A New Avenue in Ca2+ Ion-Exchanged A-Type Zeolite Chemistry
Credit?2021 Okayama University-InorgChem
Scientists report hitherto unobserved high-performance CO2 adsorption in zeolites at room temperature, opening doors to applications in air purification
Reducing the amount of CO2 in our environment is crucial for mitigating climate change and needs materials that can adsorb CO2 from air under ambient conditions. In a new study, scientists from Japan explore the CO2 adsorption properties of zeolite, which has been overlooked in this regard, and report an unprecedented selective adsorption behavior in the extremely lower pressure region and at room temperature, paving the way for its future applications in air purification.
It is now well known that carbon dioxide is the biggest contributor to climate change and originates primarily from burning of fossil fuels. While there are ongoing efforts around the world to end our dependence on fossil fuels as energy sources, the promise of green energy still lies in the future. Can something be done in the meantime to reduce the concentrations of CO2 in the atmosphere?
It would, in fact, be great if the CO2 in the atmosphere could simply be adsorbed! Turns out, this is exactly what direct air capture (DAC), or the capture of CO2 under ambient conditions, aims to do. However, no such material with the ability to adsorb CO2 efficiently under DAC conditions has so far been developed. “It is well known that CO2 is acidic in nature. Therefore, materials with basic nature are generally utilized as adsorbents for CO2. However, that often leads to corrosion of the system and is also not suitable for recycling the adsorbed CO2,” explains Professor Yasushige Kuroda from Okayama University, Japan, who conducts research on surface chemistry.
Against this backdrop, in a recent study published in the Journal of Materials Chemistry A, scientists from Okayama University and Japan Synchrotron Radiation Research Institute (JASRI) led by Prof. Kuroda explored the adsorption properties of a material that has so far remained an “underdog”: zeolites (minerals containing mainly aluminum and silicon oxides). “Zeolite materials have received little attention as adsorbents owing to their low CO2 adsorption capacity at room temperature and in the lower pressure adsorption region, as well as their poor selectivity over nitrogen,” says Prof. Kuroda.
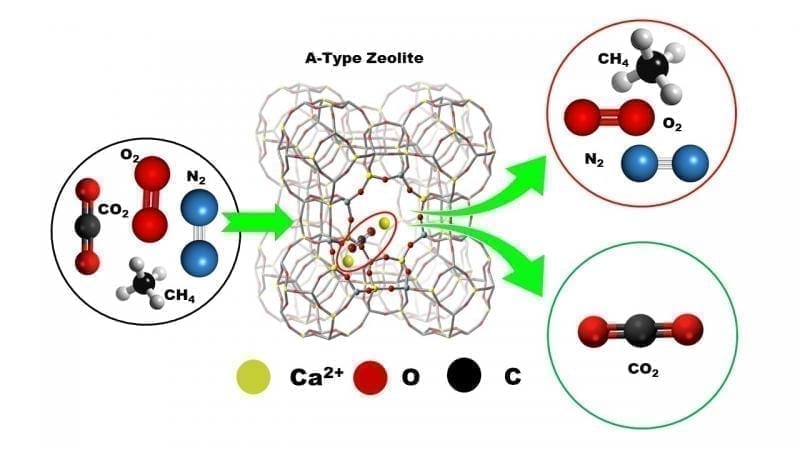
In their study, Prof. Kuroda and his team designed an ion-exchanging method of zeolite with alkaline-earth ions and achieved a remarkably high CO2 adsorption under ambient conditions. The team specifically chose an A-type zeolite (silicon/aluminum ratio of 1) because of its appropriate pore size for adsorbing CO2, while the alkaline-earth ion exchange imparted a large electric field strength that, supposedly, acted as a driving force for the adsorption. Scientists chose a doubly charged calcium ion (Ca2+) as the exchange ion since it allowed for the greatest amount of adsorption. In fact, the adsorbed volume noted was the largest amount of CO2 to have ever been adsorbed by any zeolite system, surpassing that for other materials under similar conditions!
To investigate the underlying adsorption mechanism, the scientists carried out far-infrared (far-IR) measurements and backed them up with density functional theory (DFT) calculations. The far-IR spectra, which detected the vibrational modes due to Ca2+-zeolite vibration, showed a distinct shift towards longer wavelengths following CO2 adsorption, a feature scientists could not recognize in other samples, e.g. Na-ion exchanged A-type zeolite. They further verified their observation with a model that showed good agreement with DFT calculations.
Moreover, the scientists were able to completely desorb the adsorbed CO2 and recover the original sample and its specific adsorption properties. In addition, the sample showed a superior selective adsorption of CO2 from other gases after the scientists examined the separation of CO2 using a model gas that emulated ambient air in its composition.
The findings thus bring zeolites to the forefront as an efficient adsorbent of CO2 under ambient conditions, a feat previously thought unachievable with these systems. “Our work can open doors to potentially novel applications of zeolites, such as in the cleaning of air inside semi-closed spaces including space shuttles, submarines, and concert halls, and as an adsorbent material in the anesthetic process,” speculates Prof. Kuroda excitedly.
One thing is for sure, though: chemists will never look at zeolite in the same way again.
Original Article: Zero to Hero: Overlooked Material Could Help Reduce Our Carbon Footprint
More from: Okayama University
The Latest Updates from Bing News & Google News
Go deeper with Bing News on:
Zeolites
- Self-adjusted reaction pathway enables efficient oxidation of aromatic C-H bonds over Co@Y catalyst
The selective oxidation of aromatic C-H bonds has drawn significant attention in both industrial and fine chemistry due to its crucial role in converting readily- and cheaply-available aromatic ...
- United States Antimony Corp (UAMY) Q4 2023 Earnings Call Transcript Highlights: Navigating ...
Cash and Cash Equivalents: Decreased by $7.2 million during 2023. Operational Losses: $5.9 million from Mexico operations. Inventory Purchases: $1.8 million related to Mexico operations. Noncash ...
- Advanced nuclear magnetic resonance technique reveals precise structural, dynamical details in zeolites
Zeolites are widely used in many industries, yet their intrinsic catalytic nature is not completely understood, due to the complexity of the hydroxyl-aluminum moieties.
- FMI Predicts Robust Growth in Global Refinery Catalyst Market, Reaching US$ 12,366 Million by 2030 End
Increasing use of transportation fuel is likely to have a direct impact on the demand for refinery catalyst, according to a recent report published by Future Market Insights (FMI). Catalytic processes ...
- Rice Husk Ash Market Worth $3.2 Billion By 2028, At A CAGR Of 4.5%, Says Marketsandmarkets™
The Global Rice Husk Ash Market is projected to grow from USD 2.6 billion in 2023 to USD 3.2 billion by 2028, at a CAGR of 4.5% during the forecast period, as per the recent study by ...
Go deeper with Google Headlines on:
Zeolites
[google_news title=”” keyword=”zeolites” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]
Go deeper with Bing News on:
CO2 adsorption
- The Dirtiest Side of EVs: Rare Earths and Conflict Metals
"Typical mining operations for rare earth mines vary from hard rock to heavy-mineral sands and ionic adsorption clay operations," de Jonge said. "For hard rock assets (the most common), metallurgy is ...
- Imagine powering your phone with carbon dioxide
The carbon-negative power generator, reported in the journal Nature Communications, is only a small proof-of-concept laboratory-scale device right now. But it could pave the way towards a new ...
- Can this ocean-based carbon plant help save the world? Some scientists are raising red flags
On a slice of the oceanfront in west Singapore, a startup is building a plant to turn carbon dioxide from air and seawater into the same material as seashells, in a process that will also produce ...
- Australian researchers develop prototype device to devour carbon dioxide to make electricity
Australian researchers have built an electrical generator that consumes carbon dioxide, generates electricity and admits no exhausts. They say the technology could create a new industrial-scale ...
- Carbon Dioxide Levels Have Passed a New Milestone
Source: National Oceanic and Atmospheric Administration’s Global Monitoring Laboratory The chart shows monthly numbers of carbon dioxide molecules per million molecules of dry air. Because of ...
Go deeper with Google Headlines on:
CO2 adsorption
[google_news title=”” keyword=”CO2 adsorption” num_posts=”5″ blurb_length=”0″ show_thumb=”left”]