomedical research.”
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Cryogenics
- Dr. Andrew A. Gage, 101, pioneering surgeon who helped develop heart pacemaker
Born in West Seneca, the son of Andrew B. and Estelle Stahl Gage, he was a graduate of West Seneca High School. He earned his medical degree in 1944 from the University of Buffalo, then served in the ...
- Cryogenic Pump Market Surges to US$ 2,092.2 million by 2033, Fueled by Solar Power Innovations
As projected by Future Market Insight, the global cryogenic pump Industry is poised to surge to US$ 2,092.2 million by 2033, exhibiting a CAGR of 6.8%. This growth marks a substantial leap from its ...
- Quantum challenge to be solved one mile underground
Radiation from space is a challenge for quantum computers as their computation time becomes limited by cosmic rays. Researchers from Chalmers University of Technology, Sweden, and University of ...
- Storage Dewar and Dry Shipper Market To Reach USD 2.2 Million By 2032 | DataHorizzon Research
The storage dewar and dry shipper market size was valued at USD 1.2 Million in 2022 and is anticipated to reach USD 2.2 Million by 2032 at a CAGR of 6.5% during the forecast period 2023-2032.Fort ...
- Dover Corp: Strong Q1 Performance and Promising Order Trends Support Buy Rating
Analyst Joe Ritchie from Goldman Sachs maintained a Buy rating on Dover (DOV – Research Report) and increased the price target to $191.00 ...
Go deeper with Google Headlines on:
Cryogenics
[google_news title=”” keyword=”cryogenics” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
Antifreeze glycoproteins
- Your coolant can tell you if other areas of maintenance are falling apart
You may remember we covered coolants last month, specifically how topping off coolant all winter can throw your system out of balance when the weather turns warmer and limit your coolant system's ...
- Antifreeze Protein Market Sales to Top USD 90.2 Billion in Revenues by 2033 at a CAGR of 29.6%
According to Market.us, the Antifreeze Protein Market size is projected to surpass around USD 90.2 billion by 2032 from USD 9.3 billion in 2023, and it is poised to reach a registered CAGR of 29.6% ...
- He didn't want her to have the baby. So he poisoned their newborn's bottle with antifreeze.
A Georgia man convicted of poisoning his newborn baby by adding antifreeze to the child's breastmilk bottles has been sentenced to 50 years after a jury found him guilty of the crime. Curtis Jack ...
- Man Sentenced to 50 Years in Jail for Poisoning His Baby with Antifreeze in Her Milk Bottle
A man in South Fulton, Georgia, was sentenced to 50 years in prison for attempting to kill his baby daughter by poisoning her breast milk with antifreeze, authorities said. On Oct. 1, 2020 ...
- Father charged for poisoning newborn baby’s breastmilk with antifreeze, police say
ATLANTA, Ga. (WANF/Gray News) - A Georgia father is facing 50 years in prison after police say he poisoned his newborn baby’s breastmilk with antifreeze. According to South Fulton police ...
Go deeper with Google Headlines on:
Antifreeze glycoproteins
[google_news title=”” keyword=”antifreeze glycoproteins” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]